Q: Assuming 0.10 M Na2HAsO4, 0.40 M NaOH and 0.50 M HCl solutions are available in your laboratory. Usi...
A: A solution that contains both weak acid and its conjugate base acts a buffer solution. In this solut...
Q: Which wavelength of light (in nm) is emitted if an electronmoves from the conduction band to the val...
A: Valance band and conduction band are energy levels present in an atom. The valance band is the outer...
Q: Provide the reactant(s) that would give the following aldol condensation product from the possibilit...
A: Aldol condensation:The alpha hydrogen of carbonyl compounds is acidic in nature and it is removed as...
Q: 9. Calculate each of the following: a) the volume of 2.00 mol of chlorine at STP. b) the volume of ...
A: a) the volume of 2.00 mol of chlorine at STP. b) the volume of 4.00 mol of nitrogen at STP.
Q: Chlorine, Cl2, is produced commercially by the electrolysis of aqueous sodium chloride. The anode re...
A: 1 F = 96500 C Moles of chlorine = 1090 / 71 = 15.35 moles Total charge = 2 × 15.35 F = 2 × 15.35 ...
Q: What is the stereochemical relationship between the two stereoisomers shown? OH Но Но OH ´ OH Но OH ...
A: In this question we will Identify stereochemical Relationship between these two stereoisomer Compoun...
Q: Balance the following redox reaction in a basic solution PbO + CIO3 → PbO2 + Cl2 Note: answer by ind...
A:
Q: Titration Part Molarity HCI Initial Buret Final Buret Volume of HCI Reading Reading Trial 1 6 M 0.0m...
A: 4) Since, Ca form calcium hydroxide, Ca(OH)2. Ca (OH)2 = Ca2+ + 2OH- Since, 1 mol of Ca2+ is combine...
Q: List 2 types of detectors used in chromatography?
A: Two types of detectors which is used in chromatography 1)UV visible :- fixed wavelength Variable w...
Q: A compressed gas cylinder at 13,700 kPa and 23°C, is in a room where a fire raises the temperature t...
A:
Q: 175.0 g of an unknown metal gain 5.6 kJ of heat. The metal's temperature rises from 24.3 degrees C t...
A:
Q: Octene (C8H16) can be converted into octane (C8H18) following the reaction below. ΔH° for the reacti...
A: For spontaneous reaction ∆G is negative
Q: How many ml of a 1.5 M sugar solution are needed to make 250 ml of a 0.050 M solution
A:
Q: Question C
A: Osmotic pressure is given by formula, p = i c R T i is Van't Hoff factor c is concentration (molari...
Q: 2. How is the equilibrium position of this reaction affected by the following changes? (explain each...
A: This can be explained on Le Chatelier's principle. Le Chatelier's principle is used to predict the e...
Q: Write the net ionic equation for the following reaction: CuBr2 + AgNO3 → Cu(NO3)2 + AgBr
A:
Q: 1.Convert each of the following into the unit specified. 125 mmHg into atmosphere (atm): Answer: ___...
A: Pressure we can measure in different units like atm, mmHg, Pascal, bar, torr etc. 1 atm = 101325 Pa...
Q: Use the angular overlap model, calculate the orbital energies (π-donor) for trigonal pyramidal (TP)....
A: Angular overlap model is a theory which estimates the bonding strengts and energies based on the abi...
Q: 3. Typically, increasing the temperature at which a reaction occurs results in: (a) an increase in t...
A: The details solution for this reaction is provided below in attach image.
Q: Please show all steps clearly. Thank you. Determine the equilibrium constant for the following reac...
A:
Q: Answer
A: Physical change: In a physical change the physical state of matter changes. In this change different...
Q: Two stereoisomers are shown side-by-side. The isomer on the left is R, R, R. What is the R/S configu...
A: R and S nomenclature is used to label the stereocenters of a chiral compound. If the arrow points in...
Q: # moles of c2h2 that will burn in the presence of 1.43 mol o2?
A: Given that Moles pf O2 gas = 1.43 mole when C2H2 burns in presence of oxygen it is known as combus...
Q: How many unpaired electrons are in the low-spin octahedral complex [Co(CN),Jª¯? Provide your answer ...
A: The no. Of unpaired electron in given complex = 1 (one).
Q: Consider the following equilibrium reaction at 25°C: 2SO2 (g) + O2 (g) 2SO3 (g) At equilibrium, [SO...
A: The provided reaction is- 2SO2 (g) + O2 (g) ⇆ 2SO3 (g) Here, Stoichiometric coefficient of SO2=2 Sto...
Q: At 25°C the volume of a gas sample is 380 mL. To what temperature must the gas sample be heated so i...
A: Given that, Initial temperature of gas (T1) = 25°C = 25+273.15 = 298.15 K Initial Volume of gas (V1...
Q: 6. The second-order reaction 2 NO, 2 NO + 0 has a rate constantk 0.63 M's', If the initia. concentra...
A:
Q: 2. Predict whether each of the following oxides is ionic or molecular? (a) SnO2 (b) Al½O; (с) СО2 (d...
A: Ionic compounds are formed by combining a metal and a nonmetal Molecular compounds are formed by t...
Q: Which part of the GC equipment does separation takes place?
A: GC (Gas chromatography) is an analytical technique used to separate the chemical compounds of a samp...
Q: 7. Which of the following carbocations is most stable (circle your answer)
A: In this question, we will Identify which Compound have most stable. You can see details explanation,...
Q: What is the expected major product of the boxed reaction? 1. excess CH,I 2. AGOH NH2 3. heat A. D E
A: The answer is given as follows
Q: Answer ALL 12 questions, in the png files provided, completely showing steps/work done to solve each...
A: Given dissociation reaction of propanoic acid in water as ; HPro(aq) + H2O(l) <-----> H3O+...
Q: The oxidation of ammonia is a reversible exothermic reaction at equilibrium that proceeds as follows...
A: Given that O2 is lowered
Q: What type of reaction is Na +H2O —>
A: Ans The reaction of the water with sodium metal is a redox type reaction in which Na oxidized and h...
Q: State 2 properties of detectors used in chromatography?
A: A detector used in chromatography is basically used to detect the components of mixture which is bei...
Q: Write whether it will be E1, E2, SN1, and/or SN2 Show the mechanism(s) Write the MAJOR product(s) St...
A: The answer is given as follows
Q: A chemist adds 85.0 mL of a 1.18 x 10 mM mercury(II) iodide (HgI,) solution to a reaction flask. Cal...
A:
Q: What percentage of the strontium- 90 that was made 1945 still exists now in the year 2021? The half-...
A: The decay constant for Sr-90 can be calculated as follows: k = 0.693thalf-lifek = 0.69328.9 yearsk =...
Q: Find the smallest wavelength of the spectrum of the Paschen's series (ns -> 3s). Use Rydberg cons...
A: Paschen Series appear when transition occur from higher energy level to lower level,n=3 Shortest wav...
Q: Thank you
A: Answer:- This question is answered by using the simple concept of saccharides and the bonding betwee...
Q: I am trying to balance this redox equation: CrBr3 + HNO3----> Cr2O72- + BrO4-1 + NO2 (in acidic s...
A: Step 1. Write down the unbalanced equation ('skeleton equation') of the chemical reaction. All react...
Q: 5. Rank the following groups in order of increasing leaving group ability (i.e. rank from worst LG t...
A: Order of increasing leaving group ability:
Q: need help with this question
A: Atomic mass: Atomic mass of an element is the total number of protons and neutrons present in the nu...
Q: Draw the Lewis structures for the following chemicals, predicts the angle between the bonds and say ...
A: For a. The molecular structure of KrF2 is linear shaped. so, Bond angle is: 180∘ Hybridization of t...
Q: Cesium Chromate + strontium phosphate -->
A: Cesium Chromate is Cs2CrO4 strontium phosphate is Sr3(PO4)2
Q: Possible monochlorinated isomers of methyl-2, butane are: a) 1 b) 2 c) 3 d) 4
A: This question is related to finding out the total number of isomers of given compound on monochlorin...
Q: These three statements about Substances A, B, and C are true: The average kinetic energy of Substan...
A: As we know kinetic energy of any substance is due to the motion of particles .And the solids have m...
Q: 2. Typically for a gas-phase reaction, increasing the pressure at constant temperature results in: a...
A: For a gas-phase reaction, increasing the pressure at constant temperature results in an increase in ...
Q: Using the equation MnO(OH)2(s)+2H2SO4(aq)->Mn(SO4)2+3H2O(1) and Mn(SO4)2+2I^-(aq)->Mn^2+(aq)+I...
A: Interpretation - To determine the number of mole of I2 when here 2 mole of MnO(OH)2 are formed usin...
Q: The important result from the photoelectric experiment is the confirmation that O light's energy dep...
A: Photoelectric effect is explaine on the basis of particle nature of light. In this effect when light...
*Please answer leter D and E only. Thanks!
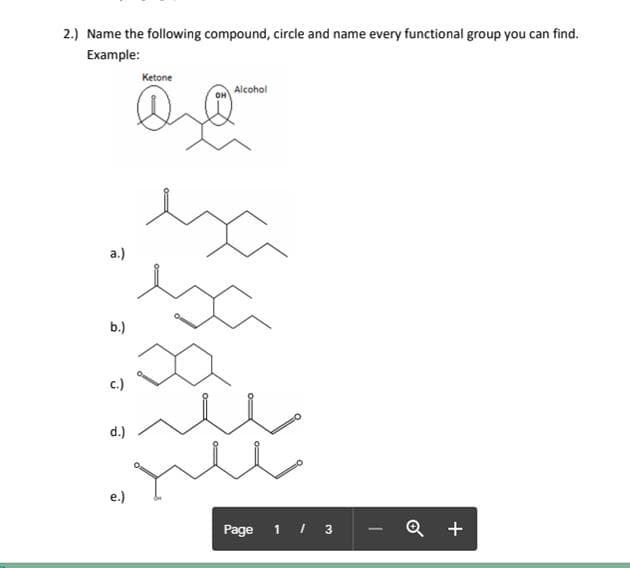
Step by step
Solved in 3 steps with 2 images

- Draw the structures based on iupac name digitally (use chem-space .com for easy drawing then screenshot)I need help answering the following. Organic chemistry seems a little confusing.(c) There is one (1) other positional isomer of X missing for reaction II. Draw the structural formula of this isomer.
- The second question was incorrect. "The IUPAC nomenclature of benzene compounds primarily will use benzene as the root name. However, common names for substituted benzene compounds exist that use the base names from its derived compounds. The principal functional group will serve as the common root name." Would you mind trying again please? Thank youName the following compound. I only need part D,H, I5. please write funtional group and iupac name, u can look at example

