Q: The following chemical reaction takes place in aqueous solution: Fe, (so,),(aq)+3…
A: Process to write net ionic equation 1. Break the aqueous species into ions in a Balance reaction…
Q: ) Balance the following equations and state whether they are redox or non-redox reactions. For the…
A: Redox reaction is the reaction in which oxidation that is loss of electrons as well as reduction…
Q: Identify the base in the following acid-base reaction: H,PO, (aq) + H,O(1)→ HPO, (aq) + H,O*(aq)…
A: To determine whether a substance is an acidor a base, before and after the reaction, count the…
Q: Balance the redox reaction occurring in basic solution. ClO−(aq)+Cr(OH)4−(aq)→CrO42−(aq)+Cl−(aq)
A: The redox reaction is: ClOaq-+CrOH4aq-→CrO4aq2-+Claq- Step 1: Separate the two half-reactions.…
Q: (aq)+Sn(s)
A: Cell reaction : Zn2+(aq) + Sn(s) ----> Zn(s) + Sn2+(aq) Reduction half reaction: Zn2+(aq) + 2e-…
Q: The following chemical reaction takes place in aqueous soluti ZnCl,(aq)+2 KOH(aq) →Zn(OH),(s)+2…
A: To write the net ionic reaction of the below reaction. ZnCl2aq+2KOHaq→ZnOH2s+2KClaq
Q: Al(OH),(s) Al°* (aq) + 30H (ag) x x.=. = +, log. cos, K = [AP][oH]³ 10
A: The equilibrium constant (K) is the ratio of the equilibrium concentration of the products over the…
Q: Hydrogen peroxide solutions can easily be decomposed by dust, trace amounts of metals or light. It…
A: The balanced reaction taking place is given as, Given: Concentration of KMnO4 = 0.372 M And volume…
Q: Compose the balanced reaction equation for the following reaction occuring in acidic aqueous medium.…
A: Given reaction: As “P” oxidation state changes from 0 to +5, it is undergoing oxidation by losing…
Q: Based on the field of civil engineering, choose particular equipment or device used, and enumerate…
A: Chromium is a metal frequently used to help alloys resist corrosion. ... Nickel is a metal often…
Q: lodine pentoxide is used as a reagent to determine the amount of carbon monoxide present in a…
A: Balanced equation for the reaction of iodine pentoxide, I2O5(g) with CO(g) is: I2O5(g) + 5CO(g)…
Q: Which of the following aqueous solutions, if any, is expected to be comprised essentially of all…
A: The following solution gives all ions from the solute rather than a mixture of ions or molecules.
Q: Calculate the concentration of CO32- in an aqueous solution of 0.123 M carbonic acid, H,CO3 (aq).…
A: Given initial molarity of H2CO3 = 0.123 M Concentration of CO32- = [CO32-] = ? Acid dissociation…
Q: The Kc for the reaction 2A (aq)=3 B (aq) is 0.085. What is Kc for the reaction 3 B (aq) = 2A (aq)?
A: Welcome to bartleby !
Q: -. For the reaction Fe* (aq) + Co²+ (aq) → Fe²*(aq) + Co³* (aq), which is the reducing and oxidizing…
A: In a chemical reaction- The oxidising agent is that chemical species which is present in higher…
Q: The following chemical reaction takes place in aqueous solution: Mn(NO3),(aq)+2 KOH(aq)…
A:
Q: Balance the following equation and write the sum of the coefficients of the reactants and products…
A: A balanced chemical equation is an equation which has an equal numbers of atoms on both side…
Q: Balance the reaction in both in acid solution and in basic solution. CrO42 (aq) + SO3² (aq) →…
A:
Q: a dilution is needed to prepare a standard of 10 mg Zn/L from 0.5g Zn/L. To carry out the dilution…
A: We will discuss which bulb pipette and which corresponding volumetric flask you would choose to…
Q: Equivalent weight is the molar mass divided by the number of reacting units (H+ or OH .ionizing) for…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: following chemical reaction takes place in aqueous solution: 2 AgF(aq)+ (NH,), S(aq)Ag,S(s)+2…
A: Given 2 AgF(aq) + (NH4)2S(aq) → Ag2S(s) + 2NH4F(aq)
Q: Potassium permanganate solutions used in oxidation-reduction titrations are often standardized…
A:
Q: The fertilizer ammonium sulfate, (NH4)2SO4, is prepared by the rxn of ammonia with sulfuric acid.…
A: The balanced chemical equation of a reaction has equal number of atoms and balanced charges on…
Q: The following chemical reaction takes place in aqueous solution: SnCl,(aq)+2KOH(aq)…
A:
Q: 1 drop 0.2m (u(NO3)2 +9 drops Hau For electrolyt, Sdn confir
A: Given: 1 drop 0.2 M Cu(NO3)2 + 9 drops water 2 drop 0.2 M Zn(NO3)2 + 14 drops water To Calculate:…
Q: Identify the acid in the following acid–base reaction.
A: The component which donates a H+ ion in the reaction is known as acid and the component which…
Q: To determine , by gravimetric analysis, the concentration of barium ions (Ba2+) in a given solution,…
A:
Q: Consider these compounds: A. Cas B. Ag,Cro4 C. Fes D. Zn(OH)2 Complete the following statements by…
A: Solubility of compounds can be compared on the basis of their Ksp ( solubility product constant )…
Q: The pH of an aqueous solution of 0.2170 M hydrosulfuric acid, H,S (aq), is
A:
Q: The pH of an aqueous solution of 0.1130 M hydrosulfuric acid, H2S (aq), is ____ .
A:
Q: The following chemical reaction takes place in aqueous solution: 2 Fe(NO;),(aq)+3 Na,S(aq)…
A:
Q: The following chemical reaction takes place in aqueous solution: 2 AgNO3(aq)+Na,S(aq)…
A: To write the net ionic equation the following steps are involved 1. Write the balanced equation 2.…
Q: Complete and balance the following redox reaction in basic solution ClO₂(g)→ClO₂⁻(aq)+ClO₃⁻(aq)
A:
Q: Equivalent weight is the molar mass divided by the number of reacting units (H+ or OH ionizing for…
A: Equivalent weight of an element is defined as the gram atomic weight divided by the n-factor
Q: Chlorine gas is used in the production of many other chemicals, including carbon tetrachloride,…
A: Given: Volume of chlorine gas produced at STP = 5.9 ✕ 105 L Known: Molar mass of NaCl = 58.44 g/mol
Q: 12.) A 2.00g sample of limestone was dissolved in hydrochloric acid and all the calcium present in…
A:
Q: When sodium acetate, CH3COONa(s), is added to CH3COOH (aq), why does the pH of the solution…
A: The dissociation of sodium acetate in aqueous solution of acetic acid can be shown as follows-
Q: The Kc for the reaction 2A (aq) ⇌ 3 B (aq) is 0.085. What is Kc for the reaction 3 B (aq) ⇌ 2A (aq)?
A: Characteristics of Kc : If reverse the equilibrium reaction, Kc value is reciprocated. In the given…
Q: K,SO,(aq) a.
A: Electrolyte is defined as a chemical compound which converts into ions and conducts electricity when…
Q: The quantity of antimony in a sample can be determined by an oxidation–reduction titration with an…
A: The question is based on the concept of quantitative estimation. it involves estimation of the…
Q: e) OO0
A: Reaction of acid with base gives water and salt. Reaction is called as neutralization reaction. In…
Q: Write a balanced net ionic equation for a redox reaction that results in the formation of potassium…
A: Here we have to write balanced net ionic equation for above given reaction-
Q: Balance the chemical equation: In aqueous solution: ZnO(s) + H+(aq) ----> ?
A:
Q: Of what use is the acid in the isolation of the sought-for constituent in determination of Sulfur as…
A: Barium is a metal that is alkaline metal and is divalent in nature, Barium Sulfate is actually a…
Q: Hydrogen peroxide solutions can casily be decomposed by dust, trace amounts of metals or light. It…
A: Given: Density of hydrogen peroxide solution = 1.11 g cm-3 = 1.11 g/mL Volume of H2O2 used for…
Q: it is determined through a titration experiment that the molarity of acetic acid, CH3COOH, in…
A:
Q: You are required to standardize a dilute solution of sulphuric acid, and undertake the following…
A: The reactants given are Na2B4O7 and sulphuric acid i.e. H2SO4.
Q: For a precipitation reaction to be useful in a gravimetric analysis, the product of the reaction…
A: Kc > 1 for the product of the reaction must be insoluble and reaction is spontaneous in the…
Q: Give the oxidation number of the underlined elements in each of the following chemical species: (a)…
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: The following chemical reaction takes place in aqueous solution: MgCl,(aq)+2 KOH(aq)…
A: We have to predict the net ionic equation.
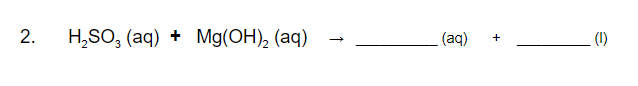
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- The iron content of hemoglobin is determined by destroying the hemoglobin molecule and producing small water-soluble ions and molecules. The iron in the aqueous solution is reduced to iron(II) ion and then titrated against potassium permanganate. In the titration, iron(ll) is oxidized to iron(III) and permanganate is reduced to manganese(II) ion. A 5.00-g sample of hemoglobin requires 32.3 mL of a 0.002100 M solution of potassium permanganate. The reaction with permanganate ion is MnO4(aq)+8H+(aq)+5Fe2+(aq)Mn2+(aq)+5Fe3+(aq)+4H2O What is the mass percent of iron in hemoglobin?The carbon dioxide exhaled in the breath of astronauts is often removed from the spacecraft by reaction with lithium hydroxide 2LiOH(s)+CO2(g)Li2CO3(s)+H2O(l) Estimate the grams of lithium hydroxide required per astronaut per day. Assume that each astronaut requires 2.50 103 kcal of energy per day. Further assume that this energy can be equated to the heat of combustion of a quantity of glucose, C6H12O6, to CO2(g) and H2O(l). From the amount of glucose required to give 2.50 103 kcal of heat, calculate the amount of CO2 produced and hence the amount of LiOH required. The H for glucose(s) is 1273 kJ/mol.Tartaric acid, H2C4H4O6, is a diprotic acid that naturally occurs in the production of apple wine. Emma was tasked to find out the acid content in a 50.0 mL wine sample by titrating it with a standardized solution of NaOH. She was able to establish the working concentration of the NaOH solution by using 17.61 mL of NaOH to titrate a primary standard of KHP that weighed 1.501 g. The grams and %w/w of Tartaric acid present in the 50.0 mL wine sample were then determined by titrating a 12.50 mL aliquot using 21.78 mL of the standardized NaOH solution. H2C4H4O6 (aq) + 2NaOH (aq) ↔ Na2C4H4O6 (aq) + 2H2O (l) Assume that the density of the wine sample is 1.000 g/mL MM of KHP = 204.22 g/mol MM of H2C4H4O6 = 150.087 g/mol What is the molar concentration of the prepared NaOH solution?…
- Whilst working in the pharmacy aseptic unit you are asked to prepare 5 litres of a solution containing potassium ions 1 mmol/mL. What weight of potassium chloride is required?The fish canning industry has a waste effluent with the specification as follows: BOD5 = 150 mg/L, COD = 230 mg/L, CaCO3 = 65 mg/L, pH = 7,0, Odor = 11 (TON, where every 1 mg/L ammonia-N equals to 2 TON), Oil = 300 mg/L. Give recommendation of what kind of treatment (physical and chemical steps) is required to treat the wastewater, and why do you select those steps.Tartaric acid, H2C4H4O6, is a diprotic acid that naturally occurs in the production of wine. Jose was tasked to find out the acid content in a 100.0 mL wine sample by titrating it with a standardized solution of NaOH. He was able to establish the working concentration of the NaOH solution by using 35.21 mL of it to titrate a primary standard of KHP that weighed 3.001 g. The grams and %w/w of Tartaric acid present in the 100.0 mL wine sample were then determined by titrating a 25.00 mL aliquot using 43.56 mL of the standardized NaOH solution. H2C4H4O6 (aq) + 2NaOH (aq) ↔ Na2C4H4O6 (aq) + 2H2O (l) Assume that the density of the wine sample is 1.000 g/mL MM of KHP = 204.22 g/mol MM of H2C4H4O6 = 150.087 g/mol What is the molar concentration of the prepared NaOH solution?…
- Come up with a dilution scheme for a test solution starting from 1000 μg/mL Mn stock solution so that the concentration of permanganate in the solution just prior to measurement is approx. 20 μg/mL MnO4-. Carefully read the units!In the standardization of HCl using pure anhydrous sodium carbonate as the primarystandard for methyl orange as an indicator, 1.0 mL HCl was found to be equivalent to 0.05gof sodium carbonate (MW =106). The normality of HCl is:The Kc for the reaction 2A (aq) ⇌ 3 B (aq) is 0.085. What is Kc for the reaction 3 B (aq) ⇌ 2A (aq)?
- Malic acid (C4H6O5) is a dicarboxylic acid that naturally occurs in the production of bignay wine. Maria was tasked to find out the acid content in a 200.0 mL wine sample by titrating it with a standardized solution of KOH. He was able to establish the working concentration of the KOH solution by using 39.12 mL of it to titrate a primary standard of KHP that weighed 3.021 g. The grams and %w/w of Malic acid present in the 200.0 mL wine sample were then determined by titrating a 20.00 mL aliquot using 48.95 mL of the standardized KOH solution. C4H6O5 (aq) + 2KOH (aq) ↔ K2C4H4O5 (aq) + 2H2O (l) Assume the density of the wine sample is 1.000 g/mL MM of KHP = 204.22 g/mol MM of C4H6O5 = 134.0874 g/mol ? What is the molar concentration of the prepared KOH solution?…Malic acid (C4H6O5) is a dicarboxylic acid that naturally occurs in the production of bignay wine. Maria was tasked to find out the acid content in a 200.0 mL wine sample by titrating it with a standardized solution of KOH. He was able to establish the working concentration of the KOH solution by using 39.12 mL of it to titrate a primary standard of KHP that weighed 3.021 g. The grams and %w/w of Malic acid present in the 200.0 mL wine sample were then determined by titrating a 20.00 mL aliquot using 48.95 mL of the standardized KOH solution. C4H6O5 (aq) + 2KOH (aq) ↔ K2C4H4O5 (aq) + 2H2O (l) Assume the density of the wine sample is 1.000 g/mL MM of KHP = 204.22 g/mol MM of C4H6O5 = 134.0874 g/mol What is the molar concentration of the prepared KOH solution?…A 12.5 mL portion of a base solution of an element from Group 2A is diluted to 500 mL, and the resulting concentration of this solution is determined to be 0.125 M. What is the normality of the original solution?











