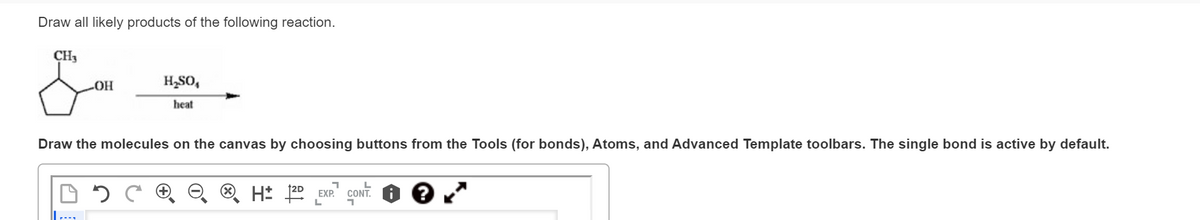
Draw all likely products of the following reaction. CH₂ -OH B H₂SO, heat Draw the molecules on the canvas by choosing buttons from the Tools (for bonds), Atoms, and Advanced Template toolbars. The single bond is active by default. A 8 U+ 12D +
Q: What is the balanced chemical equation for the chemical reaction depicted in the figure? Be sure…
A: Information of question
Q: Consider the two trials shown below and provide structures for the products formed: Trial 1 Trial 2…
A: NaBH4 is a good reducing agent and can reduce aldehyde and ketone .
Q: Rank the following chair structures in order of stability (#1 = most stable, #4 = least stable): Zox…
A: Given are cyclohexane derivatives in chair forms.In chair conformation, most stable conformation is…
Q: Provide the complete IUPAC name for the following structure. p
A: RULES for IUPAC nomenclature; In first step, we identify functional groups present. (Absent…
Q: 400 cm3 of 0.2M h2so4 is mixed with 600 cm3 of 0.1M naoh solution. Is this reaction endothermic or…
A: Endothermic reaction is the one in which heat is absorbed while in an exothermic reaction heat is…
Q: The pOH of an aqueous solution of 0.532 M acetylsalicylic acid (aspirin), ( Ka (HC9H7O4) = 3.0 ×…
A:
Q: What is the hydronium ion concentration in an aqueous hydrobromic acid solution that has a pOH of…
A: Since you posted multiple type question, then according to the guideline, post first solution for…
Q: What are the approximate bond angles between atoms in the follow molecule? :F: --F:
A:
Q: An unknown compound contains only C, H, O. Complete combustion of 5.50 g of this compound produced…
A:
Q: Draw the major product of this reaction. Include stereochemistry if applicable. Ignore byproducts.…
A:
Q: Calcium Chloride solution comes as a 10% solution in a 10 ml vial. How many ml of this solution are…
A: A quantity can be represented using multiple units. By analyzing the relationship between the two…
Q: Draw and upload the expected product of this reaction. Br. H3C X Incorrect -CEN (no answer) H₂SO4,…
A: Cyanide on acidic hydrolysis give Carboxylic acid.
Q: Imagine you react HCI (an acid) with NaOH (a base). It requires 25.0 mL of 0.500 M HCI to neutralize…
A: HCl (acid) reacts with NaOH (base) to form NaCl (salt) and H2O.Reaction is,HCl + NaOH = NaCl +…
Q: At 650 K, the reaction MgCO3(s) ∆ MgO(s) + CO2(g) has K = 0.042. A 10.0 L container at 650 K has 1.0…
A: Given:Reaction at :Equilibrium constant (K) for the reaction at 650 K:K = 0.042Volume =10.0 L Mass…
Q: The pOH of an aqueous solution of 0.532 M caffeine (a weak base with the formula C8 H10 N4O2) is…
A: Given ,Part(A)Part(B)
Q: 1. 1. 2. 3. 4. Provide IUPAC names for the given structures. Compound Br ㅁ OH ㄷ yta 게 Name
A: We have to determine the IUPAC name of the compounds shown below.
Q: Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product…
A: Here, an organic reaction steps are given with all the intermediates and products of the reaction.We…
Q: where did the x 10-6 come from on the 12.8 concentration value when it wasn't in the original…
A: Question :Calculate the extinction coefficient of each peak if the measurements were made for a…
Q: Draw the BEST lewis structure (with formal charges) of the indicated chemical species. (Draw all…
A:
Q: solution A iron (II) nitrate zinc sulfate sodium chloride solution B sodium hydroxide manganese(II)…
A: First, write the balanced reaction between compounds and check the solubility of the product using…
Q: A 24.3 mL sample of a 0.435 M aqueous hypochlorous acid solution is titrated with a 0.348 M aqueous…
A: The volume of the hypochlorous acid = The concentration of the hypochlorous acid = The concentration…
Q: .O-CH3 حمد سینہ H OH ОН
A: Functional group is a group of atoms having distinctive chemical properties responsible for the…
Q: Identify the most stable structure(s). O CH₂-CH-OCH3 O CH₂-CH_OCH3 O O e CH₂-CH=0CH, e CH₂=CH-OCH,
A: If the Lewis structure of a molecule or ion cannot be explained by a single structure then more than…
Q: 2. Draw two resonance structures for the structure shown below. Use curved arrows to indicate…
A: If the Lewis structure of a molecule or ion cannot be explained by a single structure then more than…
Q: each chemical reaction listed in the first column of the table below, predict the sign (positive or…
A:
Q: Below is an image with some separation results. Each of them is erroneous. Match the result with the…
A: The general principle used in TLC separation is the compounds in the mobile phase move over the…
Q: Suggest typical reagents needed to convert an alkene into an aldehyde (e.g. B to C). Your answer may…
A: Alkenes react with ozone to cleave the double bond oxidatively by forming ozonides. The ozonide…
Q: The densities of water, mercury and gasoline is shown below. Water 1.0 g/mL Gasoline 0.701 g/mL…
A: The mass per unit volume of a substance is known as the density of the substance. The density of a…
Q: 1. Aside from the reagent and the positive results, what is the difference between Fehling’s…
A: Fehling's test, Moore's test, and Benedict's test are all chemical tests used to detect the presence…
Q: The start drug is Tolbutamide and the product is Chlorpropamide. Which of the following is…
A: The question appears to be related to the modification of a drug molecule, specifically the…
Q: A student measures the Pb2+ concentration in a saturated aqueous solution of lead hydroxide to be…
A: Solubility product constant is defined as the product of concentration of ions present in the…
Q: What is the major product of the following Diels-Alder cycloaddition? 5. a b e Od Oc CN CN CN d. CN…
A:
Q: Which of the following combinations of buffering systems and molar ratios of compounds would work…
A: A buffer is a solution which resists any change in pH on adding a small amount of acid or base.It is…
Q: solution A sodium sulfide potassium acetate silver nitrate solution B copper(II) chloride ammonium…
A: Some combinations of ionic compounds are not soluble in water. If the mixture of the solutions…
Q: Below is some data for three different gases. Container Gas Formula Molar Mass Volume Temperature…
A: A gas that obeys the ideal gas equation (PV=nRT) at all temperature and pressure values is…
Q: Identify whether each species functions as a Brønsted-Lowry acid or a Brønsted-Lowry base in this…
A:
Q: 4. Two solutions, initially at 24.60°C, are mixed in a coffee cup calorimeter (Ccal = 15.5 J/°C).…
A:
Q: Although ester bonds are present in all of the lipid structures, which lipid structures listed below…
A: Given:a. Glycerophospholipidsb. Cholesterolc. Triacylglycerolsd. Gangliosidese.…
Q: 7. Which compound would produce the following ¹H NMR spectrum? 4 4 11 PPM fork IV
A: The question is based on the concept of organic spectroscopy. We need to analyse the spectral data…
Q: When the ammonium salt CH,CH₂CH, NHBr is treated with NaOH, the products are H₂0, NaBr, and an…
A: Tertiary ammonium bromide react NaOH to give tertiary amine , water and NaBr
Q: For each system listed in the first column of the table below, decide (if possible) whether the…
A: Answer:Entropy is a measure of system's disorderliness or randomness. In more possible microstates…
Q: Which weak acid would be best to use when preparing a buffer solution with a pH of 9.50? an acid…
A: A buffer is a solution which resists any change in pH on adding a small amount of acid or base.It is…
Q: Consider the following data on some weak acids and weak bases: acid name nitrous acid hydrofluoric…
A: Answer:Two concepts will be required to arrange the given solutions in increasing order of pH:Salt…
Q: List the name and position for each substituent in this organic molecule on a separate row of the…
A: Answer:A question based on nomenclature of organic compounds. An organic compound is given whose…
Q: Which expression best describes the relationship between the solubility product, Ksp, and the…
A: Answer:Ionic compounds that have low solubility in water are called as sparingly soluble compounds…
Q: 4. Draw product of the following reaction. 1. H30Ⓒ 2. A OEt POEt
A: Esterification reaction:Carboxylic acid reacts with alcohol in the presence of an acid catalyst to…
Q: 4. The following two compounds are protonated when treated with one mole equivalent of a strong acid…
A: Basicity is defined as the electron donation ability of a molecule. If it is able to donate…
Q: Consider the electrochemical cell reaction of hydrogen gas and chlorine gas to produce 1 mol of HCI…
A:
Q: Consider the insoluble compound zinc carbonate, ZnCO3. The zinc ion also forms a complex with…
A: Answer:Le-chatalier's principle states that on changing any parameter of the system that is in…
Q: Complete the electron-dot structure for sinapaldehyde showing lone pairs and identifying multiple…
A: Given :Incomlpete structure for sinapaldehyde.We need to complete the electron-dot structure for…
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

- [IV2] Instructions: Follow the flow of electrons indicated by the curved arrows in each of the following reactions, and predict the products that result: (refer to the photo below)Using compounds m-p from image 1please fill in the columns in image 2Repeat Problem 14.36 for [8]annulene. Draw the π MO energy diagram for [10]annulene, similar to those in Figures 14-19 and 14-21. Fill up the orbitals with the appropriate number of π electrons. Based on this diagram, should [10]annulenebe aromatic or antiaromatic? Explain.
- QUESTION- Label these spectra attached. Use them to discuss differences in reactant and product spectra that determine/support positive product formation. please no general responses, be specific to peaks in the spectra attached. EXPERIMENT: Synthesis of Benzil - Multi-Step Synthesis Part B benzoin + NH4NO3 ==== benzil ReferencesLehman, J.W. Operational Organic Chemistry: A Problem- Solving Approach to the LaboratoryCourse, 3 rd Ed., Prentice-Hall, Inc: New Jersey, 1999, p. 449.k Please don't provide handwritten solutin.Assign the stereochemical configuration (E or Z) for the alkene below. Show your work, indicating clearly which groups are assigned high priority (e.g., through assigning the groups numbers, circling only the high priority groups, or labeling groups as high or low).
- Please help. Questions in images. Solvent Rf of A Rf of B Rf of C 10% ethyl acetate-90% hexanes .12 .07 .01 20% ethyl acetate-80% hexanes .34 .29 .10 30% ethyl acetate-70% hexanes .53 .42 .22 40% ethyl acetate-60% hexanes .72 .61 .35For each of the following groups of molecules (labeled A–E), rank the resonance forms in order from least important (5) to most important (1)How to get the wt. sample, g.

