Q: Which of the following statements best describe a minimum azeotrope? I. Minimum azeotrope boils at ...
A:
Q: Here are the common abbreviation of compounds found in commercial products and the laboratory. Find ...
A: 1) MSG (added in food to give an umami flavor) - Name - monosodium glutamate. Chemical Formula - C₅H...
Q: Question 1 II A в D What is the design of figure II? diode array spectrophotometer single beam spect...
A: Spectrophotometer is an instrument which is used to measure intensity of light spectra absorbed/ tra...
Q: Write the formula of the ff a. DIAMMINEDICHLOROPLATINUM II b. potassium diamminetetrachlorochromate ...
A:
Q: A spot of black ink was placed on a strip of fiter paper. The chromatogram was developed using an ap...
A: Chromatography is based on the principle that two substances are dissolved in the same solvent but t...
Q: а. b. C. d.
A: Vitamin A also known as retinol. It is a fat soluble vitamin. It is highly essential vitamin for hum...
Q: Solve the preparation of 375ml of 40 ppt Cl-(parts per thousand) from 0.01 M PbCl2
A: Ppt means parts per thousand. 1 ppt of a substance means 1g of the substance in 1000g of its solutio...
Q: 4) Calculate the mass of copper (II) sulfate pentahydrate needed to make 250.0-mL of a 1.25 M soluti...
A: Please note- As per our company guidelines we are supposed to answer only one question. Kindly repos...
Q: 5. Wate C. Identify the following mixtures. Write SOL for Solution, SUS for suspension. and COL for ...
A: Please find your solution below : A solution is defined as a homogeneous mixture of two or more comp...
Q: How do you define chemistry?
A: How do you define chemistry ?
Q: Pheromones are a special type of compound secreted by the females of many insect species to attract ...
A: Molar mass of pheromone = 282 g/mol Mass of pheromone = 1.5 x 10-12 g
Q: PART 1: Write the half - reaction of each redox reaction and determine the oxidizing and reducing ag...
A:
Q: Determine the mass of oxygen used produced when 0.64 grams of octane (C8H18)undergoing a combustion ...
A: Given, Reaction for combustion of octane: 2C8H18(l) + 25O2(g)➝ 16CO2(g) + 18H2O(g) Mass of octane (C...
Q: In the presence of excess thiocyanate ion, SCN-, the following reaction is first order in iron(III) ...
A: As we can see here, SCN- is in excess, this is a type of pseudo 1st order reaction in which one of t...
Q: -moles of NH: wi
A: Since you have asked multiple questions, we will solve the first question for you. If you want any s...
Q: The atmospheric surface pressure on Venus is 90 bar and is composed of 96% carbon dioxide and approx...
A: Given:- Pressure of Venus surface (P) = 90 bar Composition of CO2= 96% Composition of other gases = ...
Q: Calculate the energy of blue light that has a wavelength of 400 nm. 3 significant figures
A: The relation between energy and wavelength is Energy=hc/wavelength plank constant h=6.626x10-3...
Q: The concentration of acetic acid (pKa =4.75) in vinegar is about 1.0 M. With this information, what ...
A: Acetic acid is a weak acid and it does not dissociate completely into ions. The pKa of acetic acid i...
Q: Below are the proposed elementary steps and enthalpies for the mechanism of burning coal, C(s). (1,...
A: Given mechanism: Step 1: C(s) + H2O(g) ⇌ CO(g) + H2(g) (fast) Step 2: CO(g) + H2O(g...
Q: Given the following reactions N2(g) + 202(g) → 2NO2(9) AH = 66.4 kJ 2NO(g) + O2(g) → 2NO2(g) AH = -1...
A:
Q: A sample consisting of 2.00 mol He is expanded isothermally at 0° C fron 5.0 dm^3 to 20.0 dm^3 (i) r...
A: Reversible changes are infinitesimally small and values of energy , work done etc. are small. In con...
Q: 5) Identify which of the following reactions are unfavorable and explain why: A) B) C) D) :o: CH3 +
A: In nucleophilic substitution reaction, Nucleophile has to attack to the electrophilic centre . Here ...
Q: Systematic naming
A:
Q: 3. Ozone 9. Hydrogen Fluoride 10. Table Sugar 4. Acetone 5. Water C. Identify the following mixtures...
A:
Q: Given the following reactions Fe2O3(s) + 3CO(s) → 2Fe(s) + 3CO2(9) AH = -28.0 kJ 3Fe(s) + 4CO2(s) → ...
A: Given Reaction:- Fe2O3(s) + 3CO(s) -----> 2Fe(s) + 3CO2(g) ∆H = -28.0kJ ..... (1) 3Fe(s) + 4...
Q: Which part in the figure can be a grating or prism? O A O B OD
A: Instruments for analyzing light and for determining the identity and structure of materials that emi...
Q: Using the given data above, determine the following information: What is the Amax of Jerry? O 650 nm...
A: Wavelength maximum (λmax) is defined as the wavelength at which maximum absorption occurs.
Q: e type of reaction bas
A:
Q: A solution containing 0.584 mole/L of Cuso4 and had a 0.510 absorbance reading in a 1.0cm cell at 52...
A:
Q: Given below is a diagram of separation of two components of a mixture in a chromatographic column. F...
A: Given above is a diagram of separation of two components of a mixture in a chromatographic column. F...
Q: Which of the following is/are TRUE regarding instrumental methods of analysis?
A: Option D All of the above.
Q: Which property of laser light is NOT correctly defined/described? extremely bright: high power at on...
A: Laser light have the following properties Extremely bright. Monochromatic. Collimated. But not a...
Q: Which of the following statements best describe a minimum azeotrope? 1. Minimum azeotrope boils at h...
A: Azeotrope can be defined as mixture of liquids that has a constant boiling point The boiling point o...
Q: A 2.15 L volume of KOH of an unknown concentration is mixed with 2050 mL of 0.445 M NaOH. The result...
A: If same nature of two solutions are mixed with different concentrations and volumes then the resulta...
Q: Draw the reaction mechanism of a simple addition reaction between ethene and bromine in CCl4. Label ...
A: Alkene reacts with bromine in CCl4 solvent. The double bond breaks and a bromine atom because attach...
Q: Draw an example of each of the following molecules a) Alcohols b) Aldehydes c) Carboxylic acids d) A...
A:
Q: 2. Find the mass of fructose (C6H12O6) present in a solution containing 9,370.0 mg acetic acid to el...
A:
Q: Phosphoric acid is often added to colas to give them the tart flavor people like. Coke Classic has a...
A: Phosphoric acid is a triprotic acid. It undergoes three dissociations as follows: H3PO4 <------&g...
Q: What is the pH of a 0.196 M aqueous solution of ammonium chloride, NH4Cl ? pH = __________ This sol...
A:
Q: The ethylene glycol (d = 1.11 g/mL; c = 2.42) in a car radiator cools from 37.0°C to 25.0°C by rele...
A: The volume of ethylene glycol is 5468.43 mL
Q: he copper(I) concentration in an aqueous solution is to be measured. For the cathode alf-cell, a sil...
A:
Q: The given reaction occurs in a cell with H2(g) pressure of 1.0 atm and [CI]=1.0M at 25°C. H2 (g) + 2...
A:
Q: Questions: 1) Write a balanced chemical reaction between the KHP and the NaH and between sodium hydr...
A:
Q: . At 30.0 °C, pure benzene has a vapor pressure of 121.8 mmHg. Dissolving 15.0 g of a non- volatile ...
A:
Q: A biochemist prepares two stocks of sodium phosphate buffer by titrating phosphoric acid with sodium...
A: pH = -log(H+) For pH = 2.15 solution initial [H+] = 7.08 x 10^-3 M after diluted with 30 ml of pH 7....
Q: At 20°C, protein-Z binds DNA with a free energy of -7 kJ/mole. Protein Z has an unfolding free energ...
A:
Q: 2. Molecule #2 a)What Group? (Carb, Lipid, Protein, or Nucleic Acid). b)Within the group, how would ...
A: Carbohydrates: Carbohydrates are macromolecules with the general molecular formula CnH2nOn. The mo...
Q: 375 370 365 360 355 350 345 340 0.1 0.2 03 04 05 0.6 0.7 0.8 0.9 A A Mole fraction B B What is the e...
A: Given diagram is : What is the estimated boiling point of the pure component B = ? Options are : a...
Q: Write CS if the statement describes crystalline solids and AS if it describes amorphous solids. 1. ...
A: The following are the characteristics of crystalline solids, Have a uniform structure. Have sharp a...
Q: Question 17 II в D Which part in the figure disperses light into its component wavelengths and selec...
A: The given figure is of monochromatic spectrophotometer. Its components are A - Light source B - Mo...
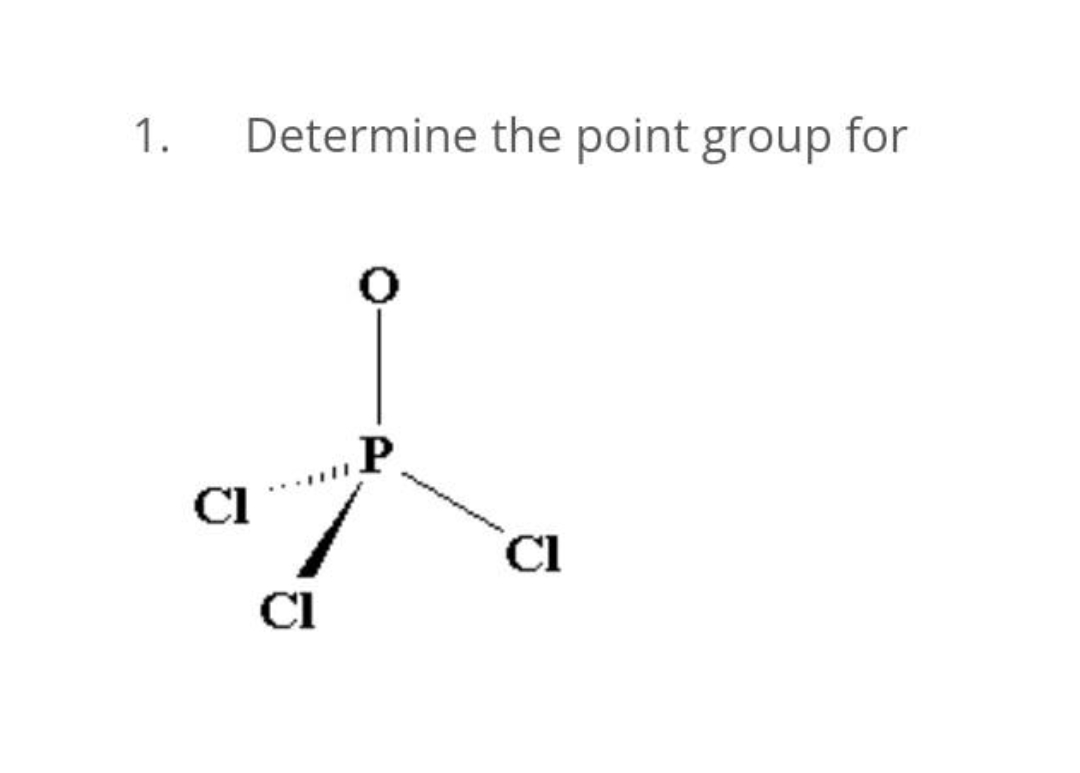
Step by step
Solved in 2 steps with 1 images

- Structural isomers can have very different point groups. There are three isomers of dichlorobenzene. Identify the point groups of the three isomers.What are the number of classes and the order of the following point groups? a C2v b D2h c D6h d S4 e Cs.Show that any irreducible representation of these point groups is normalized. a C2 b D2d c Oh d C4h e C6v
- Show that any two of the irreducible representations of the following point groups are orthogonal to each other. a C2 b C2v c D2h d Oh e TdFigure 13.27 shows the structure of the molecule porphine. Figure 13.27 The structure of porphine. Determine the symmetry elements present in the molecule, and its point group. Does the point group change if an Fe2+ ion is substituted for the two hydrogen atoms in the center of the porphine ring?In your own words, explain why an object that has more symmetry elements is said to have higher symmetry than an object with fewer symmetry elements.
- Why is it unnecessary to consider whether an irreducible representation from C4h is orthogonal to an irreducible representation of D6h?Determine the point group of the following molecules. a cis1,2 Dichloroethylene b trans1,2 Dichloroethylene c Toluene, C6H5CH3 d 1,3-Cyclohexadiene.Identify the symmetry elements present in the following objects. a A ream of blank paper, no holes. b A ream of blank three-holed paper. c A round pencil, unsharpened, with cylindrical eraser. d A round pencil, sharpened, with cylindrical eraser.
- Identify the symmetry elements present in the following objects. a The Eiffel Tower. You may have to look up a picture of it if you dont remember its shape b Any book ignore the printing. c An octagonal wood block. d A jack from the set of jacks pictured here: Note that some of the points end differently.Identify all the symmetry elements present in the tetrahedron, the cube, and the octahedron.a In the Td point group, an S41 improper rotation is equivalent to what other improper rotation? b In the D6h point group, the symmetry operation labeled C21 is equivalent to what other symmetry operation?

