Chapter21: Potentiometry
Section: Chapter Questions
Problem 21.11QAP
Related questions
Question
Did the concentration of the reactants affect the amount of electricity produced?
Transcribed Image Text:Electroch
D Lespd
View
Section
Tools
Q Click to find co
ne
Insert
Page Layout
References
Review
O Aa-
X-Al 9
AaBbCcl AaBI AaBbCcD AaBbC
A
T三▼
Normal
Heading 1 Heading 2 Heading 3
10
12
14
16
18
20
22
24
26
28
30
32
34
36
40
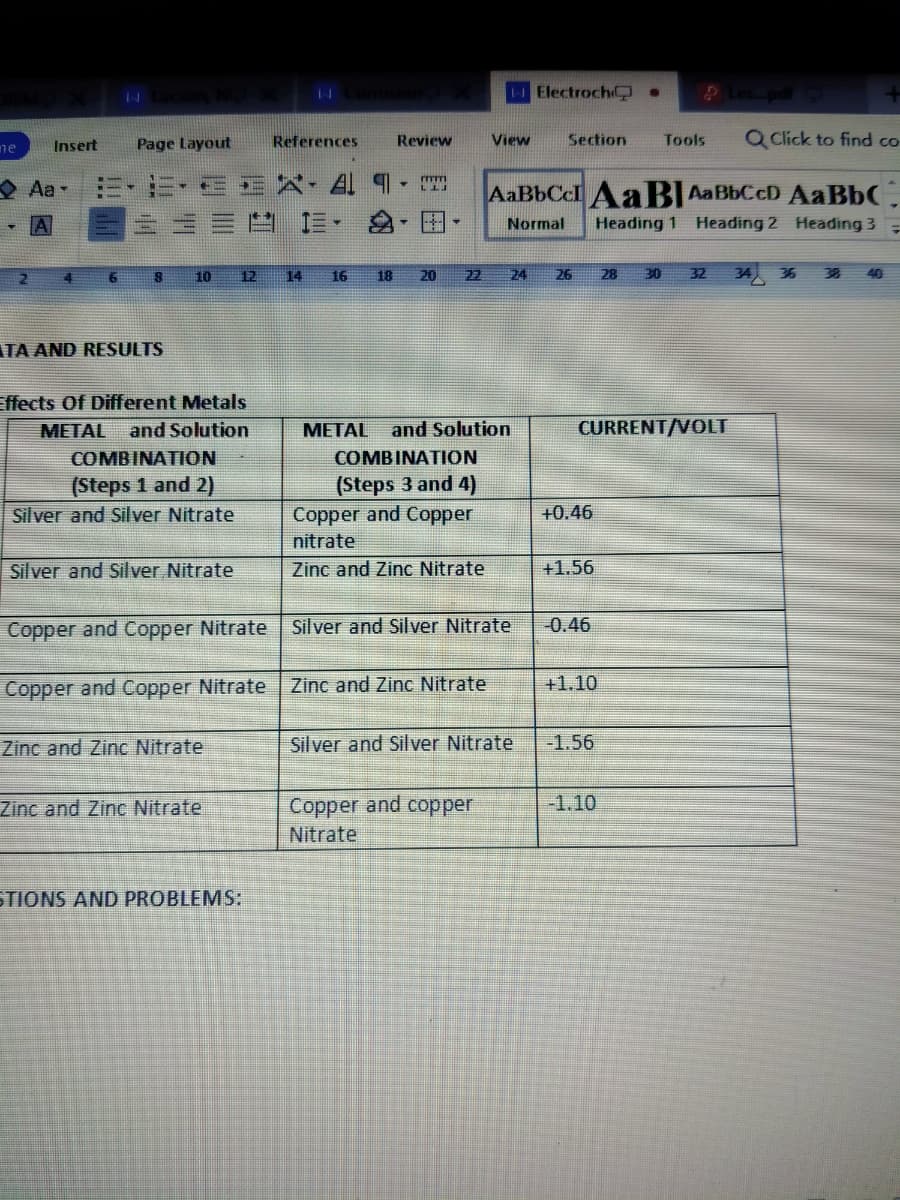
TA AND RESULTS
Effects Of Different Metals
METAL and Solution
МЕТAL
and Solution
CURRENT/VOLT
COMBINATION
COMBINATION
(Steps 1 and 2)
Silver and Silver Nitrate
(Steps 3 and 4)
Copper and Copper
+0.46
nitrate
Silver and Silver Nitrate
Zinc and Zinc Nitrate
+1.56
Copper and Copper Nitrate
Silver and Silver Nitrate
-0.46
Copper and Copper Nitrate
Zinc and Zinc Nitrate
+1.10
Zinc and Zinc Nitrate
Silver and Silver Nitrate
-1.56
Zinc and Zinc Nitrate
Copper and copper
-1.10
Nitrate
STIONS AND PROBLEMS:
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Recommended textbooks for you


Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax


Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax