Organic Chemistry
8th Edition
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Chapter22: Reactions Of Benzene And Its Derivatives
Section22.2: Disubstitution And Polysubstitution
Problem 22.4P
Related questions
Question
9
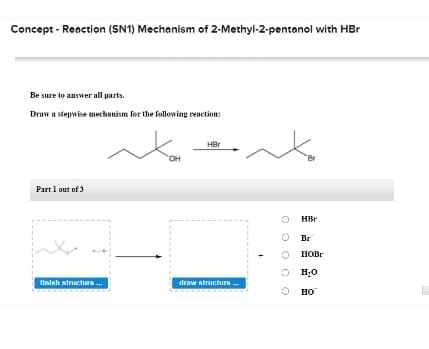
Transcribed Image Text:Concept - Reaction (SN1) Mechanism of 2-Methyl-2-pentanol with HBr
Be sure to answer all parts.
Draw a stepwise mechanism for the following reaction:
HBr
он
Br
Part 1 out of 3
HBr
O Br
HOBR
O H;0
tinish structure .
draw structure
но
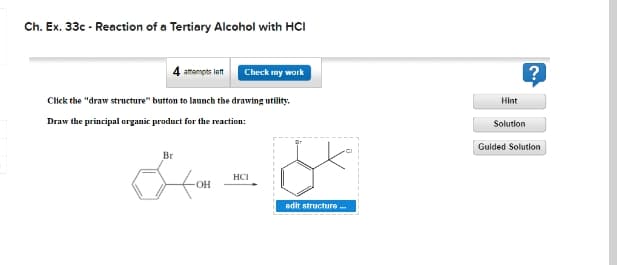
Transcribed Image Text:Ch. Ex. 33c - Reaction of a Tertiary Alcohol with HCI
attompts lett
Check my work
Click the "draw structure" button to launch the drawing utility.
Hint
Draw the principal organic product for the reaction:
Solution
Gulded Solution
Br
HCI
edit structure
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning