Q: 6) Using the following equation: 2 NaOH + H₂SO4 → 2 H₂O + Na2SO4 How many grams of sodium sulfate…
A:
Q: Write the balanced net ionic equation for the reaction of FeCl3 with NaOH in aqueous solution.…
A:
Q: Which one of the following is an intensive property? density O mass O heat content O volume moles
A: Intensive properties are physical properties of a substance that do not depend on the amount or size…
Q: ← Curved arrows are used to illustrate the flow of electrons. Using the provided starting and…
A: In the above question we have to draw the arrow pushing mechanism for above reaction. So let's…
Q: CI Br .... Ph NaSCH3 acetone NaOCH3 CH3OH
A: Strong nucleophile SN2 mechanism which proceeds through inversion in configurationStrong base favors…
Q: Consider a chemical reaction where compound A reacts with compound B to form compound C. The…
A: The observed phenomenon in this chemical reaction involves the comparison of two catalysts, X and Y,…
Q: ||| O STATES OF MATTER Drawing the unit cell of a 2D lattice Color in a unit cell of this…
A: A regular 3-D arrangement of ions, atoms, or molecules in the crystal is generally described in the…
Q: The balanced equation for the combustion of butane is: 2C4H10+1302 8CO₂ +10H₂0 di See Periodic Table…
A:
Q: Which of the following elements require Roman numerals when writing their ion names? Select all that…
A: Roman numerals are typically used when writing the ion names of transition metals, as well as some…
Q: Consider the following system at equilibrium where AH = -16.1 kJ, and K, 154, at 298 K: 2NO(g) +…
A: A question based on equilibrium constant. An equilibrium equation is given followed by few sub-part…
Q: Questions Draw the structures and provide the IUPAC names of the product(s) of the reaction of…
A: Since, there is multiple questions are present. We solve only first question for you according to…
Q: A doctor prescribes 2.5 g2.5 g of a drug, daily, for a patient. The pharmacist has only 500500 mg…
A: A doctor prescribes 2.5 g of a drug, daily, for a patient. The pharmacist has only 500 mg tablets…
Q: 5) IDENTIFY the products of the following reactions
A: In electrophilic aromatic substitution hydrogen of benzene is replaced by electrophile.
Q: Propose an efficient synthesis of the following. Make sure you identify the reagents and the…
A: In this question, we will propose a efficient synthesis from the starting material to the final…
Q: Determine the oxidation state of metals and its electronic configuration for the following complexes…
A: To determine the oxidation state of metals and their electronic configurations in the given…
Q: () ¹H NMR spectrum of an alcohol (CsH12O) is shown with related integral values of the each proton…
A: A question based on NMR spectroscopy. An NMR spectrum is given whose respective structure is to be…
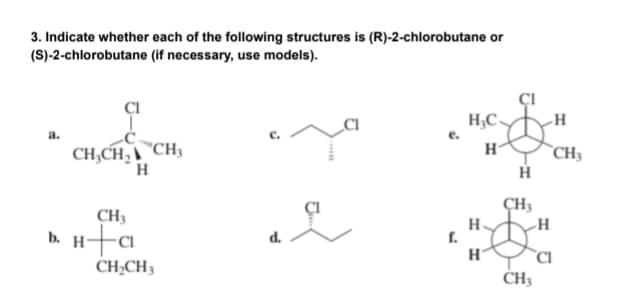
Q: Q2. Please identify chiral centers in the molecule given below and calculate total number of…
A: Chiral Carbon is that carbon to which four different groups are attached. This results in optical…
Q: Calculate the pAg values after the addition of 5.0, 40.0, and 50.0 mL of 0.050 M AgNO3 into 50.00 mL…
A: IntroductionThe titration involves mixing solutions of AgNO3 and KBr, resulting in the formation of…
Q: 1. a) Draw the product for the following reaction. THF b) is the mechanism SN1 or SN2?
A: “Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: What is the pH of 0.055 M KOH solution? What is the pH of 0.55 M KOH solution What is the pH of a…
A: 1. Concentration of KOH = 0.55 M2. Concentration of Chloroacetic acid = 2 M3. Concentration of…
Q: Provide the major organic product for each of the following reactions
A: Three-membered rings formed by two carbon and one oxygen atom is known as epoxide ring. The…
Q: int: C10H14 ¹HNMR: 6.5 (1H, t), 6.4 (1H, q), 6.20 (1H, d), 2.9 (2H, d), 2.6 (2H, s), 2.1 (1H, m),…
A: Structure of the compounds is given below.
Q: A. Add all implied hydrogen atoms and any missing lone pairs of electrons. There are many possible…
A: Implied Hydrogen: The missing hydrogen atoms in the line angle diagram or structure of molecules are…
Q: Consider the reaction below: D HBr The final product of this reaction has [Select] and may form a…
A: Protonation of alkene gives carbocation which then undergoes addition of bromide ion to produce…
Q: Identify the letters A-E. Letter A= HBr Letter B= tbOK Letter C= H3O+ Letter D= Letter E= H₂C CH3…
A: Please note that there is a double bond in the starting compound
Q: Use the attached image to answer the following question for the reaction. Given the absolute…
A:
Q: Add or subtract the following measurements. Be sure each answer you enter contains the correct…
A: To add the numbers 14.820ml and 3.97ml while considering the correct number of significant figures,…
Q: A sample of sodium carbonate, Na2CO3, weighs 40.0 g. Calculate the following quantities (a) ? mol…
A: The number of molecules or atoms present in the one mole of the substance is equal to the Avogadro…
Q: c. Balanced formula equation: Ca(OH)₂ (aq) + Complete ionic equation: Net ionic equation: + + +…
A: When solid zinc hydroxide (Zn(OH)2) reacts with aqueous nitric acid (HNO3), the complete ionic…
Q: Calculate the linear density in the [111] direction of aluminum metal. Aluminum has fcc cubic unit…
A: Unit cell is the smallest repeating unit of the crystal structure. It is the basic building block of…
Q: Which type of organic molecule is formed when a carboxylic acid and an alcohol react? a) aldehyde b)…
A: The above question is based on organic reaction and mechanism. We have to discuss the product of…
Q: Write a balanced chemical equation for each reaction.
A: A balanced chemical equation is an equation in which all the atoms of each species should be equal…
Q: How many milliliters of 0.250 M HNO3 solution can react with 3.25 g of Mg in the following reaction?…
A: Reaction involved : Mg (s) + 2HNO3 (aq) → Mg(NO3)2 (aq) + H2 (g)Mass of Mg = 3.25 gMolarity of HNO3…
Q: What are the potential limitations and challenges associated with the use of density functional…
A: Introduction:Density functional theory (DFT) is a powerful tool in theoretical chemistry, but its…
Q: Consider a chemical reaction between substances A and B. At room temperature, the reaction is known…
A: The unexpected change in the enthalpy of a chemical reaction at extremely low temperatures can be…
Q: Questions: - what type of ligands present in the compound? Compound A (first picture) compound B…
A: In the two complexes, the type of ligands (mono, bi or tri dentate) and whether anionic or neutral…
Q: What is the pH of a solution when 33.4 g of methylammonium chloride. CH3NH,CI, is dissolved in…
A: Provided dataVolume of solution is 514.2 mL or 0.5142 L.Mass of the sample(CH3NH3Cl) is 33.4…
Q: 19. A gas occupies a volume of 35.7 L at 621 torr and 741°C. What volume will the gas occupy at 5.31…
A: The relationship between the temperature, pressure, and volume of the initial and final state of an…
Q: Predict and calculate the effect of volume change on an equilibrium system. Consider the equilibrium…
A: Considering the equilibrium between PCl5, PCl3 and Cl2 as The initial number of moles of each…
Q: Question 3 What mass of carbon dioxide is produced from the combustion of 2 kg of propane? Write…
A:
Q: Give IUPAC names for the (2) Give IUPAC names for the following compounds. pay special attention to…
A: Give the IUPAC nomenclature for the compounds Based on IUPAC rules
Q: Ph. D-C-CH,CHO PHCH₂CHDCH₂CH=CHCH₂ CH₂CH=CHCHDCH₂CH₂P 0-0-04 0-0-040 Po OA OB hv Additional…
A: The Given Reaction is photolysis of aldehydesThe Aldehydes undergoes photolysis as follows:The…
Q: Consider a reaction A B C carried out in a plug flow reactor. Initial concentration of A, B. and C…
A: The initial concentrations of A, B and C are given as 4, 2 and 0 mol/m3 , respectivelyMean Axial…
Q: Read each description in the first column of the table below. If any chemical element with atomic…
A: Periodic Table compromises of the chemical elements in a systematic order. It arranges the chemical…
Q: Ph. 1) MCPBA 2) H₂O, H*
A: As per Guidelines, there are multiple questions, I can only answer one question this time. Please…
Q: 39. 2,5-diethyl-3-methylhexane is an incorrect IUPAC name for an alkane. Identify the correct name…
A:
Q: Which coefficient placed before HCI will balance the following equation? Zn + HCl → ZnCl₂ + H₂ a 2 b…
A: In this question we need to balance the given equation and then we need to answer the coefficient of…
Q: CI Br .... Ph NaSCH 3, acetone NaOCH3 CH3OH
A:
Q: To balance a redox equation in acidic medium, a student must make all these species balanced except…
A: A redox reaction is one which involves simultaneous oxidation and reduction reactions. It contains…
Q: A sample of zinc sulfide having a mass of 3.04 g was analyzed and found to contain 1.00 g of sulfur,…
A:
Step by step
Solved in 5 steps with 5 images

- Consider the following aromatic compounds a para-substituted compound?, the most reactive towards EAS? an ortho-substituted compound? does not react with RX, FeX3?i need help filling out the following SN2 reactions with appopiate reactants, products, or reagents,Consider the molecule: rate the priority functional groups from highest to lowest A. Alkyl chain B. Ketone C. Carbonyl D. Anhydride
- Compound X is insoluble in cold KMnO4, Br2 in CCl4, and conc. H2SO4. Compound X is most likely: a. an alkane b. none of these c. an alkene d. an alcohol e. an alkyl halide Indicate which of the ff. statements regarding nucleophilicity is incorrect. F- is more nucleophilic, hence, more reactive towards methyl iodide than Cl-. Second row elements are more nucleophilic than first row elements of comparable basicity. The rate of SN2 reaction may be markedly affected by the nucleophilicity of the attacking atom. Nucleophilicity is the affinity of a nucleophile to an electrophilic carbon Which of the following alkynes can be deprotonated by NaNH2 in liquid NH3? 3-Methylhex-2-yne Pent-2-yne 3-Methylbutyne none of these Hex-3-ynePlease solve correctly: Optio for Q1. a.)2-hexyne b.)3-methyl-1-pentyne c.)3-hexyne d.)1-hexyne e.)4-methy-1-pentyne Q2) a.)Alkenes react with nucleophiles and bases. b.)Alkenes react with electrophiles. c.)In the reactions of alkenes, the p bond is not always broken. d.)Alkenes contain double bonds from two sp-hybridized carbon atoms.Use the Hammond postulate to explain why (CH3)2C = CH2 reacts faster than CH3CH = CH2 in electrophilic addition of HX.
- Name the major product of the reaction between (S)-2-bromohexane and KCH3COO. Be specific with the configuration.Rank the following (in image one, the one with the chlorines) from most to least reactive in SN 1 reactions. How about ranking reactivity in nucleophilic addition (for example, Grignard) reactions in the second image?Explain Orientation Effects in Substituted Benzenes ?
- A hydrocarbon of unknown structure has the formula C8H10. On catalytichydrogenation over the Lindlar catalyst, 1 equivalent of H2 is absorbed. Onhydrogenation over a palladium catalyst, 3 equivalents of H2 are absorbed.(a) How rnany degrees of unsaturation are present in the unknown?(b) How many triple bonds are present?(c) How many double bonds are present?(d) How many rings ar e present?(e) Draw a structure that fits the data.What is the strongest IMF in pure 2-octanone? I know it is a ketone and has some polarity due to this, but the carbon chain so long, I wonder if london dispersion forces are stronger?Reactions that target the substituents on arenes can change their behavior in EAS reactions. Do not give handwriting solution.



