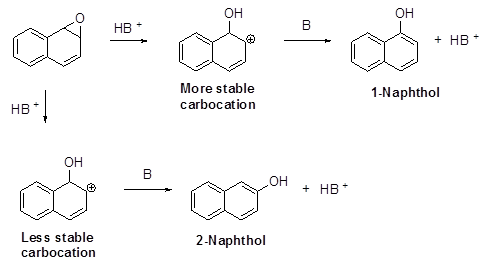
Draw all possible resonance contributors for the two carbocations in the preceding reaction. Use the resonance contributors to explain why 1-naphthol is the major product of the reaction.
Q: Why do reactions like this require a polar protic solvent even though they mechanistically follow an…
A: Polar protic solvents are the solvents which have tendency to solvate a nucleophile with the help of…
Q: Draw the resonance contributors for the following carbocation
A: Please find below the resonance contributors of the above carbocation.
Q: Which set of starting materials can be used to make the target molecule in one step? HO. Но Target…
A:
Q: OCH3 FeBr3 , Br2 LOCH3 Br
A: We have to draw resonance structures of anisole or the intermediate in the given reaction to…
Q: What starting materials or reagents are best used to carry out the following reaction?
A: This transformation can be achieved by Friedel-Crafts alkylation. But, Friedel-Crafts alkyaltion…
Q: H;O H,SO, (cat)
A: This reaction involves formation of carbocation intermediate which exapnds ring to get stable…
Q: HO
A: -> When Br2 attack to double bond first of all bromonium ion formed then nucleophile can open the…
Q: Draw a resonance structure that explains the unusual stability of the byproduct in the Gabriel…
A: Resonance structure:If the Lewis structure of a molecule or ion cannot explain by a single structure…
Q: 6. Which of the following alkyl halides CANNOT undergo E2 elimination? (circle your answer) Br Br Br
A:
Q: Provide the most accurate answer from the table of choices to best complete these reactions. Rxn L…
A:
Q: CH3 нн エ -エ、 エエ
A: As the given base is LDA i.e. Lithium diisopropylamide. It is a bulky base which favours the…
Q: How does changing the leaving group from Cl− to Br− affect the rate of an E1 reaction?
A: In the halogen family, the basicity of the halogen can be given as: F->Cl->Br->I- Fluorine…
Q: 2.) Draw the major product of the following reaction. Br
A: The ion is a very strong base and a very poor nucleophile because of the 3 bulky CH3 groups on the…
Q: A student attempted the following reaction. What was the result and why? + ОН NaCI NaOH + Isolated…
A: Reaction of alcohols with metal salts is a kind of substitution reaction which leads to the…
Q: H. This transformation can be performed with some reagent or combination of the reagents listed…
A: The question is based on the concept of chemical reactions. we have to systematically carry out…
Q: Draw the reaction mechanism of the E2 reaction between 2-chloro-2- methylpentane with potasium…
A: When 2- chloro -2- methyl pentane is treated with potassium hydroxide, it undergoes bimolecular…
Q: Propose a mechanism for the entire reaction of pent-1-yne with 2 moles of HBr. Show why…
A: In organic chemistry, Markovnikov's rule is used to describe the outcome of some addition reactions.…
Q: Br Give the reagents needed to complete the following transformation. May. use 1) & 2) over a single…
A: In the above transformation the starting material taken is isobutyl bromide. Its IUPAC name is…
Q: Which of the following anions is the best leaving group in an Sn1 reaction? а. F- b. Но- c. NH2- d.…
A:
Q: Step 6: Put it all together. Complete the two-step synthesis by selecting the reagents and starting…
A:
Q: Which reagents would best accomplish this transformation? %3D .C. HO, CH3 CH3 CH3 (A) K,Cr,O, +…
A: The reaction given is,
Q: Using resonance contributors for the carbocation intermediate, explain why a phenyl group is an…
A: Halogenation of phenyl ring in the presence of lewis acid promotes ortho - para substitution. It's…
Q: he Table of Reagents, select those that would be used to carry out the following transformation: NH2…
A: A sequential reaction is given and asked what appropriate reagents are used for this reaction.
Q: Why does the leaving group rarely act as a base in SN1 reactions? The ABC step is exothermic and…
A: In a SN1 reaction the leaving group to leave it must be able to accept electron ,if a base remove…
Q: In the third phase of the reaction, what is the primary role of compound C? H.0 CH Route 1 :OH2 CH3…
A:
Q: This reaction is an example of conjugate addition of a nucleophile to an a.B-unsaturated carbonyl.…
A: conjugate 1,4 addition is called Michael addition
Q: 9. Provide the major dehydration product of the following reaction. Recall which of the four…
A:
Q: 2.3 Determine the reagent required for the following transformation. a) Br b) Br
A:
Q: What is the most likely dehydration product of the following reaction?
A: The question is based on the concept of organic reactions. We have to identify the product formed…
Q: Which of these would be the best electrophile for an Sy1 reaction? A) CH;Br B) (CH3);CBr C)…
A:
Q: a. Indicate how each of the following factors affects an E1 reaction: 1. the strength of the base…
A: The following given factors affect an E1 reaction and an E2 reaction are explained. 1. the strength…
Q: What reagent(s) should be used to perform the following transformation most efficiently? A 1.…
A:
Q: Draw the major organic product of the reaction. Select 1. CH3CH,MgBr 2. H*
A: Grignard reagent is a special kind of reagent because normally the alkyl group acts as electrophiles…
Q: What are the conditions needed in a synthesis to promote an SN1 type of reaction? For example do we…
A: SN1 reaction is a nucleophilic substitution reaction in which the rate determining step depends on…
Q: Draw the major product of this E1 reaction. Ignore any inorganic byproducts. OH
A: Products are present on right hand side and reactants are present on left hand side in a reaction.
Q: Perform a retrosynthetic analysis by working backwards two steps in the synthesis below. Identify…
A: Given is retrosynthetic analysis
Q: Identify the reagent(s) needed for the following transformation: OH ?. + En 1. BH3 THF 2. H2O2, NAOH…
A: Given: Transformation of a reaction. To find : Best suitable reagents for the transformation.…
Q: CH3 DH H.
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: What is the proper reagent for this transformation?
A: Given transformation, Propene to 2-Bromopropane
Q: -CH, CH, HyC
A:
Q: What is the jer product of the reactron? ma a
A: Alkenes reacts with HCl via carbocation formation mechanism.
Q: Draw an alkyl bromide that would produce primarily this alkene through an E1 elimination reaction.…
A: Elimination reaction of alkyl halide: Alkyl halide gives an elimination reaction by reacting with a…
Q: KNH2 + KCI NH3 ČI -30 °C NH2 chloro is only moderately electron-withdrawing traditional SAr…
A:
Q: What is the best choice of reagent to accomplish the following transformation: HO. O H20, Br2 (1]…
A: This reaction is named HBO [hydro boration oxidation ] in this reaction, reagent are used as- (1)…
Q: Rank the attached dienophiles in order of increasing reactivity.
A: The groups having strong EDG attached are strong Diene. Reactivity order of dienes is given below:
Q: What is the right reagent in the reaction below
A: In this question, we will write the right reagent for this reaction given above. You can see the…
Q: b. Rank by rate of electrophilic aromatic substitution. The compound that reacts the fastest in an…
A: The answer is given below:
Q: CH3 .CH3 Br H. H.
A: Alkyl halide gives elimination reactions when they react with a strong base. The E2 elimination…
Q: Explain why a diazonium group on a benzene ring cannot be used to direct an incoming substituent to…
A: Generally, the electron withdrawing substituent present in benzene ring can direct an incoming…
Draw all possible resonance contributors for the two carbocations in the preceding reaction. Use the resonance contributors to explain why 1-naphthol is the major product of the reaction.
Step by step
Solved in 2 steps with 2 images

- Draw resonance structures of anisole or the intermediate in the above reaction to demonstrate why the methoxy group is an o,p-directorExplain why the trifluoromethane sulfonate (TfO-) ion is a better leaving group than the methanesulfonate (MsO-) ion.What is the first step in an SN1 reaction? Group of answer choices Attack of the nucleophile. Loss of the leaving group. Protonation of the leaving group.
- What kind of reagent is KmnO4? Draw the possible reactions of this reagent with the specific compounds tested in the following reaction?Please fill out for each reaction i.Fill in the missing starting materials, products, or reagents as necessary.If no reaction occurs, write "N.R." and explain why this is the case. ii. Label each transformation as SN1, SN2, or acid/base. iii. Indicate if the product is racemic or a single enantiomer.How do you explain why in base-catalyzed halogenation of acetone, the second (and third) halogenation occurs on the same carbon, while the other methyl group does not occur on the carbon?
- What is the right reagent in the reaction belowWhich alkyl halide in each pair is more reactive in an E2 reaction with hydroxide ion?Explain using resonance structures of the intermediates (please explain and draw them out) why the bromination of phenol is faster than the bromination of phenyl ester?
- Explain why a diazonium group on a benzene ring cannot be used to direct an incoming substituent to the meta position.How does changing the base from −OH to H2O affect the rate of an E2 reaction?If you were to add Br2 to this reagent, what would happen? Would there be an anti Br2 addition, or would the OH attack the carbon after the first bromine is attached so that a ring is formed?

