Q: 8
A: Two multiple choice questions based on polymer chemistry, which are to be accomplished.
Q: Which compound contains both ionic and covalent bonds? O CH,0 O PCla O CaCOa O MGF2
A: Ionic bond: It is the type of chemical bond which is formed by the complete transfer of electrons fr...
Q: n electrochemical cell is constructed in which both electrodes are pure iron immersed in a solution ...
A: Solution - The correct option is (A)
Q: Get a possible use of an acid-base titration with brief scientific details and include equations.
A: A acid base reaction where a acid reacts with a base to form salt and water. This is an neutralizat...
Q: Q13
A:
Q: name this structure with systematic name
A: The three dimensional representation of an organic compound can be done using the ball-stick model. ...
Q: For the below the molecule, draw a) a diastereomer of the initial alcohol and b) an enantiomer of th...
A: Enantiomers are the isomers that has non-superimposible mirror image to each other. Diastereomers ar...
Q: Does HClO2 has a conjugate acid? If yes, what is it?
A: The Acid decomposition as HClO2 + H2O --------> H3O+ + ClO2- Conjugate Acid-Base pair is [HC...
Q: Q2) a) Explain the titration curve (variation of pH with respect to addition of titrant) with suitab...
A: Dear subscriber! You have posted multiple questions . As per our policy, we have solved the first qu...
Q: Give the major organic product(s) for the below reaction
A:
Q: Which is the stronger oxidizing agent? Ag/AgNO3 (aq) or Mg/Mg(NO3)2 Cu/Cu(NO3)2 (aq) or Mg/Mg(NO3)2 ...
A: The loss of electrons is known as the oxidation while the gain of electrons is known as the reductio...
Q: For which member of the following pairs of complexes would A. be larger and why? i [Cr(H:O)k]* and [...
A: By comparing ligand field strength and central metal ion we can decide the value of Δo for a particu...
Q: For O2 at 1 atm and 298 K, what fraction of molecules has a speed between 200. and 300. m/sec? What...
A: For O2 at 1 atm and 298 K, what fraction of molecules has a speed that is greater than vmp? vave? vr...
Q: In this question, you will demonstrate your ability to convert between pressure units, use correct s...
A: According to combined gas law: P1V1/T1 = P2V2/T2 ...
Q: A strong field ligand such as CO produces_ а. high spin complexes and small crystal field splitting ...
A:
Q: Sketch out the mirror image of alpha-D-mannose and identify the carbon which defines if a sugar is D...
A: We will Sketch out the mirror image of alpha-D-mannose and will identify the carbon which defines if...
Q: The brine water collected from a reverse osmosis desalination plant. The collected water sample was ...
A:
Q: I want the answer for Q1
A: Acidic strength is a measure of ka value. Aromatics compound burns with sooty flame. Normality, mola...
Q: The hydroxide ion(OH-) concentration of an aqueous solution is 6.4 x 10-5 What is the hydronium ion(...
A: The given data contains, [OH-] = 6.4 x 10-5M.
Q: Complete the table. Indicate if the species act as a Bronsted acid, base, or both by writing the che...
A: Complete the table. Indicate if the species act as a Bronsted acid, base, or both by writing the che...
Q: i5.OmL of phosphonic is titratecd w/ 28-3m2 ofa o.520M Solution of potassium hydroxide. what is the ...
A: Given volume of H3PO4 is 15.0 mL. Given volume of KOH is 28.3 mL and concentration of KOH solution i...
Q: Three beakers are placed in a sealed container illustrated below. A B C Initially all the beakers ar...
A: Answer:- this question is answered by using the simple concept of elevation is boiling point of a so...
Q: number 4 only
A: Rice gives positive in Iodine test but not in Benedict's test.
Q: Which of the following reaction sequences can convert toluene to 4-bromo-3-nitro benzoic acid? Selec...
A:
Q: 9
A: The length of one side of the cell can be calculated from the volume of one cell.
Q: at is the composition, in weight percent, of an alloy that consists of 94.1 at% Ag and 5.9 at% Cu? T...
A:
Q: Complete and balance the given equations. Identify the acid, base, conjugate acid, and base showing ...
A: Since you have asked a question with multiple sub-parts, we are solving first three parts for you. F...
Q: It is required to analyse the metal from its ore sample. Manganese (Mn) present in the ore sample wa...
A: The mass of the ore sample is = 2.98 g The mass of the MnSO4 precipitate is = 0.19 g The percentage ...
Q: The reaction of C4H Cl with water is a first-order reaction. The reaction half-life is 340 s. If the...
A: The given problem can be solved by using the formula for the first order given below as; First ...
Q: Complete and balance the given equation. Identify the acid, base, conjugate acid, and base showing t...
A: Acid is a substance that is able of donating a H+ ion. Base can abstract the proton from an acid. Ac...
Q: Use the standard of enthalpies table to determine the change in enthalpy for: SO 2(g) + ½ O 2(g) --&...
A: Dear student I have given answer to your question in the image format.
Q: [References] Use the References to access important values if needed for this question. For the foll...
A:
Q: What effect does an increase in temperature have on the speed distribution of gaseous particles?
A: The Maxwell-Boltzmann distribution curve explains the distribution of speed. This curve gives the fo...
Q: The pOH of an aqueous solution of 0.591 M caffeine (a weak base with the formula C3H10N,O2) is 5.31
A: Caffeine is a weak base with a base dissociation constant (Kb) equals 4.1 x 10-4. For a high concent...
Q: Is there are series of treatment process for water previously qualified for industrial use and make ...
A: The treatment process for drinking water varied. It depend on type and quality of the raw water. Co...
Q: Consider the element silicon, Si. a) Write its electron configuration. b) How many valence electrons...
A:
Q: How the Heating and digestion can effect during the formation ofprecipitate
A: Those reactions in which chemical changes occurs are known as the chemical reactions. There are two ...
Q: Solve this
A: Solution - According to the question - where, In other word, and Since the charge of muon is...
Q: Chemistry Question
A: Since you have asked a question with multiple questions, we are solving first question for you. For ...
Q: Which one of the following is not the electronic configuration of atom of a noble gas? 1s? O 1s 25 1...
A:
Q: Consider a hypothetical chemical reaction: →+AB+CD (In this equation A, B, C and D stand for som...
A: the activation energy for the forward reaction is the difference in energy between the reactants and...
Q: Draw a structure of oxindole, with an aromatic ring. Make sure the diagram contains sp3 carbon atom,...
A: Chemical bonds are the attractive force that holds the atom together. Every atom has distinct number...
Q: A student makes the following observation: when you mix two solutions (each originally at the same t...
A: Given: When you mix two solution's, the temperature of the resulting solution decreases. This is an ...
Q: Draw the products of each of the following reactions and indicate whether the reaction occurs by: a)...
A: An SN1 reaction (Aromatic Nucleophilic) is possible but unfavorable. It would involve the unaided l...
Q: Predict whether these complexes would be labile or inert and explain your choices. The magnetic mome...
A: Dear Subscriber! You have posted multiple questions. As per our policy, we have solved first one he...
Q: 1
A:
Q: 10. Provide the products for the reactions shown. HgSO, HgSO, H2SO4, H2O H2SO4, H20 2 products H9SO4...
A: Hydration of alkyne in presence of HgSO4, H2SO4 gives ketone. (-OH group of enol is formed at carbon...
Q: 3H2(g) + N2(g) ⟷⟷ 2NH3(g) + Heat
A: Equilibrium Constant : Equilibrium constant can be written as ratio of product to the reactant where...
Q: e the hydronium ion concentration in an aqueous solution of 6.57×10² M hydrosulfuric acid, H,S (aq).
A:
Q: Make a PV Diagram for these two heat reservoirs Step 1: Isothermal Expansion=Q = 0, W > 0 Step 2...
A:
Design a synthetic route to prepare three of the following compounds using benzene as a raw material:
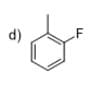
Step by step
Solved in 2 steps with 1 images


