Q: Can you explain the problem? Is it a salt?
A: Sodium chloride (NaCl) is considered a neutral salt.This is because its ion can form both strong…
Q: A student artificially pumped 1 mole of each substance seen below in a piston with a moveable lid…
A: The reaction:2 C3H7OH(g) + 9 O2(g) ←→ 6 CO2(g) +8 H2O(g) The number of molecules in reactant side =…
Q: Give the IUPAC name of the molecule.
A:
Q: Question 5 Please predict the products for each of the following reactions: 1. Na 2. PrBr Na 10 1.…
A: Reactions: Mechanism of particular reaction which is asked in question
Q: Determine the temperature of a reaction if K = 1.20 x 10⁻⁶ when ∆G° = +26.90 kJ/mol.
A: The objective of this question is to calculate the temperature of a reaction given the standard…
Q: 4. For each reaction, look at all the reagents and conditions, and determine if the reaction will…
A:
Q: 3. For medicinal substances: sodium chloride, formaldehyde solution, ascorbic acid - the general…
A: 3. For pharmaceutical compounds such as ascorbic acid, formaldehyde solution, and sodium chloride,…
Q: dont provide handwriting solution ....
A:
Q: OH CO₂H + NH₂ SO3H 1. Give the mechanism for the reaction 2. Discuss the reason(s) why the dye…
A: The first part of the question is asking for the mechanism of the reaction between OH CO2H…
Q: A chemist has a 16.0 mL sample of Ca(OH)2 with an unknown concentration. To neutralize the base,…
A: The objective of this question is to find the concentration of Ca(OH)2. This can be done by using…
Q: 7.16 Consider the isoelectronic ions F and Nat. (a) Which ion is smaller? (b) Using Equation 7.1 and…
A: A. Na+ is the smallest as it has lost an electron from its outermost shell thereby pulled a bit more…
Q: These disorders are associated with inborn errors of metabolism: Question 13 options:…
A: Option a: This option is incorrect because Types 1 and 2 diabetes mellitus can impact metabolism,…
Q: Identify the Brønsted-Lowry acid and base in each of the reactions. HF + H₂O → H3O+ + F¯ H₂O + H2CO3…
A: Step 1: Step 2:Acids: According to the Brønsted-Lowry definition, an acid is a substance that…
Q: 5. How many grams of CuSO4 * 5 H₂O are needed to prepare 100 ml of a 0.10M solution? (This is like…
A: The target volume to prepare is 100 mL and the concentration is 0.10M CuSO4. So we have…
Q: How many grams of solid sodium hypochlorite should be added to 1.50 L of a 0.232 M hypochlorous acid…
A: The objective of this question is to determine the amount of solid sodium hypochlorite (NaClO) that…
Q: For the reaction below which of the following answers (a-i) best describes the major product(s) you…
A:
Q: Question 10 Please predict the products for each of the following reactions: 1. Na 2. PrBr เวรเ 10…
A: Step 1:At first presence of Na metal ,it released one electron , which abstract alcoholic proton and…
Q: In the proton NMR spectra of aldehydes and ketones, the protons bonded to carbons adjacent to the…
A:
Q: None
A:
Q: 2:15 AM Sun Apr 14 5% Question 4 of 8 One of the commercial uses of sulfuric acid is the production…
A:
Q: What constitutes a 'reference material', and why does its utilization play a criticalrole in the…
A: A reference material is a substance with a specific characteristic that serves as a comparative…
Q: None
A: Step 1: For general reaction of gases, the equilibrium constant (Kp) can be represented as:where:P…
Q: The acid dissociation K of acetic acid (HCH3CO2) is 1.8 × 105. -4 Calculate the pH of a 4.1 × 10 M…
A: The objective of this question is to calculate the pH of an acetic acid solution given its molarity…
Q: Describe how you could achieve a higher degree of separation for the bands of food dyes on a TLC…
A: Step 1:Optimize the Mobile Phase: The choice of mobile phase, which is the solvent that moves up the…
Q: - Aktiv Chemistry ← → C M Gmail b Answered: Draw the major pro × | + app.aktiv.com ☑ ੩ ॥ YouTube…
A: Thank you.
Q: Please don't provide handwritten solution ....
A: Given:…
Q: Question 11 Choose the correct chemical structure for every single acronym used below: PrBr A B C D…
A: Step 1:Propyl bromide, represented by the chemical formula C3H7Br, is an organic compound belonging…
Q: What is the Kyoto Protocol?
A: The Kyoto Protocol is an international treaty that was adopted in Kyoto, Japan, in December 1997. It…
Q: Please fast expert answer solutions
A:
Q: What is the formula of ammonium nitrate? NH4NO3 (NH4)2NO3 NH4(NO3)2 NH4N
A: The objective of the question is to identify the correct chemical formula for ammonium nitrate.
Q: Sodium hydroxide is a substance that causes severe burns. It is used in many educational laboratory…
A: Contact with very high concentrations of sodium hydroxide can cause severe burns to the eyes, skin,…
Q: Question 29 Predict the FINAL (?) product for each of the following reaction or synthetic chain: A…
A: Thank you.
Q: MISSED THIS? Watch KCV: Solubility and The Solubility Product Constant, IWE: Calculating Molar…
A: CuCl <=> Cu+ + Cl-Mass of CuCl in 1 L (1000 mL) of solution = 1000/100 x 3.76= 37.6 mg =…
Q: Name the following molecule using the IUPAC system Br
A: The following steps are taken in consideration while naming Alkylbromides:Step 1:Identify the…
Q: B1. Draw the partial orbital diagram for nitrogen and oxygen in NH2O*. Next to each diagram draw the…
A: A partial orbital diagram of an atom shows the filling of electrons in the higher energy valence…
Q: 3. Problem: Outlined below is a synthesis of the gypsy moth sex attractant E (a type of pheromone).…
A:
Q: please answer in text form and in proper format answer with must explanation , calculation for each…
A: The reduction half-reaction involves the gain of electrons. Pb2+ is gaining electrons to form Pb,…
Q: Question 28 Predict the FINAL (?) product for each of the following reaction or synthetic chain: Mgl…
A: The sequence of reactions shown suggests the formation of a Grignard reagent followed by a reaction…
Q: ← 68 Problem 15 of 27 Submit Draw the major product of this reaction. Ignore inorganic byproducts.…
A: Step 1:Step 2:
Q: Which of the following would be a major product of the following reaction? Br Br Br hv ? Br2
A:
Q: A 1.00 L flask is filled with 1.45 g of argon at 25∘C. A sample of ethane vapor is added to the same…
A: The objective of this question is to find the partial pressures of argon and ethane in a flask. The…
Q: Predict the products of the following reaction. If no reaction will occur, use the NO REACTION…
A: Step 1:When solid strontium (Sr) reacts with hydrobromic acid (HBr), a single displacement reaction…
Q: For butane, the ∆H° of vaporization is 22.40 kJ/mol and the ∆S° of vaporization is 82.3 J/mol・K. At…
A:
Q: Can the molecule on the right-hand side of this organic reaction be made in good yield from no more…
A:
Q: Read the buret (burette) volume and report your reading with the proper number of digits. 0 mL…
A: As we can see from figure , water level is between 13.7 to 13.8 , thus correct value upto 2 decimal…
Q: Use oxidation numbers to identify the species oxidized and reduced in an oxidation- reduction…
A:
Q: Question 15 Choose the correct chemical structure for every single acronym used below: n-BuLi ? A B…
A: Step 1:The acronym "n-BuLi" stands for "normal butyllithium." "n-" Prefix: In this context, "n-"…
Q: Question 14 Choose the correct chemical structure for every single acronym used below: PhBr A B C ?…
A:
Q: Use the observations about each chemical reaction in the table below to decide the sign (positive or…
A: The objective of the question is to determine the signs of the reaction enthalpy (ΔH) and reaction…
Q: The concentration of H+ in a solution is 1×10−2 M. What is the hydroxide ion concentration?
A: The objective of this question is to find the concentration of hydroxide ions (OH-) in a solution…
Please do letter e. Give the major organic product or missing starting material for the following.
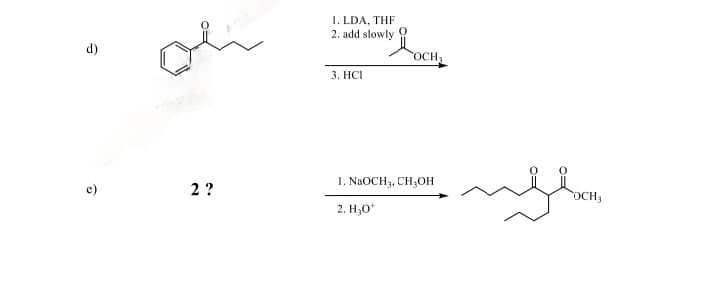
Step by step
Solved in 2 steps with 2 images




