Q: Draw a general Mechanism for Nucleophilic Aromatic Substitution (NAS) reaction
A: Aryl halides are less reactive towards nucleophilic substitution due to resonance effect (+ R) of…
Q: What alkyl halide and nucleophile are needed to prepare each compound?
A: a. The cyanide ion (CN-) acts as a nucleophile on reaction with alkyl halide. The reaction between…
Q: Devise a synthesis of attached compound from an alkyl halide using anyother organic or inorganic…
A: Ethyl bromide can be used for the formation of propionitrile through nucleophilic substitution…
Q: D. А. B.
A: Highly stable conjugated cyclic compounds that do not give addition reactions and follow the Huckel…
Q: (a) Draw two different halo ketones that can form A by an intramolecular alkylation reaction. (b)…
A: Given compound,
Q: Explain why compound A is much more stable than compound B. A B
A: Aromatic molecules are cyclic conjugated system with (4n+2) pi electrons. They are planar compounds.…
Q: What starting material is needed to synthesize each compound by a ringclosing metathesis reaction?
A: Ring closing metathesis : In order to obtain the ring closing metathesis (RCM) , Grubbs catalyst is…
Q: Explain why A is aromatic but B is not aromatic.
A: Given compounds,
Q: a.Draw all reasonable resonance structures for pyrrole, and explain why pyrrole is less resonance…
A: Pyrrole is less resonance stabilized than benzene because of the reason that the in the resonance…
Q: Explain why A is a stable compound but B is not.
A: Introduction : We have to tell which compound is more stable.
Q: Rank the following species from best nucleophile to poorest nucleophile in an aqueous solution (in…
A: Given that : We have to rank the following species form the best to the poorest nucleophile in…
Q: he cyano group is often considered to be more stable than esters or even amides vhen reacting with a…
A: Introduction : We have to tell why cyanide is a stable nucleophile.
Q: Which is the better nucleophile, A or B?
A: This question is related to the topic electrophilicity and nucleophilicity. A nucleophile is species…
Q: Which carbon will be the best site for electrophilic aromatic substitution? A C
A:
Q: In the following pairs of nucleophilic substitution reactions, which one has a higher reaction rate?…
A: Given reactions are nucleophilic substitution reactions.
Q: Draw resonance structures for each radical.
A:
Q: Devise a synthesis of attached alkyne. You may use acetylene, benzene,organic halides, ethylene…
A: Acetylene reacts with NaH to form sodium acetylide and then reacts with PhCH2CH2Br to form…
Q: AICI,
A: Friedel-Crafts alkylation is an alkylation of aromatics using an alkyl halide and a Lewis acid…
Q: Which is the major product? a. A b. Neither product would likely form c. B
A: According to Saytzeff's rule (also known as Zaitsev's rule), during dehydration, more substituted…
Q: a.How many π electrons does C contain? b.How many π electrons are delocalized in the ring? c.Explain…
A: 1. The given comound C has 16pi electrons from the 8pi electron due to teh double bonds
Q: nucleophilic
A: Dear student I have given answer to your question in the image format.
Q: A В C
A:
Q: Which ring in each compound is more reactive toward electrophiles?
A: The species that are electron deficient can be defined as electrophiles. Since, they have less…
Q: Devise a synthesis of attached compound from an alkyl halide using anyother organic or inorganic…
A: The Williamson ether synthesis is a method for the synthesis of ether which occurs by an SN2…
Q: a) electrophilic aromatic addition reaction C) nucleophilic aromatic addition reaction nucleophilic…
A: The question asks about the category under which the nitration reaction of benzene with HNO3 and…
Q: 2. Draw the mechanism (curved arrows) for each acid-base reaction b) d)
A: The given reactions are : Curved arrows mechanism of the given reactions are = ?
Q: Draw the alkyl halide and nucleophile and/or base you would need to make the products. a. Hint: only…
A:
Q: Devise a synthesis of attached compound from acetylene and any other required reagents.
A: A series of reactions has to be carried out to convert the lower hydrocarbons to the higher ones.…
Q: a) b)
A: Applying reagent and suitable chemical reaction As LDA RMgX etc.
Q: Draw curved arrows to show how one resonance contributor leads to the next one.
A: a) Please find below the resonance contributors.
Q: d provide all the necessary reagents in class and any reagent
A: In Organic synthesis the reactant is converted into desired product using specific reagent
Q: Explain why A is aromatic but B is not aromatic (see attached file)
A: Given: Ring A: 9 membered unsaturated ring with 1 unsubstituted nitrogen atom Ring B: 9 membered…
Q: Why hydroxide nucleophile is needed to synthesize an alcohol ?
A: Alcohols are organic compounds which contain hydroxyl group. These compounds are polar and water…
Q: Explain why acetanilide is less reactive toward electrophilic substitution than aniline.?
A:
Q: Which sn² reaction is each pair is faster
A: Given Two SN2 reactions varied by two different nucleophiles OH- and CH3COO-
Q: can you identify A and B
A: Identify A & B.
Q: compounds A and B ?
A:
Q: Rank the attached alkenes in order of increasing stability.
A: Alkene stability depends on Degree of hyper conjugation.
Q: What alkyne gives each compound as the only product after hydroboration-oxidation?
A: Hydroboration reaction takes place in the presence of a BH3 or B2H6 and H2O2, and will lead to the…
Q: Draw a curved arrow mechanism for reaction d
A: Reaction d is ether formation reaction by substitution.
Q: how to know whihc halide is more nucleophilic in a competition reaction
A: In compitition reaction, reactivity is depend upon the nucleophilic power of particular halide.…
Q: Which carbon is most electrophilic in Y? Explain your choice.
A: Since the carbon attached to 2 O as shown below will be most electrophilic because it is having 2 O…
Q: B) How the electrophilic substitution occurs in aromatic system?
A: Electrophilic substitution reaction is a well known reaction shown by benzene. In this reaction,…
Q: Explain why A is a stable compound but B is not.
A: The stability of Compound A and unstability of compound B can be best explained by Bredt's Rule.
Q: Does A or B undergo elimination reaction
A: Compound A cannot undergo beta-elimination reaction. Because Tungsten atom (W) has six valence…
Q: Devise a synthesis of attached compound from the indicated starting material.
A: Benzene is an aromatic compound which contain electron density and is attacked by the electrophiles.…
Q: Which of the following statement about using curved arrows in polar reaction mechanism is true?…
A: The major objective of the question is to find the true statement which is about using curved arrows…
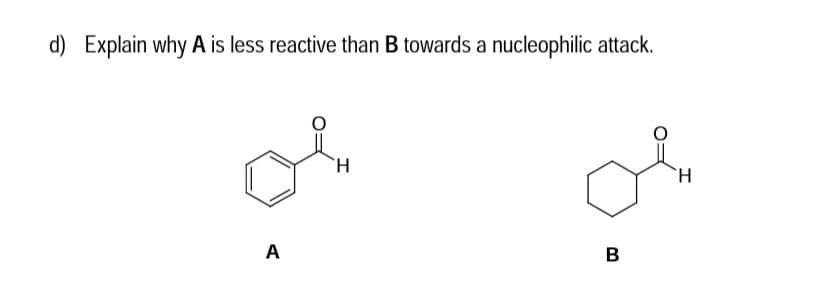
Q: d) Explain why A is less reactive than B towards a nucleophilic attack. `H. A.
A: The reactivity of aldehyde depend upon the electrophilic character of carbonyl Carbon.
Step by step
Solved in 2 steps

- (a) Draw a stepwise mechanism for the conversion of A to B. (b) What product would be formed if C was exposed to similar reaction conditions?Draw a stepwise mechanism for the attached reaction. (Hint: Conjugateaddition can occur with heteroatoms as well as carbon nucleophiles.)a.Draw all reasonable resonance structures for pyrrole, and explain why pyrrole is less resonance stabilized than benzene. b.Draw all reasonable resonance structures for furan, and explain why furan is less resonance stabilized than pyrrole.
- Draw a stepwise mechanism for the attached reaction, which involves two Friedel–Crafts reactions. B was an intermediate in the synthesis of the antidepressant sertralineDraw a stepwise mechanism for the following Friedel–Crafts acylation.susceptible to nucleophilic attack a to b c only a only a and c

