Q: Unknown compound has a solubility of 0.023 g/mL in water at 20 °C. For the purification by…
A: solubility of the unknown compound = 0.023 g/mL in water at 20 °C. The amount of compound taken for…
Q: 8 If a reaction has a rate law of: Rate = [A]²[B]2 2 What factor will the rate increase if the [A]…
A:
Q: At 35ºC, an equilibrium mixture in a 1.0-L flask contains 0.012 mol of Cl2, 0.010 mol of NO, and…
A: Since,Equilibrium constant or Kc is the ratio of the equilibrium concentrations of product over…
Q: Calculate the pH when 31.0 mL of 0.150 M KOH is mixed with 20.0 mL of 0.300 M HBrO (Ka = 2.5 × 10⁻⁹)
A: KOH + HOBr = KOBr + H2O Moles of HBrO = Molarity x volume in…
Q: When 50.2 g iron(III) oxide reacts with carbon monoxide, 32.5 g iron is produced. What is the…
A: Percent yield of a reaction is calculated using the given formula . % yield =actual yieldtheoretical…
Q: ' Determine the activation energy (kJ) for a reaction using the following information. Report your…
A: To calculate activation energy, we need to use Arrhenius equation.
Q: Exercise 4 Write a Ka (hydrolysis) reaction for the following acids. Use the appropriate type of…
A: Note: As per guidelines solution of question-4 has been made. For the expert solution of other…
Q: For the H₂SO4 and NaOH titration, the endpoint is ideally reached when a green color is observed. If…
A: Given , titration of NaOH and H2SO4
Q: 3. A proposed mechanism for the decomposition of ozone, O3, is as follows: Step 1 03 0₂ Step 2 O3 +…
A: O3 ⇔O2 +O FastO3 +O →2O2 Slowa. Overall reaction 2O3 →3O2 b . O is the…
Q: In a sample consisting of lysine, leucine, and glutamic acid, which will be eluted last from an…
A: Ion exchange chromatography separates proteins based on the net charge of amino acids at the given…
Q: Ions Reagents Formula Color and Nature Cr3+ 3%H2O2 conc H2SO4 ether(vanishing blue test)…
A:
Q: Draw the skeletal structure of a dihalide that could be used to prepare the following alkyne. There…
A: Geminal or vicinal dihalide reacts with a strong base (NaNH2) and gives an elimination reaction to…
Q: Would you expect MgCO3 to be more or less soluble in water or a saturated water solution of SrCl₂?…
A: When equilibrium shift in right direction then solubility of a compound increases that is more…
Q: Calculate the volume in liters of a 36.3 g/dL zinc nitrate solution that contains 379. g of zinc…
A:
Q: The following pH curve is for the titration of 20.0 mL of a 0.10 M solution of a weak monoprotic…
A: Given, pH curve . Volume of monoprotic base =20.0 mL concentration of strong acid = 0.1 M
Q: The vapor pressure of pure benzene (C6H6, 78.1 g/mol) is 0.53 atm at 61 °C. The vapor pressure at 61…
A:
Q: The heat of combustion (AH) for propane (molar mass 44.11 g/mol) is -2222 kJ/mol. The heat generated…
A: Given data The heat of combustion for propane is -2222 kJ/mol.Mass 2.195 g of 575.0 g of water The…
Q: Which extraction solvent, when mixed with an aqueous solution of NaHCO3, would form an upper layer…
A: Extraction is an isolation technique that is used to separate one compound from a mixture.
Q: X Y Z sample before cm 6 -5- 4 3- X Y Z sample after solvent front origin line
A:
Q: Predict whether boron trichloride is more likely to form a solution with water or carbon…
A: We have to tell whether boron trichloride is more likely to form a solution with water or carbon…
Q: A Diels-Alder reaction that shows the stereospecificity of the reaction with respect to the…
A:
Q: The dose of a drug is 50 mg/kg of body weight once daily for 7 consecutive days. For a child…
A: The required dose is 50 mg/(Kg.day) for 7 days Weight of child = 90 lbs 1 lb = 0.453592 => Weight…
Q: What is the pH of a solution made of 0.15M NH3 and 0.23M NH4Cl? where the Kb= 1.8 x 10-5. A. 4.61…
A:
Q: Kindly show all solutions. Applying the correct solution preparation procedure, calculations, and…
A: Given : strengths of solution M1=14.8M, M2=0.296M, V2=500mL M1=molarity of concentrated…
Q: Match the following descriptions or examples with the corresponding term. More than one description…
A: Concepts of titrimetric analysis.
Q: Find the volume (mL) of 0.0588 M KOH needed to reach the equivalence point in the titratio 23.4 mL…
A: At equivalence point Concentration of KOH × Volume KOH=Concentration of HNO2 × Volume HNO20.0588 M ×…
Q: 2 HO ll Br C Br- Br CI- CH3 I -H -H CH3 S-3-bromo-2- methylbutane
A: In last column the configuration which gives correct configuration is .
Q: Compute the repeat unit molecular weight for nylon 6,6
A: Given : structure of repeat unit. Tip : add masses of all atoms
Q: draw bond line ch3ch2ch2(cl)ch3 cis-2,3-dichlorobut-2-ene plz help
A: Bond line representation is the one in which carbon and hydrogen are not shown while the other…
Q: Fill in the missing information: symbol ст atom or ion? check all that apply neutral atom neutral…
A:
Q: What is the equilibrium constant (in atm to three decimal places) of HBr for the following reaction…
A:
Q: 1. a) For each of the following compounds, draw a structural isomer that changes the functional…
A: For the given organic compounds, we have to: 1. Draw a structural isomer with a different functional…
Q: QUESTION 11 Determine the molar solubility of MX (Ksp 8.0x109) in 0.012 M NaCN. The metal ion M*…
A:
Q: The compressibility factor of nitrogen does not exceed 0.9 at 126K and 300 atm True or False? Show…
A:
Q: Many common materials that we ingest, though quite safe in reasonable quantities, become toxic when…
A: Number of glasses of Allura Red can be calculated based on the total milligrams of its required.
Q: Ammonium phosphate NH43PO4 is an important ingredient in many fertilizers. It can be made by…
A: Given, mass of phosphoric acid (H3PO4) reacts = 3.3 g mass of ammonium phosphate [(NH4)3PO4]…
Q: At certain conditions, the average rate of disappearance of O3 gas in the reaction 2O3 --> 3O2…
A:
Q: Correctly complete each statement regarding the standard entropy of the substances indicated. The…
A: The entropy of a compound is related to the equation∆S=∆HT. So, entropy depends on the Temperature…
Q: What is the equilibrium concentration (in M to three decimal places) of HBr for the following…
A:
Q: 4. For the problem below please provide a high yielding synthetic route to the product from the…
A: the reaction procedure is given below .
Q: V7
A:
Q: A 22.6 mL sample of 0.221 M ethylamine, C₂H5NH2, is titrated with 0.207 M hydrobromic acid. After…
A:
Q: The pH of a solution of HBr is 2.88 at 25°C. What is the H⁺ concentration in this solution?
A:
Q: Provide the correct systematic name for the compound shown here. N,N- 2- di 4- 3- N- 5- tetra tri…
A:
Q: 1. Balance the following Redox Reactions A.MnO2 →MnO4+MnO₂ B. Crl3+Cl₂ CrO²+1O+Cl…
A: The redox reactions are balanced by using half-reactions method or oxidation number method. In…
Q: The heats of reaction for the following combustion reactions were determined experimentally:…
A:
Q: Draw a Diels-Alder reaction that shows the stereospecificity of the reaction with respect to both…
A: During Diels-Alder reactions, unsaturated substituents such as -COR, -COOR etc of dienophile…
Q: A 31.80 g sample of a substance is initially at 20.0 °C. After absorbing 1717 J of heat, the…
A:
Q: Which of the following properties determines the viscosity of a liquid molar mass intermolecular…
A: Concepts of viscosity of liquid.
Q: Draw an electrophilic addition reaction of a conjugated diene that is kinetically-controlled
A: Given : kinetically controlled product
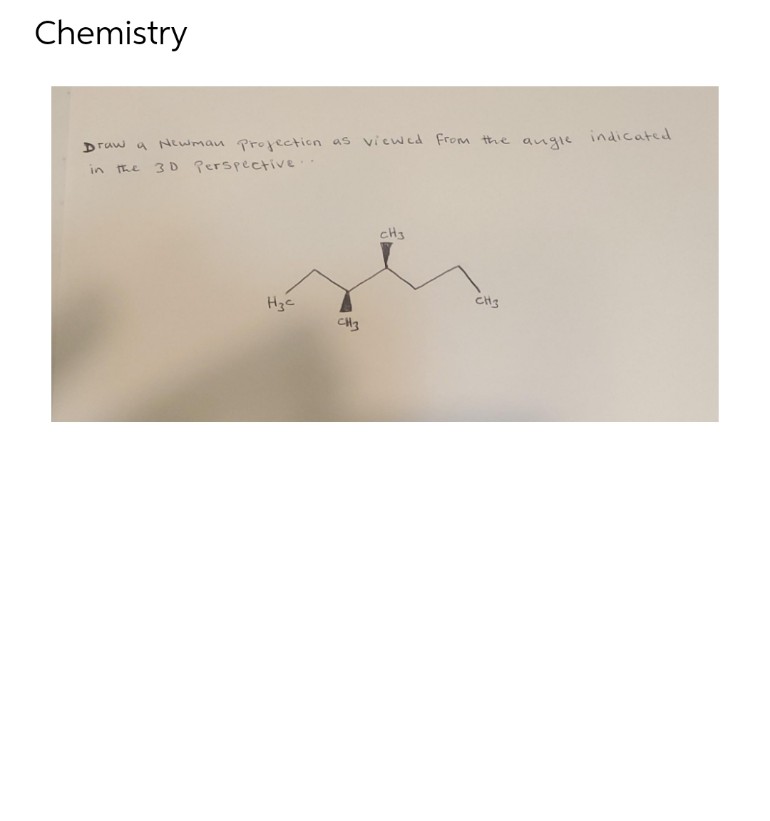
Step by step
Solved in 2 steps with 1 images

- (a) Draw the molecular orbital picture for propa-1,2-diene, H2C=C=CH2. Hint: The three-dimensional geometry is shownin the chapter. (b) Draw the MO energy diagram for propa-1,2-diene. What is the HOMO? What is the LUMO?Please refer to the Newman projections shown below to when answering questions (a) – (f)Please help wiith the fourth one (C3H8O)
- 1. Draw structures of the most stable Newman projection for the image below2. Draw the five constitutional isomers having the molecular formula C4H6select the most stable isobutyl chloride newman projectionConsider the molecule 1-bromo-2-methylbutane. C3 and C4 should be drawn as Et as in theexample. This group is called an ethyl group and can be considered a sphere about twice the sizeof a methyl group. Draw the following Newman projections sighting down the C1C2 bond... a. The lowest potential energy conformation. b. The highest potential energy staggered conformation.
- Indicate the relationship between each pair. Choose from: configurational stereoisomers,conformers, constitutional isomers, or different formulas (Each term is used at least twice.)Consider the Newman projection below. a. Draw a full Lewis structure of this molecule with R1=Me,R2=Et , and R3=iPr . b. Given the sizes of these R groups (R3R2R1) , does the Newman projection above show thelowest potential energy conformation of this bond? If not, draw a Newman projectionshowing the lowest P.E. conformation (sighting down this same bond). c. To draw a Newman projection in the lowest P.E. conformation, the following rule of thumbusually applies: Place the largest group on the front carbon anti to the largest group on theback carbon. Is your answer to the previous question consistent with this rule of thumb?I believe the first anwer is d and the second is a mixture of enantiomers

