Q: The oxidation number of N in NO2 is eTextbook and Media Save for Later
A: Answer Oxidation number of N in NO2- Oxidation No. Of O atom = -2
Q: Two carts mounted on an air track are moving toward one another. Cart 1 has a speed of 1.1 m/s and a...
A: Given, mass of cart 1 =0.45 kg speed of cart 1 = 1.1 m/s mass of cart 2 = 0.75 kg
Q: please explain and give complete answer
A: As part a is already solved I am solving for part b ., Part b :- Concentration of acetic acid = 7.44...
Q: Draw the reactants that would react to form this cyclohexene derivative in a Diels-Alder reaction. I...
A: In presence of light a Diene and a dienopile react to form a product in Diels-Alder reaction. In gen...
Q: For a one component system, when three phases are in equilibrium, then system becomes. O condensed s...
A: The correct answer about phase equilibrium is given below
Q: Consider two beams of light: Beam A and Beam B. The energy of Beam A is less than the energy of Beam...
A: Answer is :
Q: When solid potassium chlorate is heated, it decomposes to form solid potassium chloride and oxygen g...
A: According to the Law of conservation of mass " all atoms of different elements must be equal on both...
Q: How many grams of silver carbonate are present after the reaction is complete? ΑΣφ m
A:
Q: the mass of
A:
Q: Part A An iron ore sample contains Fe2O3 together with other substances. Reaction of the ore with CÓ...
A: Given, α Fe2O3 (s) + β CO (g) ϒ Fe (s) + δ CO2 (g) Weight of Fe2O3 = 0.310 ...
Q: Which statement is true concerning an oxidation-reduction reaction? O The reactant which is reduced ...
A: oxidation-reduction reaction means a reaction in which both oxidation and reduction takes place simu...
Q: 2. What is the mass of KHP that you will weigh? a. The instructions read "Weigh out accurately 700 t...
A: answer :
Q: For the pyrolysis reaction of propane proceeding at 625 Ky atmospheric pressure C2H2(g) →C2HA(g) + C...
A: Given steps :
Q: How do structures of these IUPAC names look like? 2-Propoxy-1-(2-propoxyethoxy)propane Please g...
A:
Q: How many liters of 0.750 M lead (II) nitrate are necessary to react completely with 1.00 L of 2.25 M...
A: Pb(NO3)2 (aq) + 2 NaCl (aq) → PbCl2 (s) + 2 NaNO3 (aq) Find Moles of NaCl: Using Formula : Mol...
Q: 3) Arsenic acid has 3 pK,'s: 2.19, 6.94, and 11.51. Determine the concentrations of all the species ...
A:
Q: Arrange the following steps in performing a general procedure. I. work on the data II. Carry out the...
A: we are required to outline the steps to perform a general procedure
Q: Information for Parts B, C, and D A compound is 80.0% carbon and 20.0% hydrogen by mass. Assume a 10...
A: Given, Mass of sample of the compound = 100 g Percentage of of each element in the sample of compoun...
Q: ny electrons
A:
Q: how much heat will be given off when 18.5g of C2H4 is burned in oxygen form CO2(g) and H2O(g) for th...
A: The combustion enthalpy of C2H2 of 18.5 g can be calculate with the help given stoichiometric equati...
Q: The water will start to "crawl" up the towel by capillary action. You will be able to see a moving! ...
A: Capilarry action is a process in which a liquid floe from a narrow space without any resistance or e...
Q: 1. How many MOLES of boron trichloride are present in 4.21 grams of this compound ? moles. 2. How ma...
A:
Q: molarity
A:
Q: Which of the following reactions is correctly balanced? A) CO + O2 → CO2 B) N2 + H → 2NH3 C) 2H2O...
A: Given :- A) CO + O2 → CO2 B) N2 + H → 2NH3 C) 2H2O + C → CO + 2H2 D) Zn + 2HCl → H2 + ZnCl2 To ide...
Q: Identify which conversion factor should be used in each of the following conversions using the dropd...
A:
Q: Question states. Select all forms that would definitely produce the photoelectric effect from a piec...
A: When a photon of electromagnetic radiation with sufficient energy falls on the surface of the metal,...
Q: rue or False 1. Sample preparation of infrared spectroscopy uses KBr as the blank because Kbr has 10...
A: Ans.1. Sample preparation of infrared spectroscopy uses KBr as the blank because KBr has 100% transm...
Q: How many electrons in an atom could have these sets of quantum numbers? n = 3 18 electrons n = 5, € ...
A:
Q: Consider the following exothermic reaction: C2H4 (9) + Cl2 (9) = C2H¾C12 (g) Part A If you were a ch...
A:
Q: What happens to ice when sugar and ice are added, respectively?
A: Here we will apply the colligative properties of solute in a solvent.
Q: The equilibrium constant, Ka, for the following reaction is 1.29x10-2 at 600 K. coCI2(g) co(g) + Cl2...
A: The balanced equilibrium reaction given is COCl2 (g) ⇌ CO (g) + Cl2 (g) The equilibrium constant K...
Q: Unknown compound D has a melting point of 102-3C. Carbon/hydrogen analysis of compound D showed 30.4...
A: Given: Compound I contains 30.4% Carbon nad 2.1% H Conclusion: For every 30.4g of C, moles of C =...
Q: Which one of the following is not a redox reaction? O 2H2O(g) O Zn(s) + 2MNO2(s) – O PBO2(s) + Pb(s)...
A:
Q: What is the mole fraction, X, of solute and the molality, m (or b), for an aqueous solution that is ...
A:
Q: AS° for the reaction shown is equal to -667.8 (J/mol-K). Use the information given in the table to d...
A: Given,
Q: ne molarity of
A:
Q: Suppose a 3.00 L scuba tank is filled with about 29.7 moles of air at the ocean surface. If the tank...
A:
Q: A student attempts to identify six solutions that contained either iron(II) bromide, potassium bromi...
A:
Q: Atomic number
A: The atomic number is generally use to uniquely identifies a chemical element.
Q: What is the molar mass of ibuprofen, C13H18O2? A) 206.3 g/mol B) 289.4 g/mol C) 29.0 g/mol D) 37...
A: Molar mass of C = 12.01 g/mol Molar mass of H = 1.008 g/mol Molar mass of O = 16.00 g/mol
Q: Object A has a charge of 3.6 × 10 °C and object B has a charge of 4.7 × 10 °C. What is the electric ...
A:
Q: buffer
A: Here 0.350 M of HCN is given and pH is 9.25 Also Buffer solution is a solution which can resist the ...
Q: Problem 20 - IR spectrum Problem 26 100- C;H10,Br MW = 238 80- 60- - CDCI; 40- - CDCI; 20- 3500 3000...
A:
Q: Under certain conditions the rate of this reaction is zero order in hydrogen iodide with a rate cons...
A: A zero order reaction is the independent of the reactant concentration. And formula used [A]t = - k...
Q: 50.0 g of copper (II) oxide reacts with an excess of ammonia gas in the following unbalanced reactio...
A: Given :- CuO + NH3 → N2 + H2O + Cu (unbalanced) Amount of CuO = 50.0 g To calculate :- Amount of...
Q: A sample of water with a mass of 23.31 g and an initial temperature of 367.67 K loses 8615 Joules....
A:
Q: Draw the lowest energy conformation for the compound shown below. CH 1. Choose a chair from the Temp...
A: Conformational isomers: The conformational isomers are formed by the rotation of a carbon-carbon sin...
Q: Part 3
A: For the reaction of CaCO3 with 6M HCl and with 6M CH3COOH., CaCO3 + 2HCl ---> CaCl2 + H2O + CO2 C...
Q: What coefficients (in order from left to right) are needed to balance the following chemical equatio...
A: What coefficients (in order from left to right) are needed to balance the following chemical equatio...
Q: Ethanol is an organic polar solvent with many commercial uses. A sample of this compound(which conta...
A: Given, Ethanol is an organic polar solvent with many commercial uses. A sample of this compound(whic...
Help me with inorganic chemisyey
Step by step
Solved in 2 steps with 2 images

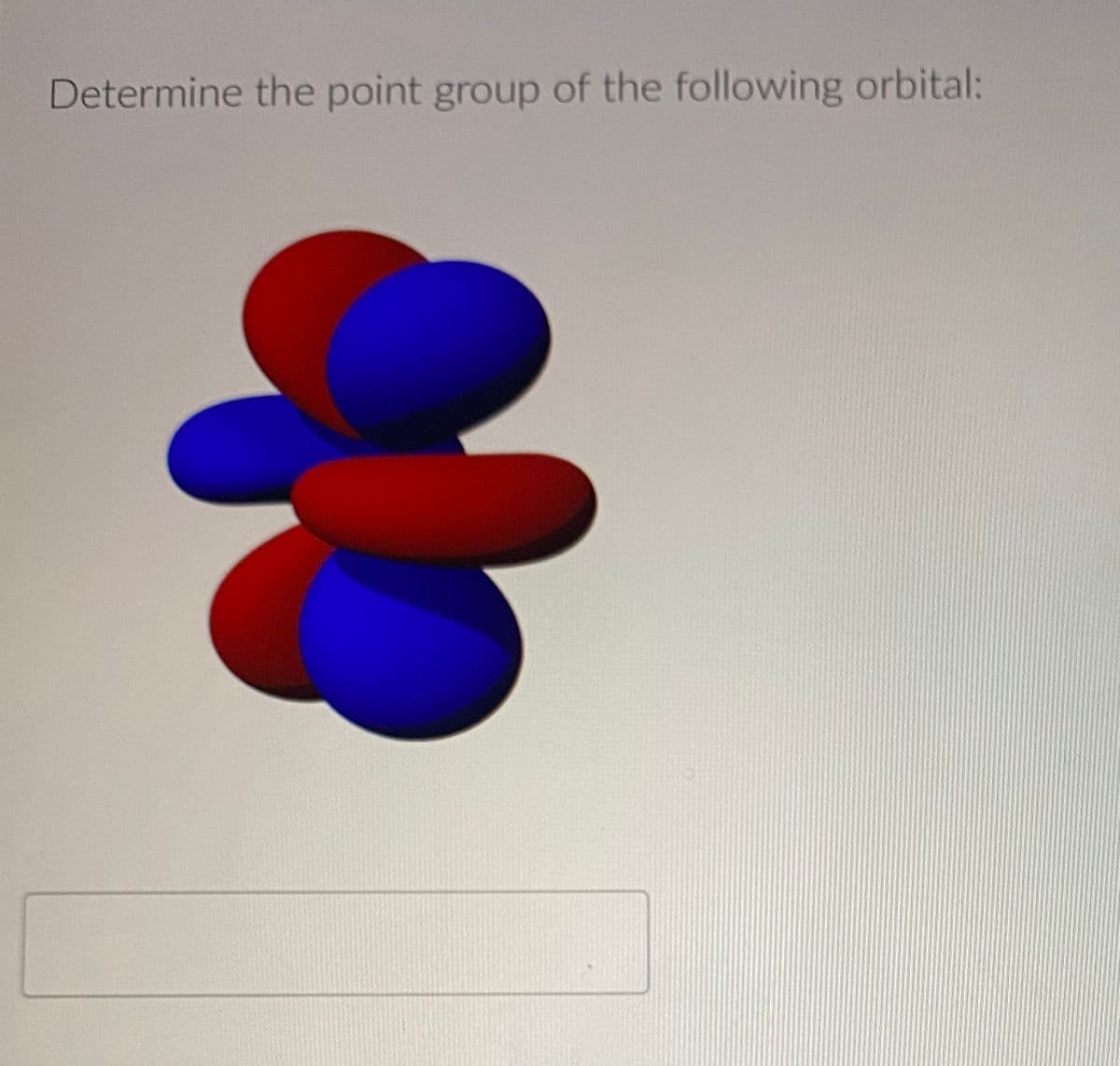
- Show that any two of the irreducible representations of the following point groups are orthogonal to each other. a C2 b C2v c D2h d Oh e TdWhat are the number of classes and the order of the following point groups? a C2v b D2h c D6h d S4 e Cs.Show that any irreducible representation of these point groups is normalized. a C2 b D2d c Oh d C4h e C6v
- Determine the point group of the following molecules. a cis1,2 Dichloroethylene b trans1,2 Dichloroethylene c Toluene, C6H5CH3 d 1,3-Cyclohexadiene.Determine the point groups of the following molecules. a Fe(CO)5, which has a trigonal bipyramidal structure, b CO32, which has three resonance structures that contribute to its overall shape, c The perfectly staggered conformation of ethane, d The perfectly eclipsed conformer of ethane.Why is it unnecessary to consider whether an irreducible representation from C4h is orthogonal to an irreducible representation of D6h?
- Structural isomers can have very different point groups. Determine the point groups of 1,4cyclohexadiene and 1,3cyclohexadiene, which both have the molecular formula C6H8.Use molecular orbital arguments to decide whether or not the difluoride dianion, F22, should exist as a stable ion.Construct the symmetry-adapted linear combination molecular orbitals for hydrogen sulfide, H2S.
- Figure 13.27 shows the structure of the molecule porphine. Figure 13.27 The structure of porphine. Determine the symmetry elements present in the molecule, and its point group. Does the point group change if an Fe2+ ion is substituted for the two hydrogen atoms in the center of the porphine ring?Identify the symmetry elements present in the following objects. a The Eiffel Tower. You may have to look up a picture of it if you dont remember its shape b Any book ignore the printing. c An octagonal wood block. d A jack from the set of jacks pictured here: Note that some of the points end differently.Determine the symmetry species of the D3h point group for the sp2 hybrid orbitals, assuming that the C3 axis is coincident with the z-axis and that one of the orbitals lies along the positive x-axis. See Example 13.16.

