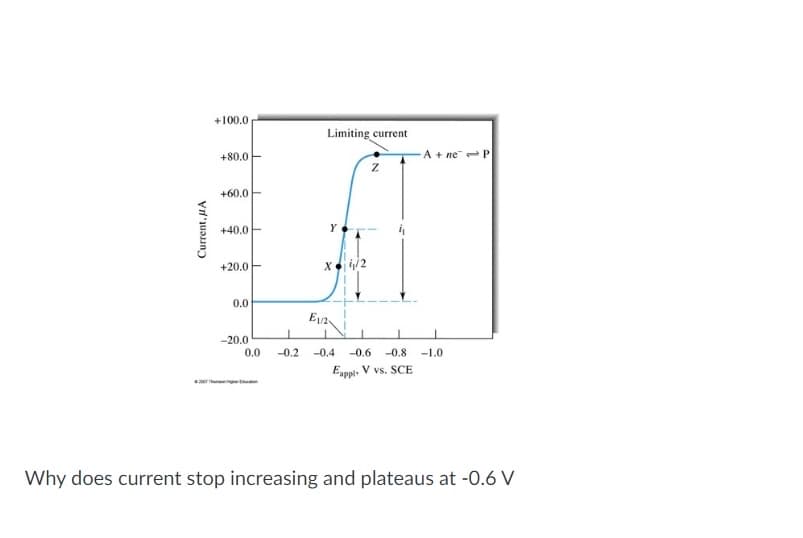
Current, A +100.0 +80.0 +60.0 +40.0 +20.0 0.0 -20.0 0.0 Limiting current Y X 4/2 E1/2 Z A+ ne P 1 -0.2 -0.4 -0.6 -0.8 -1.0 Eappl. V vs. SCE Why does current stop increasing and plateaus at -0.6 V
Q: For the chemical reaction 2 AgNO3 + Na₂ CrO4 Ag₂ CrO4 + 2 NaNO3 what mass of silver chromate is…
A:
Q: Macmillan Learning Plavix is used to treat acute coronary syndrome. Patients with a recent history…
A:
Q: Hh.119. Find the pH of a solution that is 9.0×10−2 M in H2CO3 express answer to two decimal places
A: Given Concentration of H2CO3 = 9.0 x 10-2 M
Q: > Calculate the percent of acetic acid in a 20 mL vinegar sample. Titration was used, and it took…
A: Given data is Volume of the vinegar sample = 20 mL Volume of NaOH used in the titration= 35.19 mL…
Q: For Q3, write the balanced equation, construct your pathway, and write out the calculation. Use the…
A: Given that Sodium hydroxide reacts with sulfuric acid to form sodium sulfate and water. Molarity of…
Q: VALENCE ELECTRONS FORMULA FROM EACH ATOM H₂ HCI H₂O2 TOTAL # OF VALENCE ELECTRONS LEWIS DOT…
A: Since you asked multiple questions so as per Q&A guidelines of portal I solved first three parts…
Q: 1. Draw the Full Lewis chemical structure of a simple ester of your choice containing exactly five…
A:
Q: Select the major product from the reaction sequence shown. ميل (A) 1) Me₂NH, [TSOH], -H₂O 2) 3) H3O+…
A: enamine is an unsaturated compound derived by the condensation of an aldehyde or ketone with a…
Q: When you cool down water to 10°C, Kw 2.9 x 10-15, as compared to the value of Kw at 25°C which is 1…
A:
Q: Flame Emission Spectra Step 1: Record the color of each flame in the table below NaCl CaCl₂ NiCl₂…
A: Flame emission spectra is also called as atomic emission spectra. Lines produced by each element in…
Q: Question 3 Hexane(C6H14) combusts with oxygen to produce CO2 and steam. If 550 g of H2O(g) are…
A: Given, Hexane( C6H14) combusts with oxygen to produce CO2 and steam ( H2O ) mass of H2O = 550 g.…
Q: What is the total number of oxygen atoms on the right-hand side of this chemical equation? 3HNO2 aq…
A: We need to count all the atoms on left side of the reactions as well as right side of reactions to…
Q: What volume of a 0.178 M barium hydroxide solution is required to neutralize 22.0 mL of a 0.171 M…
A: 1. we have to calculate the volume of barium hydroxide needed for neutralization
Q: O3(g) 2 SO₂(g) + O = 2.62 x 10-6 at 400° (g) at an initial press ilibrium. What will be
A: Given reaction is : 2 SO₃(g) ⇌ 2 SO₂(g) + O₂(g) Pressure equilibrium constant = Kp = 2.62 × 10⁻⁶…
Q: 4 Compare the activation energy in uncatalyzed reac- tions and in enzyme-catalyzed reactions. w…
A:
Q: ▾ Part B -C=N
A: IUPAC nomenclature used for the systematic naming of the organic molecules.According to IUPAC…
Q: 5. A given molecule has a one-dimensional conjugated ¹-electron networ that is distributed to…
A: 16. A given molecule has a one-dimensional conjugated -electron network that is distributed to…
Q: 78 Write the expected products of the acid hydrolysis of the following tetrapeptide:d to apofle sdi…
A:
Q: CH,(CH,),CHO OH
A: The IUPAC name of the compound is octanal. It has an aldehyde group at one end. The other end has 7…
Q: note: round all molar masses from the Periodic Table to two decimal places 3. Calculate the mass in…
A: “Since you have posted multiple questions with multiple subparts, we will provide the solution…
Q: The molarity (M) of an aqueous solution containing 0.065 mol of sucrose in 85 mL of solution is 4 A)…
A: Molarity is defined as moles of solute present in 1 L of solution. it is a method of expressing…
Q: 14 Draw the structures of L- and D-valine.
A: D/L- nomenclature is relative configuration system. It is mainly used for carbohydrates. In D/L…
Q: H روه Base Aldol Product the Mal NaOH H₂0 Condensation Product
A: Organic reactions are those in which organic reactant react to form organic products. In the given…
Q: The National Institute of Science and Technology (NIST) kinetics database lists the rate constant,…
A:
Q: A chemistry graduate student is given 450. mL of a 1.10M diethylamine ((C₂H), NH) solution.…
A:
Q: What is the IUPAC name for the compound shown? HHO H IUPAC name: -O-H
A: Rule of IUPAC- 1) Choose the longest carbon chain as parent chain. 2) Numbering start from those…
Q: For the chemical reaction shown, 2 H₂O₂(1) + N₂H4 (1) 4 H₂O(g) + N₂(g) determine how many grams of…
A: To solve this problem we have use the stoichiometry of the balanced chemical equation.
Q: Question 4. The titration curve for histidine is shown below. The pK values for the carboxy group…
A:
Q: Give the number of atoms of oxygen on the product side of the following balanced chemical equation.…
A: Answer: This question is based on law of conservation of mass which states that total mass of…
Q: Draw the OH- molecular orbital diagram
A: The basis orbital for H is 1s and for O are 2s and 2p. Z* for O 2s and 2p orbitals are similar so…
Q: Apply the significant figure rules to the final answer. The rules are attached. The final answer…
A: In multiplication/decision : Answer should not have the significant figures more than the original…
Q: does the (Z) have to be there or can you write the same thing without it
A: Answer: This is question is based on the stereoisomerism of alkene.
Q: Question 9 Dr. Cavendish prepared hydrogen in 1766 by the novel method of passing steam through a…
A:
Q: The concept of determining which reactant is limiting and which is in excess is akin to determining…
A: Answer: This question is based on the concept of limiting reagent. It is the reactant that exhaust…
Q: Draw the product of the reaction shown below. Use a dash or wedge bond to indicate stereochemistry…
A:
Q: Given the reversibility of the acid-catalyzed DAGUI er! obivo17 hydrolysis/Fischer esterification…
A:
Q: Sodium hydroxide reacts with sulfuric acid to form sodium sulfate and water. What is the molarity of…
A:
Q: 4. Predict the products (draw their structures) of the fatty acid with the reagents below. (a) (b) О…
A:
Q: CaCO3(s)→ CO2(g) + Cao(s) - The standard enthalpy of reaction for the reaction shown above is kJ. O
A:
Q: Polymers may be composed of thousands of monomers. Draw two repeat units of the polymer (dimer)…
A:
Q: Starting with benzene and using any other necessary reagents of your choice, design a synthesis the…
A:
Q: CH3 1 CH₂ 1 C=C ` 애 애 대로 a -CH-CH2-CH3 0-12-13 CH3-H
A: The structures of the given compounds are The IUPAC names of the given compounds are
Q: For the chemical reaction, 2 KI + Pb(NO3)2 Pbl₂ + 2 KNO3 what mass of lead(II) nitrate in grams is…
A: Suppose a balanced chemical equation aA +bB —>cC Then c mol of C needs b mol of B Where a, b,…
Q: TriCor (fenofibrate) is used to lower cholesterol levels in patients at high risk of developing…
A: An organic compound usually contains carbon atoms bonded to hydrogen atoms. Certain functional…
Q: A man who has recently moved to live in a city that sits at approximately 1,000 meters above sea…
A: The boiling point of a liquid is the temperature at which the substance transitions from a liquid to…
Q: whats the correct synthesis from one to two m Br [1] O 1. KOtBu/HOtBu; 2. a.Hg(OAc)2/H2O b.NaBH4 O…
A:
Q: Evaluate the commutator
A: It is based on the concept of the operator in quantum mechanics. Here we are required to find the…
Q: What is the systematic name for the given compound? Br (R)-2-bromobutane O(S)-2-bromobutane…
A: Since, According to Cahn-Ingold-Prelog rule- 1) More atomic number having more priority.2) If first…
Q: Draw the molecule on the canvas by choosing buttons from the Tools (for bonds), Atoms, and Advanced…
A: Chemical structure refers to the arrangement of atoms, bonds, and functional groups in a molecule.…
Q: How many stereoisomers are possible for 2,4-octadiene? A) 2 B) 4 C) 6 D) 8 E) No stereoisomers exist
A: Since, Isomer are those whose molecular formula same but structural formula is different. Thus,
Step by step
Solved in 2 steps

- The resistance of a conductivity cell containing a 0.01 mol KCl solution cm-3 is 150 Ω and the conductivity of this KCl solution is 1.41 × 10-3 Ω-1 cm-1. The same conductivity cell resulting in a resistance of 51.40 Ω for 0.01 HCl solution mol cm-3. Calculate a cell constant and conductivity of the HCl solution.A conductivity cell filled with a 0.01 M KCl solution was found to have a resistance of 189 ohms at 25oC. When filled with 0.01 M HCl solution, the cell gave a resistance of 64.8 ohms at the same temperature. At 25oC, the conductivity of 0.01 M KCl solution was 1.4088 x 10-3 S cm-1. Calculate (a) the cell constant and (b) the conductivity of the HCl solution. answer is [0.266; 4.105 x 10-3] please explain. thank you!) 0.025 M CH3COOH solution has a specific conductance of 2.33X10-4 Scm-1,and molar conductivity at infinite dilution is 364.13 S.Cm2.mol-1. What is itsdegree of dissociation?
- 4.In a certain conductivity cell, the resistance of a 0.01 M KCl solution is 150 Ω. Theknown molar conductivity of the solution is 141.27 Ω-1 cm2 mol-1. Calculate the cell constant (Kcell).*(Kcell unit is cm-1) 5. Using the same conductance cell as in Question No. 4, a student measured the resistanceof a 0.10 M NaCl solution to be 19.9Ω. Calculate the experimental value of the molarconductivity of this solution.Zinc is to be used as an internal standard for the polarographic analysis of thallium. A standard solution containing twice the concentration of zinc as Thallium has a diffusion current of 1.89 µA for Ti and 3.50 µA for Zn. A 10.0g alloy was dissolved in 500 mL, 25 mL of this solution was mixed with 25 mL of 1.0 x 10-3 M Zn 2+ solution. The diffusion currents of this final solution are 18.2µA for Tl+ and 14.5µA for Zn+. Calculate % Ti in the sample analysedb) In one conductivity cell, the resistance of a 0.1 M KCl solution is 1.5x102 Ω. The knownmolar conductivity of the solution is 101 Ω-1 cm2 mol-1 . Calculate the cell constant, Kcell. (Kcell unit is cm-1).
- The electrolytic conductivity of NaCl solution at 18oC is 0.0124 ohm-1cm2 mol-1 and the resistance of the cell containing the solution at the same temperature is 50 ohm. The cell constant will be a. 0.62 b. 0.31 c. 0.124 d. 0.0002480.5M HA electrolyte with a degree of dissociation of 0.2; It is placed in a conductivity cell with a length of 0.9 cm and an area of 2cm2 and its conductivity is read as 300 mS. According to these data; a) Equivalent conductivity of the electrolyte b) Boundary equivalent conductivity c) Decomposition constantCalculate the hardness of water in terms of ppm CaCo3, V used, M EDTA, and Average M EDTA, Ave. ppm CaCO3
- The resistance of a conductivity cell containing a 0.02 M aqueous solution of KCl was fond to be 83 ohms at 298 K. The conductivity of the 0.02 M KCl solution at this temperature is 0.00277 S cm-1. When the cell was filled with a 0.005 M aqueous solution of K2SO4, it exhibited a resistance of 326 ohms at the same temperature. What is the molar conductivity of the potassium sulfate solution? [ answer should be 141 S cm2 mol-1]thank u0.5M HA sample with 0.2 dissociation; It is placed in a conductivity cell with a length of 0.9 cm and an area of 2 cm2 and its conductivity is read as 300 mS. For this purpose;a) Equivalent conductivity of the electrolyte ?b) Boundary equivalent conductivity ?c) Decomposition constant ?In a polarographic experiment of a 60 mL of 0.08 M Cu2+solution, a limitingcurrent was left on for 15 minutes. If the average current during the time of theexperiment is 6.0 μA, what fraction of the copper is removed from the solution? TheFaraday constant is 96485 C/mol of electron.

