Q: 6. Which one of the following solvents is not a good choice when using CHMgBr? CH,OH Et,0 THF 6.…
A: Grignard reagents (RMgX) highly react with proton. So, protic solvents are not suitable for them.
Q: 4) 250 ML Solution of 25 ppm of Na', from sodium suifate CNasSo4)
A: f) Given that - Volume of sodium sulfate Solution = 250 mL Parts Per Million concentration = 25…
Q: S+ Ni(OH)2+ 20H"s2 + Ni02+ 2H20 In the above reaction, the oxidation state of nickel changes from 8…
A:
Q: Status. TNOL If an electron transition involves 2.81 x 10 19 J of energy, what is the wavelength (in…
A:
Q: a) Briefly describe what chemical reaction occurs when an acid chloride reacts with an amine. b) In…
A: In this question, we have to explain the chemical reaction occurs when an acid chloride reacts with…
Q: In the reaction H2O2 (aq) --> H2O (l) + 1/2 O2 (g) the initial concentration of H2O2 is 0.2546 M,…
A:
Q: How much energy (in kJ) is released when 71.0 g of oxygen difluoride decomposes? Answer: kJ (enter a…
A:
Q: 1. Give monoborylated products for the following reactions. Include any isomers that you would…
A: [Ir(OMe)cod]2 is a CH borylating agent
Q: MASS SPECTRUM 100 - Relative Intensities: 80 m/z 121- 100% m/z 194- 21% 60 m/z 195- 3% 40 20 0.0 0.0…
A: Please find the below attachment. If you do not want to rate please let it be. It will effect my…
Q: Classify the following fatty acids as saturated, monounsaturated, or polyunsaturated fatty acids.…
A: Given, The saturated, mono unsaturated and poly unsaturated fatty acids are:
Q: What volume in liters of fluorine gas is needed to form 261 L of sulfur hexafluoride gas if the…
A:
Q: 2. Which of the following are TRUE about the formation of light elements lighter than iron in the…
A:
Q: One Erlenmeyer contains 50 mL of 0.1 M Na2CO3 solution, the other Erlenmeyer contains 50 ml of 0.1 M…
A: Here we have to determine the volume of HCl solution of 0.1M needed to react with 50 ml of 0f 0.1 M…
Q: Consider that the determination of potassium iodide (MM-166.00 g/mol) from a food supplement was…
A: Given the mass of the sample containing potassium iodide(KI) taken for analysis = 0.5240 g The…
Q: Calculate the problems below. Write your solution and box your final answer.
A: 1)The reaction takes place as: Al(NO3)3 ----->Al3+ + 3NO3- Now here the mole of Aluminium…
Q: A chem V 1gineer is studying the two reactions shown in the table below. In each case, he fills a…
A:
Q: 2Br" + F2 Br2 + 2F In the above reaction, the oxidation state of fluorine changes from 9 to Mow many…
A: 2Br- + F2 ----> Br2 + 2F-
Q: Help only needed on D. Thank
A: We know epoxide ring due to ring strain is unstable and undergoes ring opening reaction even in weak…
Q: H. -Br thylaziridine thylaztidene ethylazete
A: When NH group is a part of cyclic ring it is called as aziridine Now on position 3 bromo group is…
Q: Propose an efficient synthesis for the following transformation: Br - The transformation above can…
A: Lindlar's catalyst used to reduce alkyne into cis Alkene... Alkene with peroxyacid gives epoxide…
Q: QI List therc examples on the importance of analytical chemistry in environmental seienee one of…
A: 1. Answer - Analytical chemistry - Analytical chemistry is the science of obtaining, processing, and…
Q: What quantity in moles of H, are required to give off 2301 kJ of heat in the following reaction? N,…
A: We have to calculate the moles of hydrogen gas required.
Q: What is the key for getting no leftovers(leftover molecules)?
A:
Q: Calculate the concentrations of all ions after 50.0 mL of a 0.100 M AgNO3 solution is mixed with…
A: Given : Concentration of AgNO3 = 0.100 M Volume of AgNO3 solution = 50 ml Concentration of Na2SO4…
Q: Ca(OH)2) = 4.155. What amount of Ca2+-ions (in [mol] and [g]) would be present in 100mL of a…
A: a) pKL(Ca(OH)2) = 4.155 pKL(Ca(OH)2) = -log(K(Ca(OH)2)) 4.155 = -log(K(Ca(OH)2)) K(Ca(OH)2 =…
Q: The specific heat of liquid mercury is 0.140 J g C-1. How many joules of heat are needed to raise…
A: Given, Specific heat capacity of liquid Mercury (Cp) = 0.140 J/g.°C Mass of Mercury (m) = 16.94 g…
Q: 6.43 Draw the products of each reaction. [1 (CH2),CCI, AICI, а. [2] KMNO4 b. [1] Br2, hv [2]…
A: The product of the above organic transformation is given below
Q: H3, H2O, HF CH3OH, CH3NH2, CH3CH3 HC=CCH,CH, CH,CH=CHCH, CH;CH2CH2CH3 HCI, H2O, H2S CH3CH2CH3,…
A: 4) Acidic strength is depends upon ease of tendency to H+ donate. A) NH3 , H2O , HF b) CH3CH3 ,…
Q: For each of the salts on the left, match the salts on the right that can be compared directly, using…
A: Ksp or solubility product of a sparingly soluble salt is the product of concentrations of its ions…
Q: calculate the concentration of free Zn2+ in 0.020M Zn(EDTA)2 at pH 10.00
A: The solution of the question is given below:
Q: Homework Exercise Propose a synthesis for each of the following molecules from malonic ester. (a)…
A:
Q: Question 2 Only one of the statements below is correct; which one? O Two solutions are hypotonic…
A: In this question, we have to choose the statement which is given correct. Isotonic solution:…
Q: Part c Your answer is incorrect. How many grams of NaCHO2 would have to be ad m(NACHO2) =
A:
Q: A neon gas at 660 mm Hg has a volume of 12.0 L and a temperature of 44ºC. Find the new volume of the…
A:
Q: 15.0 L of an ideal gas at 298 K and 3.36 atm are heated to 383 K with a new pressure of 5.45 atm.…
A: Given data of the following, V1=15.0 L, T1=298 K, P1=3.36 atm V2= ?, T2=383 K, P2=5.45 atm
Q: An argon laser emits photons at a wavelength of 610 nm. c = 3x108 m/s h = 6.6x1034 J/s 1 nm = 10-9 m
A:
Q: What products are obtained when (R)-2-bromobutane reacts with CH 30 /CH 30H under conditions for…
A:
Q: d. After subtracting 2 electrons per is e. The number of electrons needed atom in the molecule is f.…
A: In NH₄⁺ ion, N atom forms 4 N-H bonds and thus it has 8 electrons in its outermost shell.…
Q: Hello can this problem please be simplified, I would like to make sure I am getting each step down…
A: P1 = 193 psi V1 = 39 mL T1 = 331 K P2 = 795 psi V2 = ? T2 = 675 K
Q: following food groups is NOT a source of carbohydrate? A. Breads and pasta B. Fish and poultry C.…
A: Note : As you have uploaded multiple questions, so I'll answer first three questions. Please upload…
Q: 3. Circle the compounds that are soluble in water: NaNO, (NH),SO, CACO, 4. KCI Ag,S LiBr РЫ, PBCI,…
A: Compounds that are soluble in water is given below
Q: Make a retrosynthetic analysis to design a synthesis plan. It should be ay least 2 steps synthesis
A:
Q: N. NH O imidazo[2,1,b]oxazole O imidazo[1,2,b]oxazole oxazolo[2,1,b]imidazole
A: A question based on nomenclature that is to be accomplished.
Q: write introduction Experiment Acid-Base Titration Using a pH Meter and Phenolphthalein Strong acid…
A: The purpose of strong acid vs strong base titration is to determine the concentration of acidic…
Q: 1. NaOCH2CH3, CH3CH2OH 2. Acidic workup +
A: Ester which has alpha hydrogen present is form nucleophile when react with base.then it give…
Q: Determine the concentration (M) of H3O+ in a 0.589 M solution of a triprotic weak acid, H3A, given…
A: Given, the concentration (M) of H3O+ in a 0.589 M solution of a triprotic weak acid, H3A is:
Q: How much heat is liberated (in kJ) from 2.57E2 g of silver when it cools from 8.2E1 °C to 2.67E1 °C?…
A: We have to predict the heat released.
Q: Calculate the ∆S 0 for each given reaction. 1. 2H2 (g) + O2 (g) ---> 2H2O (g) 2. Ca3(PO4)2 (s)…
A: 2H2 (g) + O2 (g) ---> 2H2O (g) Ca3(PO4)2 (s) ---> 3Ca+2 (aq) + 2PO43- (aq)
Q: 4. Predict the products of the following reactions., 1. DIBAL-H 2. Н,о 1. PhMgBr (excess 2. Но 1.…
A: “Since you have posted a question with multiple sub-parts, we will solve the first three subparts…
Q: Write the complete mechanism (including intermediates) for the reaction of trans-stilbene with…
A:
Step by step
Solved in 5 steps with 5 images

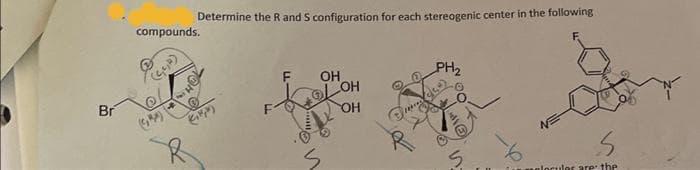
- For the following molecules: a. How many stereogenic centers are present? b. How many stereoisomers are possible for these molecules?Label the stereogenic center in attached compound as R or S.What are the number of stereogenic centers in each and what are the maximum number of stereoisomers in each case? Explain please.
- Considering all stereogenic centers, what is the maximum number ofstereoisomers possible for discodermolide?Draw all stereoisomers and indicate which ones if any, are meso compounds...Determine the configuration of each stereogenic center in the compound below. Highlight the carbon in red if the configuration is R, and in blue if the configuration is S.
- Convert the following chair conformation of the sugar beta-D-galactose into a structure that shows all via and trans relationships.If the two stereogenic centers of a compound are R,S in configuration, what are the R,S assignments for its enantiomer and two diastereomers?the Leu side chain has 2 rotatable bonds that can each each adopt the g+, g- and t conformation. how many possible rotamers does the Leu side chain have?

