Q: ie vapor pressure a
A:
Q: 2. Lone pair occupies a hybridized p-orbital allowing it to delocalization to a nearby side-way over...
A:
Q: Show the calculations on how to prepare 2.5 L of 1.5 M buffer with pH=10.0 from CH3NH3Cl and CH3NH2....
A: Here we have determine how to prepare 2.5 L of 1.5 M buffer solution with pH= 10 from CH3NH3Cl and C...
Q: What is the volume of the following sample of gas at STP? 0.236 H2 Express the volume to three si...
A:
Q: Type of delocalization that occurs in a conjugated system? A pi electron delocalization B lone pair ...
A:
Q: Which strain occurs when carbon-carbon bonds do not meet the 109.5 degree angle? (A Angle strain (B)...
A: A molecule experiences strain only when chemical structure of a molecule undergoes some stress which...
Q: Which of the following acids has the weakest conjugate base? Chlorous acid, Ka = 1.1 x 10-2 ...
A: The strongest an acid , the weakest is its conjugate base. The acid which has highest Ka value is s...
Q: 1. How many MOLES of dinitrogen monoxide are present in 1.90 grams of this compound ? moles. 2. How ...
A: Mole concept: The number of molecules or atoms present in the one mole of the substance is equal to ...
Q: Balance the given molecular equation using redox (electron-transfer method). Identify the element ox...
A:
Q: Of the initial radte ol a tion are yiven in the labie belo N2 H2 initial rate of reaction 0.776M 2.2...
A:
Q: (d) (e) (f) CH3CHCOH CI NH2
A: Answer of the question given below,
Q: BUFFERS CH3COOH and CH3COONa NH,OH and NH&Cl H3PO4 and NaH2PO4 H2CO3 and NaHCO3 NH3 and (NH4)2CO3
A: Buffers are the solutions which resist the pH change. Acidic buffer is the mixture of weak Acid/bas...
Q: Activity 2: Complete the table below. Lewis Bond Molecular Polarity of Molecule Structure EN Polarit...
A:
Q: HO In reference to chiral carbon A, what is the configuration of the structure? (A)S B)R
A: Answer is given below:-
Q: Aluminum oxide, Al203, is used as a filler for paints and varnishes as well as in the manufacture of...
A:
Q: Indicate whether or not each of the structures is considered to be aromatic. NH A Which is true? A i...
A: We have to find whether these compounds are aromatic or not.
Q: The acid HOCI (hypochlorous acid) is produced by bubbling chlorine gas through a suspension of solid...
A: The correct expression of equilibrium constant is given below
Q: Consider the following reaction at equilibrium: C(s) + H20(g) = CO(g) + H2 (g) Predict whether the r...
A:
Q: Draw the major thermodynamic and kinetic products of the reaction. 1 eq. HBr thermodynamic product k...
A:
Q: E2B.3(a) The constant-pressure heat capacity of a sample of a perfect gas was found to vary with tem...
A:
Q: e boiling point of
A: Since you have asked multiple questions, we will solve the first question for you. If you want any s...
Q: 1) O, 2)Me,S 1)KMNO,/heat 2) H,O*(aq) NBS RO-OR 1) BHSTHF 2) NAOH/ROOR H,O°(aq)
A:
Q: Calculate the mass in milligrams of 4.67 x 104 formula units of Bi(NO3)3 5H,0. Enter your answer in ...
A:
Q: Give the conjugate base of H2O.
A: Acid is substance which release hydrogen ions and base is substance which release hydroxyl ions when...
Q: Example 3 A sample of zinc anode corrodes uniformly with a current density of 4.27 x 107 A/cm² aqueo...
A: Step = 1st Solution The following reaction: Zn→ Zn 2+ + 2e Corrosion Rate = (...
Q: How does the following affect the basicity of compounds? Explain clearly and thoroughly. a. Bond le...
A: Basicity is the number of protons given by an acid when it is dissolved in water. Acidic strength or...
Q: 2. Given a reaction in acidic medium: MnO, (aq) + Fe" (ao) → Fe3 (aq) + Mn2* (aq) a. What is oxidize...
A: Dear student since it is a multiple subparts questions according to the guidelines here I am solving...
Q: Balance the following equation with the smallest whole number coefficients. What is the coefficient ...
A:
Q: given a stock solution of 2.0% dextrose ,how would you prepare 10mL of the following solution? find ...
A: The volume of the solution can be determined by the relationship between the concentration and solut...
Q: K 3: STRONG OR WEAK! ions: Relate the properties of liquids to intermolecular forces. Tell whether e...
A: Intermolecular forces of attraction is the force that exist between two molecules.
Q: 30- 25 22.285 20- atm 15 10- 5- 50 100 150 200 250 300 seconds created Is BF, being created or destr...
A:
Q: Classify the diene as conjugated, isolated, or cumulated. Classify the diene. cumulated O conjugated...
A: The correct answer is given below
Q: A student calculated that the average neutralizing power of their antacid was 102 grams of acid neut...
A:
Q: Read carefully the given partial names in IUPAC alkane nomenclature and check the columns where they...
A:
Q: 3) Predict the products of the ff reagents upon ozonolysis (reductive-work up): A. В. - CH
A: Ozonolysis of alkene: One of the important reactions of alkene is ozonolysis. In the ozonolysis reac...
Q: When 0.200 mol of CACO,(s) and 0.300 mol of Ca(s) are placed in an evacuated, sealed 10.0-L containe...
A: ANSWER : Given chemical reaction is : CaCO3(s)⇔ CaO(s) + CO2(g)Given : PCO2= 0.220 atmV = 10.0 LT ...
Q: Some measurements of the initial rate of a certain reaction are given in the table below. [H.] 12 in...
A: Rate constant is proportionality constant of rate law expression, rate law is experimental analysis ...
Q: pH
A:
Q: Specify the formal charges (both sign and magnitude) on the atoms labeled a-c. H c/ H2C=CH-C: a b 1)...
A: Formal charge is an electric charge over the atom in molecules.
Q: 4
A: Given :- Cu-Ni alloy contains 64 wt% Cu and 36 wt% Ni. wt% of Cu in liquid is 54% and in solid is 3...
Q: A student carried out an experiment and used 20.00 mL of 0.0100 M NaOH to titrate the excess HCl in ...
A: We are given an amount of NaOH being used to titrate the acid in an antacid reaction. We have to fin...
Q: Determine the heat of reaction ( in k) for the following reaction. Mg(s) + 2HCI(aq)- MgCl2(aq) + H2l...
A: We have to find heat of reaction.
Q: QUESTION 1 After removing the pH meter electrode from the 3.0M KCL solution, the glass electrode is ...
A:
Q: What is the volume of the following sample of gas at STP? 7.90 mol SF6 (Express the volume to th...
A: 1 mole of substance is equal to avogadro's constant which is equal to 6.022 * 10^ 23 atoms.
Q: Questions: 1) Explain the molecular basis of the reaction of bromine water with phenol. 2) What is/a...
A: These given reactions are the preliminary tests of functional group’s detection in organic chemistry...
Q: Example 5 A sample of zinc corrodes uniformly with a current density of 4.2 x 10° A/cm in solution. ...
A:
Q: Dehydration of alcohol is an example of
A: Dehydration of alcohol is an example of ?
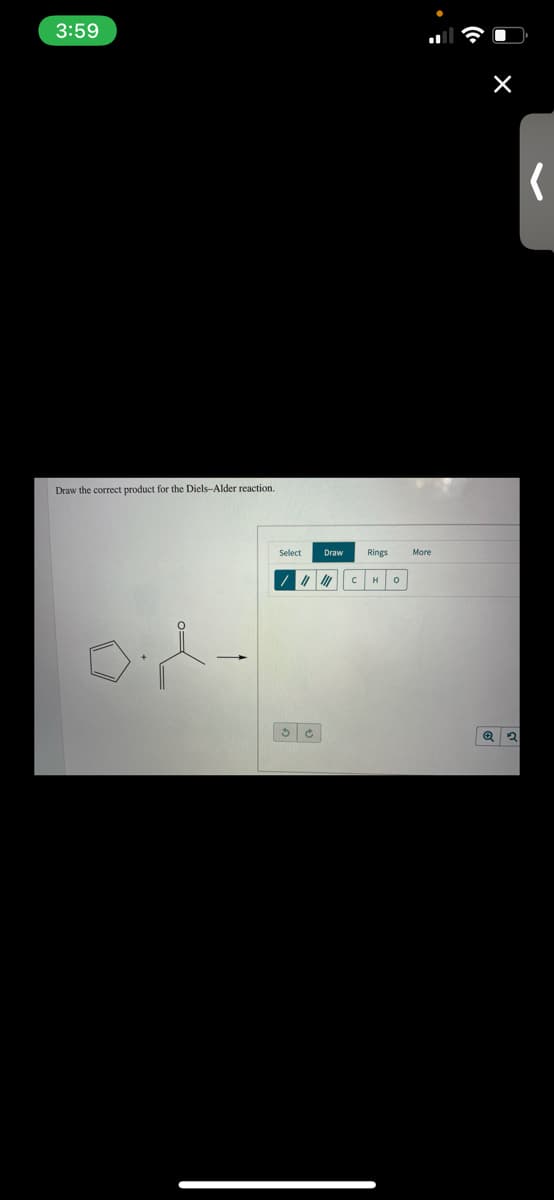
Q: Deconstruct the given Diels–Alder adduct. Draw the reactants, in any order, that would be needed to ...
A:
Q: Rank the isomers in order of increasing heat of formation. Highest heat of formation Lowest heat of ...
A: The alkenes are compound which composed of carbon and hydrogen and contains at least one double bond...
Q: At present, automobile batteries are sealed. When lead storage batteries discharge, they produce hyd...
A: Molar mass of hydrogen = 1.00794 g/mol Molar mass of H2 = 1.00794 × 2 = 2.01588 gram/mole Given Mas...

Step by step
Solved in 2 steps with 2 images

- A compound of formala ( C20H29NO) absorbs 4 molar equivalents of hydrogen. How many rings does it contain ? select one : A) 3 B) 2 C) 1 D) 4Diels–Alder reaction of a monosubstituted diene (such as CH2=CH–CH=CHOCH3) with a monosubstituted dienophile (such as CH2=CHCHO)gives a mixture of products, but the 1,2-disubstituted product oftenpredominates. Draw the resonance hybrid for each reactant, and use thecharge distribution of the hybrids to explain why the 1,2-disubstitutedproduct is the major product.Compound MM (g/mol) Volume Used (mL) d (g/mL) Weight Used (g) Moles Used Equivalence 9-anthracenemethanol 208.26 g g/mol N/A N/A 0.065 g (given) 0.0003 (rounded) 1 N-methylmaleimide 111.10 g/mol N/A N/A 0.104 g (given) 0.0003*3 = 0.0009 (rounded) 3 Diels-Alder Adduct/Product 319.4 g/mol N/A N/A 1 Please figure out the theoretical yield and moles of the product based on the table and image.
- How did you convert the c to k that's my only question like 25.5c to k and you got 298.65?Assign a priority order (from least to highest priority) to each of the folliowing sets of groups: I. -CH(CH3)2 II. -CH3 III. -H IV. -NH2 A. III, IV, II, I B. IV, I, III, II C. III, II, IV, I D. III, II, I, IVAssign a priority order (from least to highest priority) to each of the following sets of groups: I. -OCH II. -NH(CH3)2 III. CH2NH2 IV. -OH A. IV, I, III, II B. III, II, IV, I C. II, III, IV, I D. III, IV, I, II
- According to the conventions above, what is the sign ( + or ) of the P.E. change (H) for Rxn 3?Calculate the Keq for the following reaction, given that Eo Cd2+/Cd = -0.403 V and Eo Zn2+/Zn = -0.763. Overall rxn: Cd2+ + Zn <-> Zn2+ + CdRank the following groups in order of decreasing priority. −CH=CH2, −CH3, −C≡CH, −H
- 1. The trans effect trend is shown below: NO2- > Cl- > py Which sequence of reactions would lead to the synthesis of cis-[PtCl2(NO2)(py)]-? Pick one from the choices belowDraw the resonance structures of [CH3C(OCH3)2]+ and identify its minor and major contributors.In a Diels-Alder expirment, what does a 73% yield tell you



