Q: What is the formula of the ionic compound formed between Pb2+ and O?
A:
Q: Decide which intermolecular forces act between the molecules of each compound in the table below.…
A: Compounds which contain non-polar covalent bond contain Dispersion forcesCompounds which contain…
Q: What is the correct name for the following compound? CH3 OH H3C-CH-CH-CH3
A:
Q: HNO3(aq) + + Complete ionic equation: + Net ionic equation: + b. Balanced formula equation: Net…
A:
Q: Search ChP/USP/EP/BP..., find monograph of FLUOROURACIL; What kind of method used for "Impurities F…
A: IntroductionFluorouracil (also known as 5-fluorouracil or 5-FU) is a medication used in the…
Q: Given three (3) titration curve for acid-base titration. Identify a suitable acid-base pair for the…
A: Since you have posted multiple questions, we will answer all three subparts of first question only.…
Q: Consider three isolated ions: Mg²+, O²- and Nat. Which is true about these ions? a. All of the ions…
A: Atomic radius is the distance between the central nucleus and the outermost electron. Atomic radius…
Q: Write a condensed electronic configuration, for an imaginary element X with atomic number 120
A: Condensed electronic configuration includes the chemical symbol of the nearest noble gas of the…
Q: 12. Select the major product(s) of the following metathesis reaction. A. 1 only 3 B. 2 only HOOC C.…
A: Grubbs catalyst: This catalyst is used for olefin metathesis.
Q: Question: Consider a hypothetical chemical reaction where molecule A reacts with molecule B to form…
A: The slow rate of a hypothetical exothermic reaction, even at high temperatures, can be attributed to…
Q: Pls help ASAP on all pls i beg.
A: There can be different types of reaction in organic compounds. Oxidation reaction : it is a reaction…
Q: Fill in the major products to complete the transformations. Be sure to pay careful attention to…
A: It is based on the concept of reactivity of alkene. Here we are required to find the product of the…
Q: Draw a mechanism for the following reaction. OH H+ + H₂O
A: This is an example of SN1 reaction that proceeds through carbon cation intermediate. The carbocation…
Q: What is the formula for barium phosphate
A: Symbol of barium : BaBa is present in 2nd group in periodic table. It has +2 charge
Q: A particular reaction is found to be second order. The reactant has an initial concentration of 1.12…
A:
Q: In finding the pH of the solution containing amphiprotic salts, diluted solutions contain…
A: Concentration of NaHCO3 solution = 0.1 (M)Ka1 = 1.5*10-4, Ka2 = 4.69*10-11
Q: A researcher raises the temperature from 26.1 to 40.1 o C and finds that the rate of the reaction…
A: Initial temperature = 26.1oCFinal temperature = 40.1o CInitial rate = kFinal rate = 2kR = 8.3145…
Q: Predict the structure of the major product for the reaction shown. 00 CH₂CH₂CH₂COCCH₂CH₂CH₂ AICI
A: Catalyst activation: Aluminum chloride, a Lewis acid catalyst, is typically activated by reacting it…
Q: When 6.919 grams of a hydrocarbon, CxHy, were burned in a combustion analysis apparatus, 21.20 grams…
A:
Q: the behaviour of sponge material under stress
A: When a sponge material is subjected to stress,it's behaviour can depend on different factors such as…
Q: Consider a hypothetical reaction where one mole of substance A reacts with one mole of substance B…
A: Introduction:The hypothetical chemistry question involves calculating the enthalpy change (ΔH) for…
Q: You have 150.0 mL of a 0.877 M solution of Ce(NO₂),. X What mass (in grams) of Ce(NO₂), would be…
A: The volume of Ce(NO3)4 is 150.0 mL.The concentration of Ce(NO3)4 is 0.877 M.To find:a. The mass of…
Q: A 1.00 L solution contains propanoic acid (CH3CH₂COOH) with a concentration of 0.300 mol/L, and…
A:
Q: Balance the following equation: A C5H12 (9) + O2 (9) CO₂ (g) + H₂O (9)
A: -> In balanced equation number of each atom from both sides are equal.
Q: analytical chemistry. Given the following titration curve, what is the suitable indicator for this…
A: The above question is based on analytical chemistry . We have to discuss about the roll about the…
Q: 3. Match each of the following IR spectra with the appropriate molecule. A B с TRANSMITTANCE…
A: We have to determine the structure of the molecule from the given infrared (IR) spectrum.By using…
Q: or 500.0 mLmL of a buffer solution that is 0.165 M� in CH3CH2NH2CH3CH2NH2 and 0.100 M� in…
A:
Q: What is the pH of a 1.0 L solution that is 0.50 M PO43- and 0.50 M HPO42-? (The pKa values for…
A: As per the Henderson–Hasselbalch equation:pH = pKa + log [PO4-3]/[HPO4-2]pKa = 12.4 as ( third…
Q: Suppose 0.476 g of potassium acetate is dissolved in 50. mL of a 72.0 m Maqueous solution of…
A: It is based on the concept of concentration.Here we are required to find the Molarity of potassium…
Q: What is the IUPAC name of propyl isobutyl ketone? a) 4-octanone b) 2-methyl-4-heptanone…
A: We have to draw the structure of propyl isobutyl ketone and then we have to predict the IUPAC name…
Q: What is the concentration of it if a solution has a starting concentration of 0.100 M H₂C₂O4 and…
A:
Q: A quantity of liquid methanol, CH3OH, is introduced into a rigid 2.45-L vessel, the vessel is sealed…
A: The concentration of any substance at equillibrium can be calculated using an ICE diagram. This is…
Q: HO NaOH OH H₂O* H₂SO4 KMnO4 0₁ Za Acid major product minor product /OsO4, NaHSO,, water CHCI KOHT 1.…
A: Alcohol is dehydrated using an acid catalyst under heating conditions.The dehydration proceeds…
Q: Carbon monoxide , CO(g),can react with hydrogen, H2 (g), to produce methane, CH4 (g), and water…
A: Using Hess's Law, we were able to combine and manipulate the given equations to obtain the…
Q: Which of the following is true of tautomers? a) They are constitutional isomers that differ in…
A: Tautomers: These are the constitutional isomers which differ in the position of one atomic nuclei…
Q: The isoelectric point of the tripeptide Gly-Ser-Ala occurs at a value below pH 7 at pH 7 at a value…
A: The isoelectric point (pI) of a tripeptide such as Gly-Ser-Ala is determined by the presence of…
Q: Provide the structure of three compounds with the molecular formula C4H₂O that give an absorption…
A: A question based on IR spectrum of organic compounds. An empirical formula is given for which 3…
Q: Part D [s] (a) An unknown solution of sodium hydroxide was standardized by titrating it with a known…
A: Answer:-This question is answered by using the simple concept of stoichiometry which involves the…
Q: 3. For each of the following molecules rank the protons from highest to lowest chemical shift. See…
A: Rank the given protons according to their highest to lowest chemical shift.Based on standard…
Q: for the reaction 2 A(aq) B(aq) + C(aq), the standard Gibbs free enthalpy change is 1.29 kJ at 25°C.…
A: Initial concentration of A = 0.554 MInitial concentration of B = 0.307 MInitial concentration of C =…
Q: What is the pH of a 1.0 L solution that is 0.50 M PO4³- and 0.50 M HPO4²-? a. If 0.01 moles of OH is…
A:
Q: A weak acid HX has Ka = 4.0 x 10-6. What is the concentration of the anion X- in a solution in which…
A:
Q: In the periodic table, which element has the lowest electronegativity value, is a transition metal,…
A: Introduction:The periodic table is a fundamental tool in chemistry that organizes elements based on…
Q: The London dispersion force between two neon atoms compared to the London dispersion force between…
A: London Dispersion Forces (LDFs) are the weakest type of intermolecular force that occur between…
Q: Estimate KI for a competitive inhibitor when [I] = 5 mM gives an apparent value of KM that is three…
A: We have to estimate KI for a competitive inhibitor so we will use Michaelis-Menten equation which is…
Q: ant values if needed for this question. The stopcock connecting a 1.63 L bulb containing carbon…
A:
Q: ОН X N₁ N= Zn ОН
A: As it is a complicated macromolecule zinc coordination compound IUPAC nomenclature wouldn't be…
Q: How many of the following species can be represented with the resonance structures? CS2, POF3, NH3,…
A: When a single lewis structure is incapable to explain all properties of a molecule then we need two…
Q: Explain the effect that each of the following changes would have on the Rate of Reaction in this…
A: To assess the effect of each change on the rate of reaction in the given experiment, we will…
Q: Which compound or compounds in the following groups is (are) expected to be insoluble in water:…
A: Soluble salt : If atleast 1g of a compound can be dissolved in 100ml of water at 298 K, it is called…
Determine the name of the following structures:
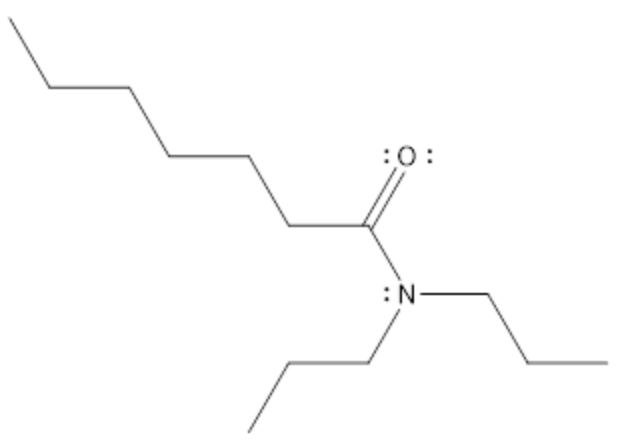
Step by step
Solved in 3 steps with 2 images


