Q: A mixture of gaseous reactants is put into a cylinder, where a chemical reaction turns them into gas...
A:
Q: A reaction at 20.0 °C exhibits a rate constant of 0.555 s–1. If the reaction is performed at 60 °C, ...
A: Consider the given information is as follows; Initial temperature (T1) = 20.0°C = 20.0 + 273.15 = 29...
Q: 3. Use the internet and your data table to figure out which of the following POLAR COVALENT COMPOUND...
A:
Q: Hydrochloric acid can be prepared by the following reaction: 2NACI(s) + H2S04(aq) 2HCI(g) + Na2SO4(s...
A: 2NaCl (s) + H2SO4(aq) -----> 2HCl(g) + Na2SO4(s) Number of moles of H2SO4 = 2 mol Mass of NaCl = ...
Q: Use the following calorimetrically dtermined enthalphy changes to predict the standard enthalpy chan...
A:
Q: What is electronegativity?
A: The tendency to attract shared pair of electron is called electronegativity. Fluorine is most elect...
Q: Consider the following molecules: phosgene formaldehyde 1,1,1-trifluoroacetone (also known as ...
A: Given molecules are : a). phosgene b). formaldehyde c). 1,1,1-trifluoroacetone (also known as triflu...
Q: 2. A metal with atomic weight of 63.65 g/mol crystallizes with a face-centered cubic unit cell. The ...
A:
Q: A sample of gas at 27°C has a pressure of 0.7 atm. Compute for the new temperature in Kelvin at 740 ...
A:
Q: A sample of polystyrene, which has a specific heat capacity of 1.880 J-g .°c', is put into a calorim...
A: Given data,Specific heat capacity of polystyrene=1.880J.g-1.oC-1Mass of water=250.0gInitial temperat...
Q: 4. Use the internet and your data table to figure out which of the following NON-POLAR COVALENT COMP...
A: Answer of given question is as follows
Q: 10 draw the condensed and line bond structure of the compound
A:
Q: glass of skim milk supplies 0.1 mg of iron, 8.5 g of protein, and 1 g of carbohydrates. A quarter po...
A: The data has been tabulated and solved in the following step.
Q: Consider the skeletal structure of 1-Isopropyl-2-methylcyclohexane. Identify the number of hydrogen ...
A: Identify the number of hydrogen atoms bound to each carbon in the structure.
Q: using the following thermochemical equation determine the % yield form the following reaction if 3.0...
A: Given mass of octane =3.080×10=30.80 g Molar mass of octane C8H18=8×12+18×1=114g/mol Mass of CO2 pro...
Q: Supposed the reaction: X(s) + 2Y(g) → Z(l) is exothermic. At what temperatures is it spontaneous? a...
A: For the reaction., X(s) + 2Y(g) → Z(l)
Q: What is required for a reaction to occur? O a. collisions O b. collisions with sufficient energy O c...
A: This question is related to collision theory. Collision theory is used to predict the rate of chemic...
Q: Draw the molecular mass fragments for the following m/z rations. Show all the atoms (including) all ...
A:
Q: An imaginary machine takes 9,000 J of heat from a hot reservoir and 3,000 J of heat from a cold rese...
A: Given : An imaginary machine takes 9,000 J of heat from a hot reservoir and 3,000 J of heat from a c...
Q: n a quality control lab at the Fairlife brand (owned by the Cocaine-Cola company), a technician prep...
A: Answer: This is question based on Raoult's law which tells that on dissolving non-volatile solute in...
Q: classification of carbon of atenolol
A: The correct answer is given below
Q: Methanol (CH3OH) is converted to bromomethane (CH3B1) as follows: CH3OH + HBr CH3B + H20 If 12.23 g ...
A: Given equation is : CH3OH + HBr ---------> CH3Br + H2O Mass of CH3Br produced = Actual yield = ...
Q: Captain Marvelibrium discovered a figure with data from an experiment left by Le Châtelier Flipdime....
A: Value of equilibrium constant is calculated in the following step.
Q: Instruction: Using the given molar mass, Calculate the mass-mole and number of entities of the given...
A: Note: As per company policy we are supposed to attempt first question only. Please repost other ques...
Q: Draw the following compounds: C4 epimer of Tagatose
A:
Q: An action will release HOCI to water. Determine expressions for each of the equilibrium concentratio...
A: Reaction of HOCl with water and find equilibrium expression and pH expression.
Q: 7) 4-ethylcycloheptene
A:
Q: A cylindrical glass tube 20.0 cm in length is filled with acetone. The mass of the acetone to fill t...
A:
Q: What is the pH of a 0.200 M H2S solution? Ka1 of H2S = 8.9 × 10−8 and Ka2 = 1 × 10−19
A: H2S is diprotic acid
Q: A scientist measures the standard enthalpy change for the following reaction to be -234.6 k) : 2502(...
A: Answer: To solve this question we have to use the relation between enthalpies of formation of reacta...
Q: H2SO4 SO3 d) -HO- H* но- H* H* H20
A: Answer of the question given below,
Q: What would be the implication if there are a lot of acidic constituents in the hydrocarbon? explain ...
A: Hydrocarbons are formed by H and C.
Q: The solutes below are soluble in water. Which solute(s) could you increase the solubility of in the ...
A: Pressure is directly proportional to solubility. Increasing the pressure leads to the increase in co...
Q: Boyle's Law Wrap-Up Use the knowledge of Boyle's Law that you have gained from your reading and calc...
A:
Q: U STATES OF MATTER Understanding how average molecular speed scales wit Rank the samples of gas desc...
A:
Q: Order of reaction with respect to [Fructose]: ; with respect to [OH]: K key, [COH2O,][ OH][ MB] Theo...
A: Consider a chemical equation as follows: aA+bB→cC Rate Law: It is defined as the relationship betw...
Q: The figure shows the skeletal structure of 2,5-dimethylhexa-1,4-diene. Identify the number of hydrog...
A:
Q: A mixture of gaseous reactants is put into a cylinder, where a chemical reaction turns them into gas...
A: As energy is absorbed so the given reaction is endothermic reaction. In this process energy is abso...
Q: mass
A:
Q: thermometer -1 A sample of glass, which has a specific heat capacity of 0.670 J-g .°C is put into a ...
A:
Q: Which of the following would be heterogeneous? A mixture of sand and water. A mixture ...
A: A heterogeneous mixture is a mixture with non-uniform composition or we can say it contains two diff...
Q: thermometer A 56.2 g sample of polystyrene is put into a calorimeter (see sketch at right) that cont...
A:
Q: xamine the curved arrow below, and answer the question in the box. If the curved arrow creates a rea...
A: Resonance structures are sets of Lewis structures that describes delocalization of electrons in poly...
Q: Draw a better resonance contributor than the one shown below. Interactive 3D display mode H;C NH Dra...
A: Resonance structure: If the Lewis structure of a molecule or ion cannot explain by a single structur...
Q: Long answers A. Define and explain the mode of action when used in hair conditioners Quaternary co...
A: Given :- A question based on general chemistry that is to be accomplished.
Q: A catalyst: a. changes the equilibrium concentration of the products. b. always increases the activa...
A: Answer A catalyst effect the both reaction forward reaction and back...
Q: Find the standard molar enthalpy for the reaction C + 1/2 O2 -> CO given that C + O2 -> CO2 Delta H ...
A:
Q: 6. Match the correct non: alkane functional group to the molecule in the table "There may be more th...
A:
Q: Calculate the molar concentration of ethanol, C2H5OH, in an aqueous solution that contains 3.40 g of...
A: Note : Since you have posted multiple questions, we are entitled to answer the first only. Please re...
Q: acid base titration
A: In this question we have to explain that how does a redox titration differ from an acid base titrati...
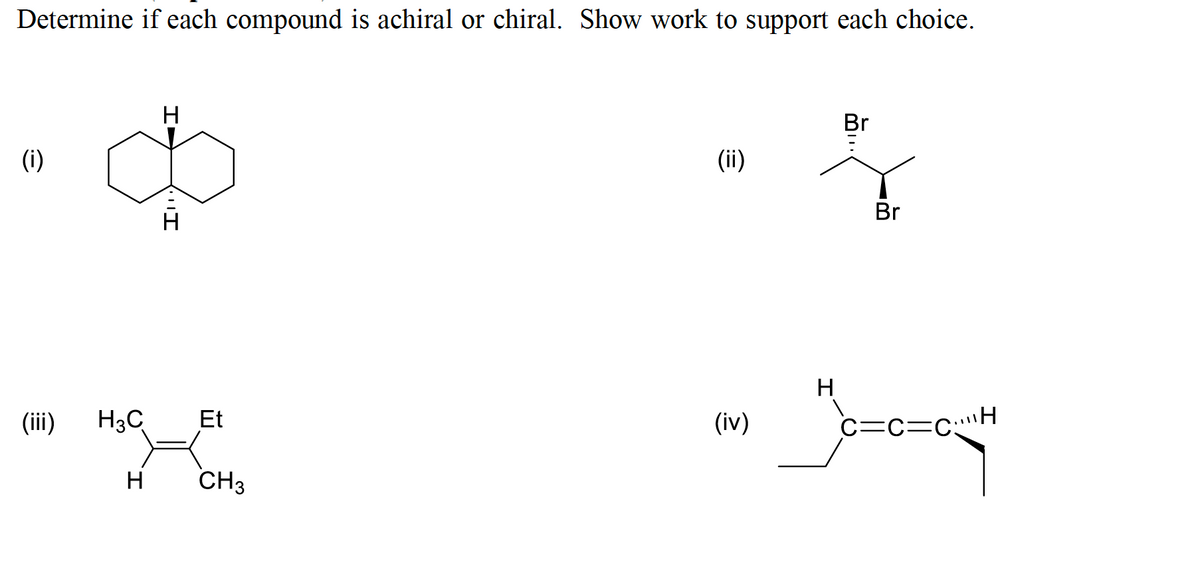
Pls help.
Step by step
Solved in 2 steps with 4 images

- Draw three-dimensional representations of the following compounds. Which have asymmetric carbon atoms? Which have no asymmetric carbons but are chiral anyway? Use your models for parts (a) through (d) and any others that seem unclear.1,3-dichloropropadieneWhat is configuration (R or S) for the centers and the enantiomer for the whole isomer? Also tell if they are Z or E.1. Determine whether the molecule is chiral or achiral 2. Oseltamivir phosphate (sold under the trade name Tamiflu®) is a prescription antiviral drug that is used in the treatment of both Influenza virus type A and Influenza virus type B. Identify all the stereocenters by labelling with an asterisk (*).
- Which compound(s) does not posses a plane of symmetry ? A.) III and IV B.) II, III, and IV C.) I, II, and V D.) I, III, and IVA) For a molecule with three chiral carbons the possible number of stereoisomers is: 4, 8, 6 or 2 B)Which of the following has a S configuration: (Image)How would I go about listing the groups from highest priority to lowest priority, and then determining the absolute configuration of the chirality center?
- Construct a model of methane (CH4) using MolView and answer the following: Can it be superimposed on its mirror image? (Y/N) Does methane contain a plane of symmetry? (Y/N) Is methane chiral? (Y/N)Why does "Zincblende" have a F(-4)3m space group designation? F stands for face centered and the 4-overbar means 90o rotation followed by inversion through the center gives the starting position, but what does the 3m mean?The molecule below has 2 stereocenters. TRUE OR FALSE
- Chloramlhenicol, a powerful antibiotic in 1947 from the streptomyces venezuelae bacterium, is active against a broad spectrum of bacterial infections and is particularly valuable against typhoid fever. Assign R or S configurations to the chirality centers in chloramphenicolHi, I need help with 7c. For 7a I got 3 asymmetric centers and for 7b I got 8 possible stereoisomers.1.What is the absolute configuration of this isomer of 2,3-dihydroxybutanal? pls explain.2. What is the absolute configuration of the enantiomer of the isomer?

