Q: Can oxalix acid and oxalate form a buffer? If not, what would be the species of a buffer if it would…
A: Oxalic acid, H2C2O4(aq) is a diprotic weak acid with pKa1 = 1.27 and pKa2 = 4.28 The dissociation…
Q: 1. Name the following structures according to IUPAC rules: a. b. C. d. Name: SH Name: CO₂H Name:…
A:
Q: Gaseous ethane CH3CH3 reacts with gaseous oxygen gas O2 to produce gaseous carbon dioxide…
A:
Q: If you have 34.7 grams of chromium that react with excess oxygen, how much chromium(III) oxide will…
A:
Q: 2 H₂C Br Convert the following Newman projections into their corresponding line structures. CH₂CH₂…
A: Confirmation isomers are stereoisomers which produced by rotation/ twisting sigma bonds. These…
Q: What is the effect of pH on the binding of oxygen to hemoglobin (the Bohr Effect)? What happens to…
A:
Q: Iron reacts with oxygen gas to form a mixture of FeO and Fe₂O₃ when the supply of oxygen is limited.…
A:
Q: On the box of a cake mix, the recipe says that it makes 24 cupcakes. When the baker makes the cake…
A:
Q: 2. The concentration of Mg2+ in seawater is 0.052 M. At what pH will 99% of the Mg2+ b precipitated…
A: Given that The concentration of Mg+2 in seawater is 0.052 M. Ksp for Mg(OH)2 = 8.9X10-12
Q: An apparatus was set up like in this experiment. A student measured 13.0 cm of water in the tube…
A: Given data: Atmospheric pressure = 30.0 in Hg Height of water above the water surface in flask =…
Q: Using the table of standard formation enthalpies that you'll find under the ALEKS Data tab,…
A:
Q: 1) The compounds below are considered: Br x-x and CH3 stereoisomers O constitutional isomers O…
A: when two isomers have same molecular formula but bond connectivity are different then isomers are…
Q: -Chloroaniline eneoic Acid -phenyl THER 4-Chlorooniline Hydrochloride) Excess H₂0 CI 4₂0 Trace Ether…
A: Solution: Ternary organic mixture can be separated by simple acid-base principle. But the…
Q: A buffer contains 0.035 mol of boric acid (pKa = 9.15) and 0.015 mol of sodium borate per liter.…
A: Here we have to determine the pH of buffer solution containing 0.035 mol of boric acid pka= 9.15 and…
Q: 18. Predict this element based on the image:
A: For a given element : Atomic number = number of electron = number of proton Atomic mass = number of…
Q: Calculate the pH of a 0.123 M solution of ethylenediamine (H₂NCH₂CH₂NH₂). The pKą values for the…
A: Given data :
Q: CHO H₂On Hot к-тонно CHO Choon H "H CH₂CH B. This noncyclic structure is correctly identified as…
A:
Q: One day in lab, while taking apart a complicated distillation apparatus, your friend Teresa (an…
A: Balanced equation: It can be defined as the chemical equation where the number of each atom on the…
Q: g. h. i. OH HBr to HCI Br excess HS:
A: To find out the missing reagent as well as products we have to look out the detail mechanistic…
Q: Electrolytes are substances that Group of answer choices A) form covalent bonds with water. B)…
A: In this question some statements about electrolytes are given We need to identify the correct…
Q: How many grams of phosphorus will be needed to produce 3.25 mol of P4010 ? 4P + 50₂ → P4010
A: Given that - Moles of P4O10 = 3.25 mol Also given following balanced chemical reaction - 4P + 5O2…
Q: When drawing a Lewis structure for a covalent compound, you need to know the total number of valence…
A: we have to calculate the number of valence electrons in Cl2CO
Q: 4. Would the separation of the grape soda dyes work equally well if you reversed the order of IPA…
A:
Q: In the balanced oxidation-reduction reaction (occurring in acidic condition) the coefficients before…
A:
Q: 1. The ionization constant (Kb) of pyridine (C5H5N) is 5.62 x 10–4. If 11.3 grams of sample of…
A:
Q: Gaseous methane CH4 reacts with gaseous oxygen gas O2 to produce gaseous carbon dioxide CO2…
A:
Q: Draw the structure of (E)-2-chloro-2-pentene.
A:
Q: Identify the reaction type.
A: Given : reaction Tip : in decomposition reaction one molecule dissociate into two or more molecules.
Q: gas tank containing an air mixture of helium, nitrogen and oxygen has a total pressure of 45.0 psi.…
A:
Q: Phosphorus pentachloride gas is obtained by the reaction of chlorine gas and solid phosphorus. Write…
A: We have to write the balanced chemical equation of chlorine gas and solid phoshorous
Q: Name the following organic compounds:
A: Given : structure of molecules. Tip : select longest carbon chain for parent chain.
Q: Rank cork, concrete, copper and glass from most heat conductive to least.
A: HEAT CONDUCTION:- Heat conduction is transfer of heat from higher temperatures to low temperature.…
Q: 2) Express the equilibrium constant for the following reaction. 2 CH3C1(g) + Cl2 (g) 2 CH2Cl2 (g) +…
A: At equilibrium, the rate of the forward and the reverse reaction is the same and the concentration…
Q: Which of the following is the weakest intermolecular bond? Hydrogen bond Polar covalent bond London…
A: Here we are required to find the weakest intermolecular force
Q: The drug cyclophosphamide is administered for breast cancer at a daily dose of 100 mg/m² for up to…
A: The administered dose is 100 mg/(m2.day) for 14 days Height in ft = 5 ft. 1 in. 1 ft = 12 in and 1…
Q: How many moles of sucrose (342 g/mol) must be added to 552 g of water (18.02 g/mol) to give a…
A: Given - Weight of water =552 gram Molar mass of sucrose =342 g/mol Molar mass of water =18.02g/mol…
Q: Exercise calculate the mean and standard deviation of the following set of analytical data. 15.67,…
A: Mean : It is defined as the average of all the observations. It is expressed as Mean = {Sum of…
Q: Aqueous sulfuric acid H2SO4 reacts with solid sodium hydroxide NaOH to produce aqueous sodium…
A:
Q: The enthalpy of dissolution of potassium iodide (166.0 g/mol) in water is +20.3 kJ/mol. A 6.2 g…
A:
Q: 13. As the mass number of an element's isotopes increases, the number of protons OA. decreases O B.…
A:
Q: How many gauche interactions for this compound?
A: gauche interactions for this compound.
Q: One of 7.74 There are two stereoisomers of 1-tert-butyl-4-chloro-cyclohexane. these isomers reacts…
A: Note - Since the given question is a multiple question, hence I solved first question according to…
Q: energy absorbed 19. A negative enthalpy change is a measure of the amount of A. True B. False the…
A: We have to mention the given statement in the question is true or false.
Q: A decomposition reaction can be symbolized by Group of answer choices A) A + B → C + D. B) A + B →…
A: Combination reaction: In this reaction, two reactants combine to form a single product; hence the…
Q: Calculate the standard enthalpy of reaction (AHºrxn). Use the standard enthalpy of formation values…
A: According to the question we have, The standard enthalpy of the reaction ∆H°rxn = -1932.84 KJ/mol…
Q: Draw a Newman projection as viewed from the angle indicated in the 3D Perspective. H3c CH3 CH3 CH3
A: A question based on introduction to organic chemistry that is to be accomplished.
Q: 7. 8. 9. Mn(ClO3)2 4 HgO Ni+ HCl-
A:
Q: cat. H+ excess CH3OH -H₂O O.
A: Here we are required to draw the curly arrow mechanism for the reaction.
Q: What is the expected bond angle of PBr3? A) 109.5o B) 180o…
A: What is the expected bond angle of PBr3?
Q: Aqueous hydrochloric acid HCl reacts with solid sodium hydroxide NaOH to produce aqueous sodium…
A:
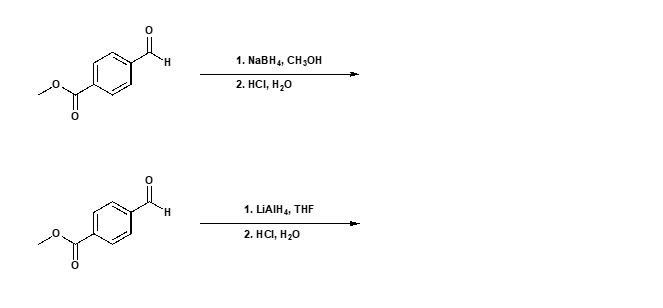
Step by step
Solved in 2 steps with 1 images

- Fumaric acid is quite common in the plant world and is added to many foods due to its acidity and fruity flavor. It has a molar mass of 116.07 g/mol and a formula of C4H4O4. A solution of sodium hydroxide is used to determine the molarity of a fumaric acid acid solution before it is used to prepare some sour gummi worms. C4H4O4(aq) + 2NaOH(aq) --> Na2C4H2O4(aq) +2 H2O(l) Calculate the Molarity of the fumaric acid if 32.08 mL of 0.09973 M NaOH was used to neutralized 75.00 mL of fumaric acid solution. Assume the fumaric acid is acting as a strong electrolyte acid in this reaction. (It is a weak acid, but a very strong weak electrolyte) Fumaric acid is quite common in the plant world and is added to many foods due to its acidity and fruity flavor. It has a molar mass of 116.07 g/mol and a formula of C4H4O4. A solution of sodium hydroxide is used to determine the molarity of a fumaric acid acid solution before it is used to prepare some sour gummi worms.…Potassium cyanide reacts with acids to produce hydrogen cyanide gas via the following reaction: ???(??) + ???(??) → ???(?) + ???(?) If 0.150 g of potassium cyanide reacts with an excess of HCl (i.e., enough HCl to completely react away all of the KCN), how many grams of hydrogen cyanide gas can theoretically be produced?One of the challenges astronauts face is in space travel is the build-up of carbon dioxide in their spacecraft. The average human exhales about 42.0 g of carbon dioxide per hour. One method to remove the excess carbon dioxide from the air is using solid lithium hydroxide: CO2(g) + 2 LiOH(s) → Li2CO3(2) + H2O(l) If a typical cannister of LiOH contains 750.0 g, how many hours will it last for one person before it has to be replaced?
- What volume of 3.00 M NaOH contains 1.75 mol NaOH? __?__ LWhat is net ionic for CuSo4+NH4Cl ? Does CUCl2 Considered Insoluable ?When heated, calcium carbonate decomposes according to the equation: CaCO3(s) ---> CaO(s) + CO2(g) A 2.255g sample of a mixture of CaCO3 and CaO was heated to completely decompose the CaCO3. After heating, the sample weighed 1.860g. What was the percent by weight of CaCO3 in the sample?
- when methane gas ch4 ang chlorine gas Cl2 react, liquid carbon tetrachloride CCl4 and hydrochloric acid HCI are produced.When (NH4 )2SO4 , and BaCl2 are mixed, a precipitate will form. Which ions should act as reactants in the net ionic equation?What percentage of NaCN is carbon? A. 25 % B. 31 % C. 11 % D. 22 %
- Natural waters often contains relatively high levels of calcium ion, Ca2+, and hydrogen carbonate ion (bicarbonate), HCO3, from the leaching of minerals into the water. When such water is used commercially or in the home, heating of the water leads to the formation of solid calcium carbonate, CaCO3, which forms a deposit ('scale') on the interior of boilers, pipes, and other plumbing fixtures. Ca(HCO3)2(aq) --> CaCO3(s) + CO2(g) + H2O(l) If a sample of well water contains 2.0 X 10-3 mg of Ca(HCO3)2 per milliliter, what mass of CaCo3 scale would 1.0ml of this water be capable of depositing?An experiment using the reaction below produces 23.26 grams of chlorine gas (Cl2). What mass of HF is produced by this experiment? Round your answer to the nearest 0.01 and remember to include units and substance! HCl + F2 -- > HF + Cl2The fizz produced when an Alka-Seltzer tablet is dissolvedin water is due to the reaction between sodium bicarbonate(NaHCO3) and citric acid (H3C6H5O72)3 NaHCO3(aq) + H3C6H5O7(aq)-------->3 CO2(g) + 3H2O(l) + Na3C6H5O7(aq) In a certain experiment 1.00 g of sodium bicarbonate and1.00 g of citric acid are allowed to react. (a) Which is the limitingreactant? (b) How many grams of carbon dioxide form?(c) How many grams of the excess reactant remain after thelimiting reactant is completely consumed?

