Determine the atomic mass of potassium, given the information about potassium's stable isotopes. sotope K-39 K-41 mass 38.96370 amu |40.96183 amu percent abundance 93.258% 6.730% First, select the math steps needed to perform this calculation correctly. 39.96370 amu + 40.96183 amu 38.96370 amu x 0.5000 40.96183 amu x 6.730 38.96370 amu x 0.93258 40.96183 amu x 0.06730 38.96370 amu x 93.258 40.96183 amu x 0.5000 Next, perform the calculation and enter your numerical answer here. amu (Enter this answer with three decimal places. It must be exact and will not match the value on the periodic table.)
Determine the atomic mass of potassium, given the information about potassium's stable isotopes. sotope K-39 K-41 mass 38.96370 amu |40.96183 amu percent abundance 93.258% 6.730% First, select the math steps needed to perform this calculation correctly. 39.96370 amu + 40.96183 amu 38.96370 amu x 0.5000 40.96183 amu x 6.730 38.96370 amu x 0.93258 40.96183 amu x 0.06730 38.96370 amu x 93.258 40.96183 amu x 0.5000 Next, perform the calculation and enter your numerical answer here. amu (Enter this answer with three decimal places. It must be exact and will not match the value on the periodic table.)
Chemistry by OpenStax (2015-05-04)
1st Edition
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Chapter2: Atoms, Molecules, And Ions
Section: Chapter Questions
Problem 23E: Average atomic masses listed by JUPAC are based on a study of experimental results. Bromine has two...
Related questions
Concept explainers
Atomic Structure
The basic structure of an atom is defined as the component-level of atomic structure of an atom. Precisely speaking an atom consists of three major subatomic particles which are protons, neutrons, and electrons. Many theories have been stated for explaining the structure of an atom.
Shape of the D Orbital
Shapes of orbitals are an approximate representation of boundaries in space for finding electrons occupied in that respective orbital. D orbitals are known to have a clover leaf shape or dumbbell inside where electrons can be found.
Question
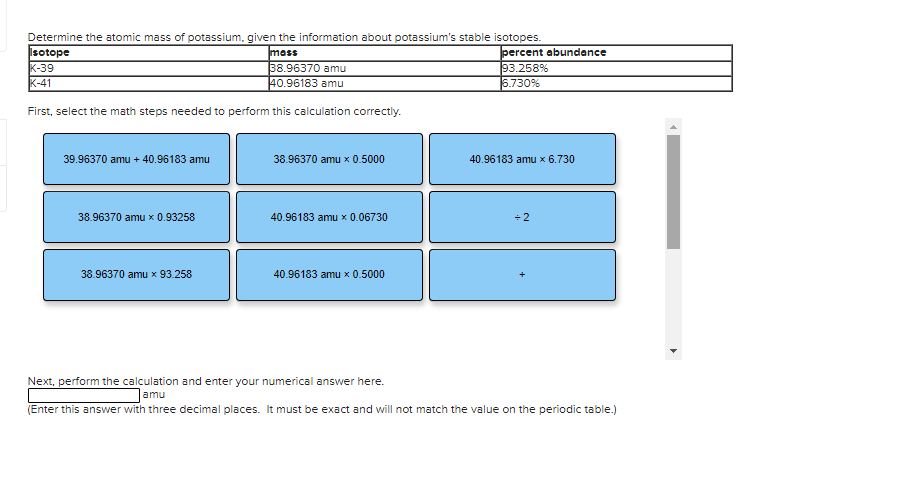
Transcribed Image Text:Determine the atomic mass of potassium, given the information about potassium's stable isotopes.
Isotope
K-39
K-41
mass
38.96370 amu
40.96183 amu
percent abundance
93.258%
6.730%
First, select the math steps needed to perform this calculation correctly.
39.96370 amu + 40.96183 amu
38.96370 amu x 0.5000
40.96183 amu x 6.730
38.96370 amu x 0.93258
40.96183 amu x 0.06730
÷ 2
38.96370 amu x 93.258
40.96183 amu x 0.5000
Next, perform the calculation and enter your numerical answer here.
amu
(Enter this answer with three decimal places. It must be exact and will not match the value on the periodic table.)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning