Q: CH3 is exposed to excess conc. HBr at high temperature
A:
Q: 1. Which of these gases would behave the least ideally? (O2, N2, and HF)
A: "As you didn't give me a question number I'm answering first question only... Please upload again an...
Q: The equilibrium constant Kp for the interconversion of phosphorous pentachloride and phosphorous tri...
A:
Q: Hydrogen phosphate ion (HPO4 ^2-) is also an amphoteric molecule. Give the hydrolytic reaction for...
A: We know that, Hydrolytic reaction is hydrolysis reaction that is a type of chemical reaction in whic...
Q: Most of the ultraviolet absorption of proteins at 280 nm is due to their content of a. Glutamate b....
A:
Q: Please calculate the molar solubility of HgBr2 (K sp = 6.20 x 10-20 ) in a 2.58 molar FeBr3 solution...
A: Given :: Ksp of HgBr2 = 6.20×10-20 [FeBr3] = 2.58 M molar solubility of HgBr2 = ?
Q: Part A lodine atoms will combine to form I2 in liquid hexane solvent with a rate constant of 1.5 x 1...
A:
Q: How does how the molecular identity of the element change in space and/or time? Be sure to make the ...
A: A name that uniquely identifies a compound, molecule or substance is known as chemical identity. In ...
Q: A solution is saturated at 25 g per 100 g of solution; this solution has 22 grams in 100 g of soluti...
A: Solutions are classified as unsaturated, saturated,super saturated depending upon the amount of solu...
Q: Describe the cleaning action of soap
A:
Q: Review Topics] (References] Either the reactant (X) or the major organic product is missing from the...
A:
Q: Identify the stronger acid between each pair and justify your answer. 1. HI аnd HF 2. ΗΝΟ, and HNO, ...
A: Given : Identify the stronger acid between each pair 1. HI and HF 2. HNO3 and HNO2 3. HBrO₂ and HC...
Q: For the following endothermic reaction at equilibrium: 2 SO3 (g) 2 SO2(g) + O2(g) Decide if each ...
A: Given endothermic reaction is : 2 SO3 (g) <---------> 2 SO2(g) + O2(g) Decide if each of the ...
Q: using magnesium hydroxide as an example, provide definitions for solubility product and saturation r...
A: The solubility product constant is the equilibrium constant for the dissolution of a solid into an a...
Q: What is the pH of a solution of 2.0 x 10–8M Ca(OH),?What color would you expect it to have with cabb...
A:
Q: How many grams of O2 gas occupy 19.0 L at STP? Enter your answer in the provided box.
A:
Q: Also please help me with Question 4.
A: 1) Given: %(w/w) of acetic acid = 5 % Density of vinegar = 1.05 g/mL. And volume of vinegar solution...
Q: 8Ca → 3. 65 Sc 65 4. Be + He C + -> P + H 31 15 32 15 5. 16S + Hе 35 6. 1ci +
A: Dear student, As you have asked multiple question but according to guidelines we will solve first...
Q: Draw tructural orm or he roduct ormed pon ydroboration/oxidation f he lkene below. CH3 • Use wedge a...
A: Here we have to write the product formed in the Hydroboration and /oxidation of 1-methyl-1-cycl...
Q: Explain the MO theory using the formation of H2.Use the information to construct the MO diagram of h...
A: Atomic orbitals of atom combine together to form molecular orbitals. If the bond order is greater t...
Q: Why composite biomaterials are preferred over ceramics, explain with examples.
A: Being able to use artificial intelligence to identify and automatically respond to data breaches is ...
Q: RECRYSTALLIZATION Principle/s Involved: What is the principle behind the process?
A: Recrystallization is a process of purification using proper solvent.
Q: TReview TOpics] [References Draw a structural formula for the major product of the reaction shown. C...
A:
Q: Answer in full sentence: What do you predict would have occurred in the reaction baking soda + vine...
A: The reaction is: Sodium bicarbonate and acetic acid reacts to carbon dioxide, water and sodium aceta...
Q: (a) NaCN (d) NEt H2SO4, 0°C dropwise EINH3 + (b) MEOH (exs.) (e) TSOH, toluene heat 1) Ph3P Br (c) 2...
A:
Q: Calculate AG° (in kJ) for the following reaction: 31, (s) + 2Fe(s) →2FE** (aq) +6I¯(aq) 232 O -111 O...
A:
Q: Consider the following system at equilibrium where Kc = 5.10×10-6 and AH° = 268 kJ/mol at 548 K. NH4...
A: According to Le Chatlier's principle, a system at equilibrium tends to remain in equilibrium. If any...
Q: Part A Concentration-time data for the conversion of A and B to D are listed in the following table:...
A: The data given is,
Q: I need help calculating some of the following values please. reaction equation: MnO4− + 5 Fe2+ + 8 H...
A:
Q: [Review Topics] [References] An alkene having the molecular formula C11H20 is treated sequentially w...
A:
Q: Br2+ Cl2: O 2.56 x 104
A: Chemical equilibrium is defined as the stage at which both reactant and product are in equilibrium.
Q: For the following solutions, estimate the buffer intensity (E) at a pH of 5. Which solution is the b...
A: Since you have asked multiple questions, we will solve the first question for you. If you want any s...
Q: Relerences) Draw a structural formula for the major organic product of the following reaction: CH2CI...
A:
Q: CH3 ters S. te)
A:
Q: A. 3. FeCls + K;Cr20, + FeSO, + )H;SO4 → Fer(SO4)s + Cra(SO4)3 + NO + K2SO4 + H20 HNO; + H25 H20 SO,...
A: In the balanced redox reactions (below), the total electrons on both the reactant and product side h...
Q: Part A The following data show the rate constant of a reaction measured at several different tempera...
A:
Q: References] Draw a structural formula for the major organic product of the reaction shown. =CHCH3 HB...
A:
Q: 4) Would you expect cis-1,2-dimethylcyclohexane and trans-1,2-dimethylcyclohexane to have a good sep...
A: TLC is a type of chromatography used in separation of non volatile Mixture. Its stationary phase is ...
Q: A. GIVE THE IUPAC NAME OF TH ОН b) (CH3)3СОН c) CH3CH2CH2OH d) OH H3C CH3
A: Following steps are required for IUPAC naming. 1-Find and name the longest continuous carbon chain t...
Q: Some chemical reactants are listed in the table below. Complete th oxidation state of highlighted at...
A: Oxidation state is the total number of electrons that an atom either gains or loses in order to form...
Q: CH3 is mixed with H,SO4 at high temperature OH
A:
Q: e. Some have proposed releasing ethane, C2H6, or other hydrocarbons into the stratosphere as a means...
A: Given: Releasing ethane in the stratosphere to reduce the concentration of chlorine radicals.
Q: calculate the titration curve of a 0.01 m aqueous solution of NH3 with 0.03M HBr. A minimum of five ...
A: Ammonia reacts with HBr as per the following reaction: NH3+HBr↔NH4++Br-
Q: A student ran the following reaction in the laboratory at 70 K: H2(g) + I2(g) 2HI(g) When she introd...
A:
Q: pressure
A:
Q: Determine the pH of 5.416 x 10-8 M of HCl. (use the systematic treatment) Determine the pH of 7.814 ...
A: 1) Concentration of HCl solution = [HCl] = 5.416 x 10-8 M pH of the solution = ? Note: HCl is a st...
Q: Order each of the sets of compounds with respect to Sn1 reactivity (1 = fastest). CH3 .CH2BR .CH21 C...
A: in sN1 mechanism carbocation intermediate is formed...
Q: Calculate the pH at the equivalence point for each titration: a) in the burette 1.0 M HCl. in the be...
A: a) In this case, HCl will react with the basic component of the buffer, i.e. acetate ions.
Q: calculate for the standardized concentration of sodium hydroxide (MM of KHP = 204.2 g/mol) using t...
A:
Q: Calculate the pH at the equivalence point in titrating 0.028 M solutions of each of the following wi...
A: a)
Step by step
Solved in 2 steps with 2 images

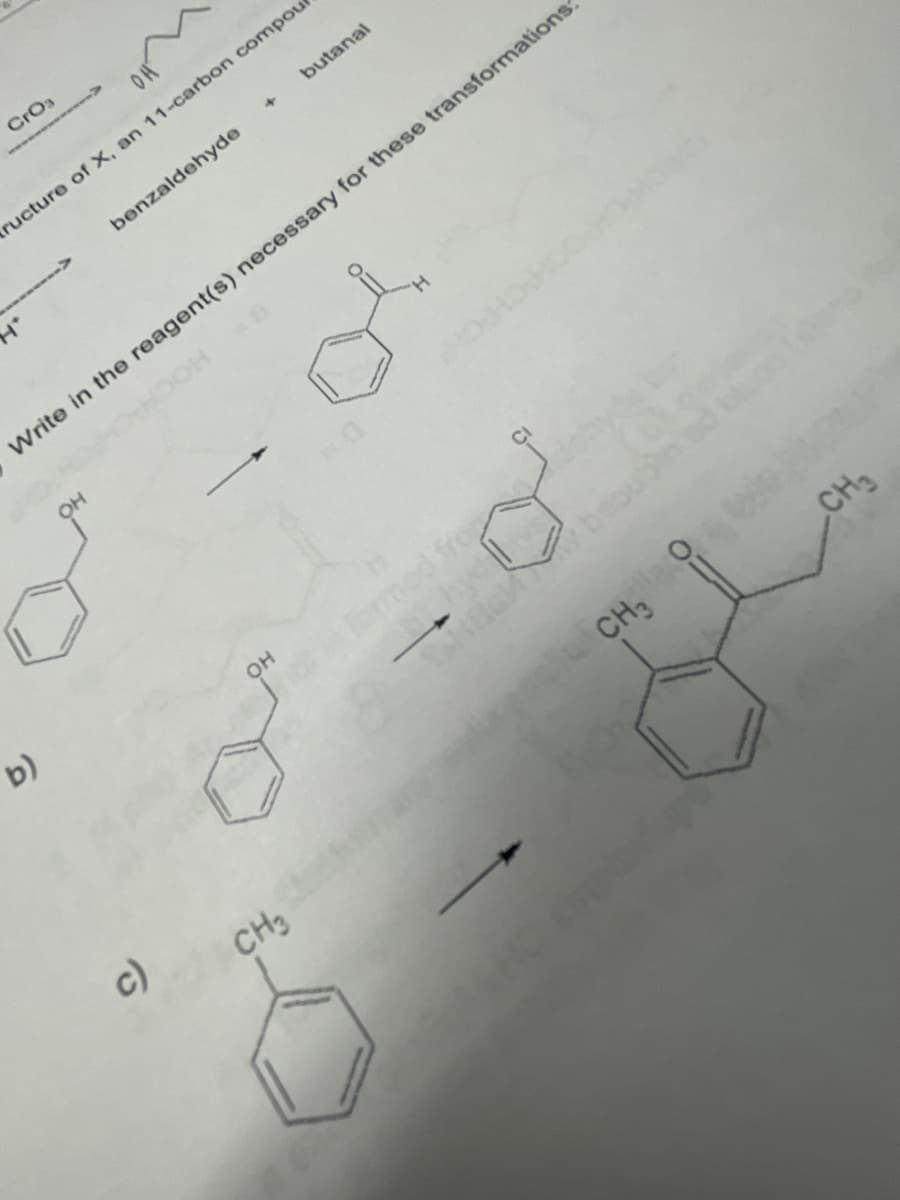
- Provide the suitable reagents to effect the following transformations. I specifically need help on sub-parts d, e, f, g, h, and i.Draw the product Y of the following reaction sequence. Y was anintermediate in the remarkable synthesis of cyclooctatetraene by RichardWillstatter in 1911.Answer the following question about curcumin, a yellow pigmentisolated from turmeric, a tropical perennial in the ginger family and aprincipal ingredient in curry powder. Most enols, compounds that contain a hydroxy group bonded to a C=C, are unstable and tautomerize to carbonyl groups. Draw the keto form of the enol of curcumin, and explain why the enol is more stable than many other enols.
- Provide the reagents necessary to affect each transformationRank the following compounds in order of increasing reactivity in a substitution reaction with −CN as nucleophile.Draw the product X of the following SN2 reaction. X was a key intermediate in the synthesis of rizatriptan, a drug introduced in 1998 for the treatment of migraines.
- Rank the compounds in each group in order of increasing reactivity in nucleophilic acyl substitution. C6H5CO2CH3, C6H5COCl, C6H5CONH2Draw a stepwise mechanism for the conversion of hex-5-en-1-ol to the cyclic ether A.Provide the reagents necessary to achieve each transformation. Draw the intermediate structure(s).
- Draw a stepwise mechanism for the attached reaction that illustrateshow two substitution products are formed. Explain why 1-bromohex-2-ene reacts rapidly with a weak nucleophile (CH3OH) under SN1 reactionconditions, even though it is a 1 ° alkyl halide.Label the alkene in each attached drug as E or Z. Enclomiphene is one component of the fertility drug Clomid. Tamoxifen is an anticancer drug. Clavulanic acid is sold in combination with the antibiotic amoxicillin under the trade name AugmentinWhat epoxide and organometallic reagent are needed to synthesize each alcohol?

