Chapter3: Statistical Tests With Excel
Section: Chapter Questions
Problem 7P
Related questions
Question
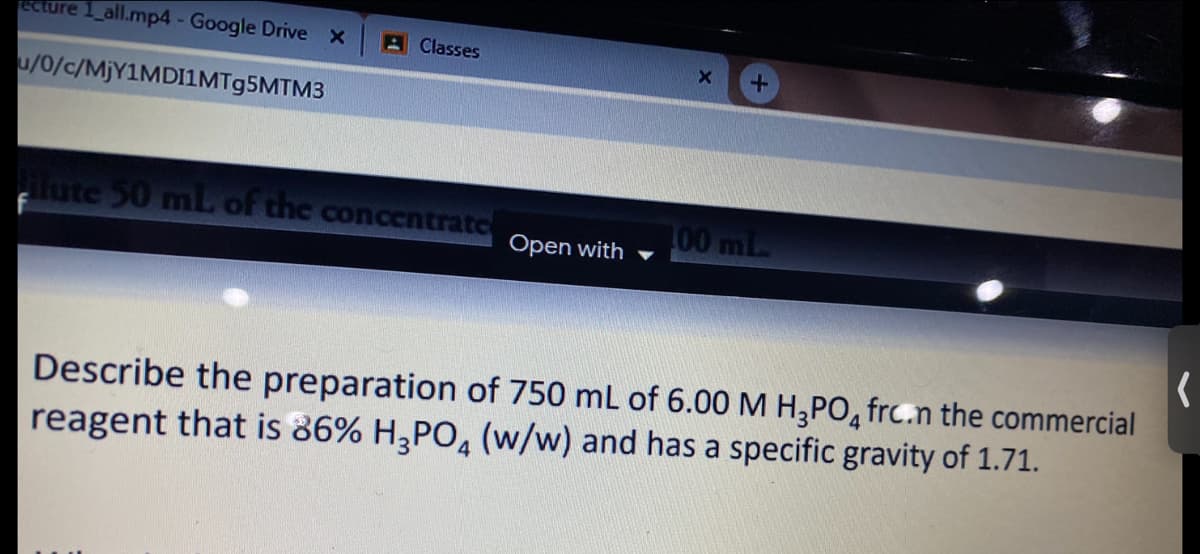
Transcribed Image Text:ECture 1_all.mp4 - Google Drive X
A Classes
u/0/c/M)Y1MDI1MT95MTM3
lute 50 mlL of the concentratc
00 mL
Open with
Describe the preparation of 750 mL of 6.00 M H;PO, frc.n the commercial
reagent that is 86% H2PO, (w/w) and has a specific gravity of 1.71.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you



