Q: 11:52 l 5G Question 4 of 25 Submit Draw one of the two enantiomers of the major product from this re...
A: Answer Draw one of the two enentiomer of the major product f...
Q: 2. Calculate the number of moles of H2 produced when 0.500 g of Mg (s) and 500. mL of 0.200 M HCl ar...
A: Given data: The mass of Mg is 0.500 g. The volume of HCl is 500 mL=0.500 L. The molarity of HCl is ...
Q: Consider a system with Hamiltonian operator A that is in a state , with energy Ex. where and k is th...
A:
Q: A app.101edu.co A Apps MyUSU O Outlook Canvas Labflow OneNote 101 Chem101 Tophat U USU Tutoring Ques...
A: Given, Moles of an unknown solid = 0.700 mol. Volume of the solution = 150.0 mL. The density of the ...
Q: An electric immersion heater rated at 100 W is used to heat up 10 kg of water initially at 20 Degre...
A:
Q: A particular antioxidant is 56.5% C, 5.7% H, and 37.7% O by mass. Determine the empirical formula ...
A: Let consider the mass for compound = 100 gram Therefore 100 gram compound contains., mass of carbon...
Q: What is the enthalpy of the reaction P406 (s) + 202(g)→P4010(s), given a) P4(s) + 302(g)→P406 AH = -...
A:
Q: Complete the following sentence by typing a number in each of the answer boxes. HO. Not considering ...
A:
Q: Sulfur dioxide and oxygen react to form sulfur trioxide, like this: 2s0,(e)+0,(2) → 2S0,(2) Suppose ...
A: The balanced reaction given is, Given: The reaction is in equilibrium initially.
Q: hich of the following statements is true for real gases? Pick those that apply. The volume occupied...
A: Given : We have to tell the true statement regarding real gas.
Q: H2SO4 (aq) + 2Al(s). ----> Al2(SO4)3 (aq) + 3H2(g) DeltaH = - 1055kJ a. What is the enthalpy c...
A: Given : We have to calculate the enthalpy of formation of Al2(SO4)3 (aq) .
Q: For a 1.0 x 10-6 M solution of HNO, (aq) at 25 °C, arrange the species by their relative molar amoun...
A: Nitric acid is a strong acid and it dissociates completely into the aqueous solution: HNO3aq + H2Ol ...
Q: Using the equations N, (g) + 3 H, (g) → 2 NH, (g) AH° = -91.8 kJ/mol C(s) + 2 H, (g) – CH, (g) AH° =...
A:
Q: Which hazard statement is false in regards to dichloromethane? 1) May be corrosive to metals 2) Ca...
A: Answer False statement about Dichloromethane Dichloromethane is a toxic ...
Q: Consider the data in the table. Compound Melting point (°C) | AHfus (kJ/mol) Boiling point (°C) | AH...
A: The data given is,
Q: Retention time can be used to identify a compound in a mixture using gas chromatography. Choose a pa...
A: We have to tall which parameter will not affect the retention time of a compound in a gas chromatogr...
Q: Which elements are primarily used in NMR? Select all that apply P F O H C Cl Br N
A: Answer Which elements are primarily used in NMR spectroscopy Many nucle...
Q: What was the concentration of my stock solution if I used 26mL of it to make 116ml of a 1.5 M diluti...
A: Given that, V1= 26 mL , M1=? V2=116 mL , M2= 1.5 M
Q: Which of these processes result in an entropy decrease of the system?
A: Entropy means randomness. All spontaneous processes have positive or zero entropy change. This means...
Q: Chemists graph kinetic data to determine rate constants and the order of reactions. Analyze this dat...
A:
Q: Which of the following isomers is more soluble in water: diethyl ether (CH3CH2OCH2CH3) or butan-1-ol...
A: The solubility of a compound depends on the nature of the solute and the solvent. If the nature of b...
Q: A. Draw the bonding and antibonding MO for the linear combination of the following sets of orbitals....
A:
Q: Correct Answer List Question List (4 images) (Drag and drop into the appropriate area) Most reactive...
A: The compounds given are,
Q: O Attempt Calculate the pH of a blood plasma sample with a total CO2 concentration of 27.7 mM and bi...
A: Introduction : Henderson- Hasselbalch Equation is the equation that provides a relationship betwee...
Q: 2N,0, (g) – 2N,0, (g) +0, (g) At a certain temperature it obeys this rate law. -(3.0 M")[N,0, rate =...
A: 2N2O5(g) -----> 2N2O4(g) + O2(g) Rate = 3.0 M-1 s-1[N2O5]2 Initial concentration of N2O5. = 0.450...
Q: Identify the functional groups in the following molecules. (Use names from the table below. List eac...
A: Functional groups : Functional groups are the specific group of atom within molecules which having t...
Q: 2. Consider the following potential energy diagram for a reversible reaction: 40 30 PE ) 20 10 Progr...
A:
Q: Which of the following sets of atomic orbitals will form a pi bond? s+ px O px + px O py + py s+ pz
A: Interaction between two atoms results yhe formation of a chemical bond between them. By this , two o...
Q: Draw the major and minor monochlorination products of this reaction. Cl2 (1 equiv) hv
A: Draw major and minor product on monochlorination ---
Q: For each of the compounds below, A В , and C state whether the molecule is polar or nonpolar. A o=C=...
A: A) The given molecule is, The given molecule is non-polar.
Q: Column resolution is an indicator to identify the selectivities and efficiencies of the column abili...
A: Coloumn resolution is an indicator to identify selectivities and efficiencies of coloumn ability to ...
Q: Methane and chlorine react to form chloroform and hydrogen chloride, like this: CH,(9)+3Cl,(9) -→ CH...
A: Le Chatelier's principle: This principle is used to predict the effect of a change in conditions on ...
Q: Reversed phase HPLC refers to the condition of stationary phase and a mobile ph separation. Select t...
A: Given : We have to choose the correct statement for reverse HPLC.
Q: Ammonia has been studied as an alternative "clean" fuel for internal combustion engines, since its r...
A: Molarity = Number of moles of compound / Volume of solution (L)
Q: a Write the molecular equation which completes the following. The reaction occurs in an aqueous solu...
A: In balanced reaction number of each atom on reactant side is equal to number of each atom on product...
Q: 1. The measured electromotive force (EMF) for the cell Pt(s) H2(g, p = 1 atm) H*(aq, a = 1)||Cu²*(aq...
A: In an electrochemical cell notation, the oxidation half-reaction is represented on the left side, wh...
Q: What mass of salt (NaCl) should you add to 1.64 L of water in an ice-cream maker to make a solution ...
A:
Q: Write the IUPAC names of the following organic compounds if given the structure. Draw the organic co...
A: We select longest carbon chain first and if functional groups are present we try to have all of them...
Q: Classify the following substituents according to whether they are electron donors or electron accept...
A: Inductive effect: Attraction of bond pair of electrons towards itself through sigma bond is called ...
Q: Exactly 3.5 moles of N204 is placed in an empty 2.0-L container and allowed to reach equilibrium des...
A:
Q: AIs questioL Tlas lmuiti a Complete the right side of the following molecular equation. Assume all s...
A:
Q: Consider this reaction: 2H,PO, (aq) → P,05 (aq) +3H,0 (aq) At a certain temperature it obeys this ra...
A:
Q: A signal for an allylic proton was found to come at 2.43 PPM using a 300 MHz NMR instrument. What wo...
A:
Q: Write a rate law for the reaction, including the value of k. 0.11M/s=K[0.8] to the zero order.
A:
Q: 3. (a) Calculate the fraction of atom sites that are vacant for aluminum at its melting temperature ...
A:
Q: Calculate how much 1500 µg/dL stock solution is needed to make 50.00 mL a 300 ug/dL solution. Answer...
A: Dilution of the solution: When we dilute a concentrated solution with pure water, the mole of solute...
Q: What will happen to the sample size if the percentage error becomes lower e.g. 5% instead of 10%? Ex...
A: A question based on tools in analytical chemistry that is to be accomplished.
Q: The diol shown undergoes reactions with reducing and oxidizing agents to yield two different product...
A: Given : We have to draw the product for the given reactants.
Q: :0: Paraphrasingewriting Tool Car note Pirate Ship BLACKBOARD [Review Topica) [References) For each ...
A: A single structure is not able to tell the properties of the compound. Hence , resonance come in to ...
Q: Please answer with detailed mechanism, with all steps otherwise I'll downvote
A:
Please explain part D

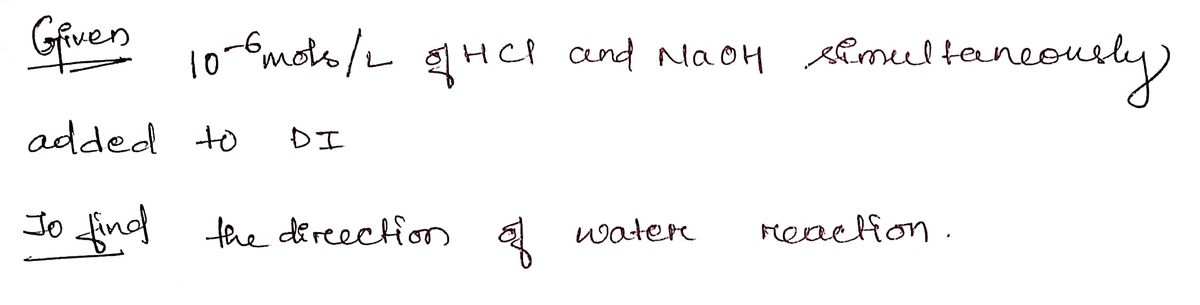
Step by step
Solved in 2 steps with 2 images

- Dapsone tablets, labelled 50 mg has an average tablet weight of 0.6509 g. 2.0g of powdered tablets reacted (see reaction below) with 26.05 mL of 0.0506N sodium nitrite (NaNO2) solution. Determine the amount of Dapsone as a percentage of the Label Strength (or Label Claim). Report your answer to 1 decimal place. Given: Dapsone molar mass, 248.302 g/mol Dapsone structure Reaction: Sodium nitrite (NaNO2) reacts with hydrochloric acid (HCl) to generate nitrous acid (HNO2). NaNO2 + HCl ® HNO2 + NaCl Nitrous acid (HNO2) then reacts with each amino group on Dapsone, and an indicator indicates the endpoint. R-NH2 (full structure shown above) + HNO2 + HCl ® R-N2Cl + 2H2ONa2CO3(aq) +2HCl(aq) 2NaCl(aq) + CO2(g) + H2O(l) How many moles are found in 15mL of 4.5M HCl? How many moles of CO2(g) will be produced when 15mL of 4.5M HCl completely reacts according to the reaction above? How many liters of CO2(g) will be produced when 15mL of 4.5M HCl completely reacts according to the reaction above at 25C at 1.05atm?Run 1 Run 2 Molarity of KMnO4 solution (M) from bottle 0.00101 0.00101 Initial reading of buret KMnO4 (mL) 0.62 11.35 Final reading of buret KMnO4 (mL) 11.33 21.70 Run 1 Run 2 Volume of KMnO4 solution (mL) Moles of MnO4- used for titration (mol) Moles of C2O42- in 100.0 mL of solution (mol) Molarity of C2O42- (M) Molarity of Cd2+ (M) Ksp of CdC2O4 Average Ksp of CdC2O4 Calculations: Moles of MnO4- used for titration of saturated solution CdC2O4 Moles of C2O42- in 100.0 mL of saturated solution of CdC2O4 Molarity of C2O42- in saturated solution of CdC2O4 Molarity of Cd2+ in saturated solution of CdC2O4 Solubility product, Ksp, of CdC2O4
- Will the calculated Molarity of NaOH be too high or too low or unaffected if the following happen: When you answer the question, consider how the situation affects the calculation of molarity of NaOH and moles NaOH, which is calculated from the mass of KHP. For each answer, you must supply a clear explanation for your answer. Often times, students will restate the facts rather than explaining why they chose the answer they chose. a) You add the weighed KHP to a flask containing a 60mL of water rather than 50 mL of water. Explain. Your answer must contain a well-thought-out and clearly written explanation. b) The buret is still wet with water on the inside when you add your NaOH solution. Explain c) The KHP is wet when you weigh it. Explain. d) You titrate past the equivalence point by 0.50mL Answer a-dIt is known that acid content has a major effect on theflavor of vinegars, but most cheaper vinegars are diluted similarly to 5% acidity Wt./vol. % is equivalent to gsolute per 100mL solution (so 5% is equivalent to 5 g acid/100 mL solution). a.) First, calculate the approximate molar concentration of acetic acid in the 5% wt./vol vinegar. b.) Next, calculate the expected molarity of acetic acid in the solution upon dilution by a factor of 5. Thank you!(1) Careless Carlie weighed her KHP sample, but forgot to bring her report sheet along, so she recorded the mass of KHP on a paper towel. During her titration, which required 18.46 mL of base, she spilled some base on her hands. She remembered to wash her hands, but forgot about the data on the towel, and used it to dry her hands. When she went to calculate the molarity of the base, Carlie discovered that she didn’t have the mass of her KHP. Her kindhearted instructor told Carlie that her base was 0.2987 M. Calculate the mass of Carlie’s KHP sample. Show calculations (2) What mass of solid NaOH would be needed to make 645 mL of Carlie’s NaOH solution? Show calculations.
- Hello. I am having issues understanding acetic acid and the correlation between its concentration and molarity. Can you help explain to me if the concentration and molarity of acetic acid in vinegar remains the same during its shelf life in the kithcen or does it change? I understand that it does not technically expire and it has an indefinite shelf life, but does the concentration and molarity change? If so, how. If it doesn't, why not? What is the molarity/concentration of acetic acid to begin with?? Thank you for your helpThe first goal is to make the oxalic acid standard solution. You measure 1.5232 g of oxalic acid on an analytical balance, add it to a 250-mL volumetric flask and add deionized H2O to a final volume of 250.0 mL. Molar mass of H2C2O4•2H2O = 126.07 g/mol Mass of H2C2O4•2H2O = 1.5232g what is the Number of moles of H2C2O4•2H2O?1) How much total glucose is present in a 75 Kg female person? (a normal person (70 kg BW) has approximately 43L body fluid).2) Please express the normal salt (NaCl) concentration in body fluid into molarity (mM).3) Commercial fuming Sulphuric acid (Oleum-H2S2O6) is 99.9% solution. Please convert it into molarity.4) Find out the Volume (dm3) of product (gas) at RTP when 0.58 M, 150 mL NaOH (aq.) reacts with 350 mL, 0.25 NH4Cl.5) The above reaction has the product Ammonia, which when dissolved in 650 mL ethanol makes an alkaline ethanolic solution. Find its molarity (M).6) Calculate the adult dose as per the BW of the baby. (Child dose-50 mg and the BW of the baby is 48 lb (British pound) (1lb=0.453 Kg)
- 10) How can you prepare 500 ml of 0.1M HCI, if the percentage is 35%, and density = 1.18g/ml, M.mass=36.5 g/molI’m so lost. How do I attempt this problem? I think I have to get NaC2H3O2 and MC2H3O2 in moles. And then combine the moles together for the A^- portion of the Henderson equation. But how do I do the rest?Kepone, a carcinogenic insecticide, fungicide, and larvicide for bananas, tobacco, and other domestic plants is insoluble in water. The US banned it's use in 1975, but the chemical is still used in some other countries. Before itsbanning, large amounts were depositing in the upper James River, posing a threat to the marine animals and groundwater supply. Most has settled into sands and sediments in the river.Kpis needing to be determined for a sediment sample. A solution of 0.02453 mg/L Kepone is prepared. 100.0 mL of solution is placed in a vial with 0.000100 kg of dry sediment. The sample is mixed for 3 days and the aqueous and solid phases are separated via filtration. The mass of Kepone in the aqueous phase is measured and the concentration is determined to be 0.02108 mg/L. The blank analysis shows no loss of Kepone to the vial walls. Determine the Kpusing the stepwise process outlinedbelow. Note, some of these may not becalculations, but rather givens from the problem. Total mass…

