Question 29 Predict the FINAL (?) product for each of the following reaction or synthetic chain: A MgBr 1. 03 (excess) ? (2 equivalents) 2. H₂O 2. Me₂S но B 1. CH3MgCl (2 equivalents) 2. H₂O C OH HO OH на зон مند شد HO A B C D OH HO OH Na2Cr2O7 H2SO4 (2 equivalents) D
Question 29 Predict the FINAL (?) product for each of the following reaction or synthetic chain: A MgBr 1. 03 (excess) ? (2 equivalents) 2. H₂O 2. Me₂S но B 1. CH3MgCl (2 equivalents) 2. H₂O C OH HO OH на зон مند شد HO A B C D OH HO OH Na2Cr2O7 H2SO4 (2 equivalents) D
Organic Chemistry
8th Edition
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Chapter24: Catalytic Carbon-carbon Bond Formation
Section: Chapter Questions
Problem 24.30P
Related questions
Question
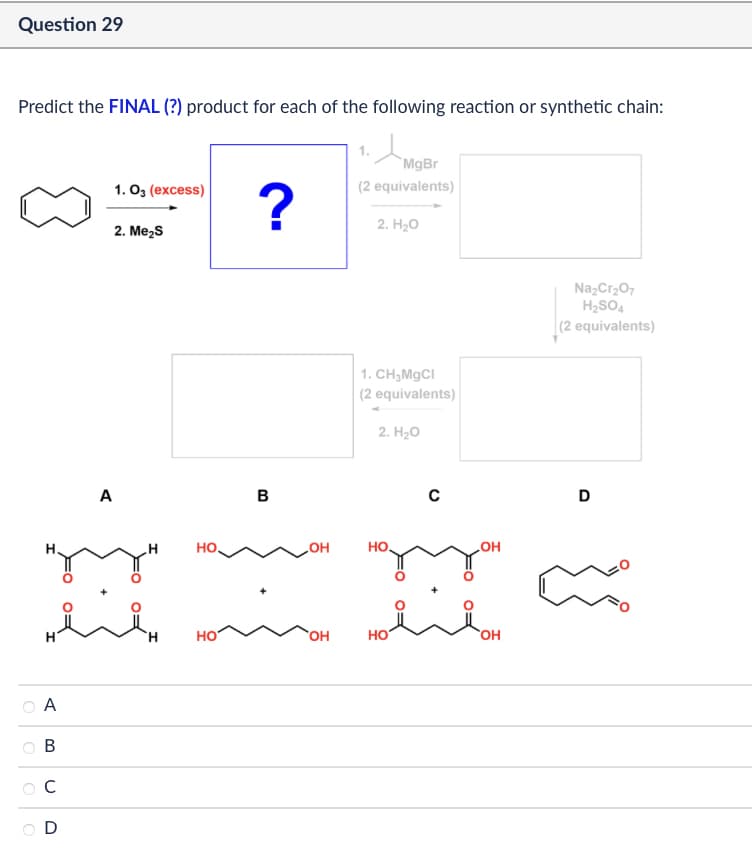
Transcribed Image Text:Question 29
Predict the FINAL (?) product for each of the following reaction or synthetic chain:
A
MgBr
1. 03 (excess)
?
(2 equivalents)
2. H₂O
2. Me₂S
но
B
1. CH3MgCl
(2 equivalents)
2. H₂O
C
OH
HO
OH
на зон
مند شد
HO
A
B
C
D
OH
HO
OH
Na2Cr2O7
H2SO4
(2 equivalents)
D
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning
