Q: Use the References to access important values if needed for this question. The pH of an aqueous…
A:
Q: Question 27 Predict the FINAL (?) product for each of the following reaction or synthetic chain: A B…
A:
Q: ← Problem 43 of 46 Submit Draw the major product of this reaction. Ignore inorganic byproducts and…
A: 1. 2.
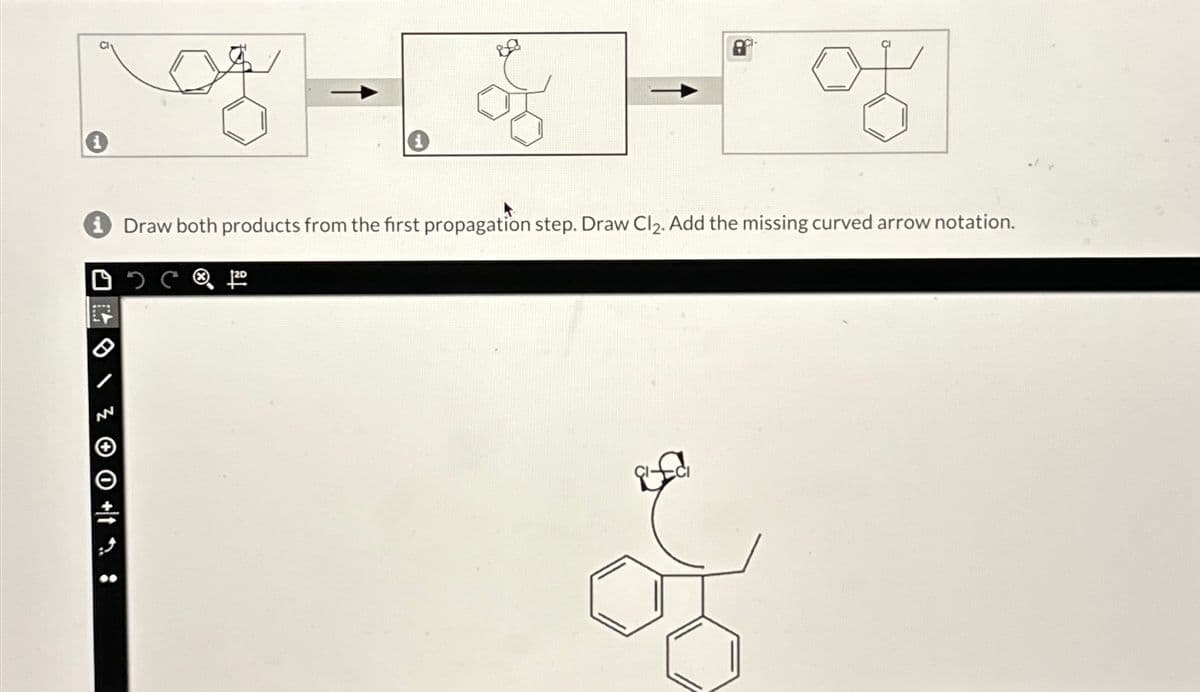
Q: Draw the bridged bromonium ion that is formed as an intermediate during the bromination of this…
A: Step 1: Step 2: Step 3: Step 4:
Q: Palmitic acid (C16H32O2) is a major component of the oil from the fruit of oil palms (palm oil). The…
A: Bomb calorimetry is also known as constant volume calorimetry and the heat absorbed by the…
Q: Do not use carboxylation mechanism but show full mechanism for the question below
A: Step 1:• Firstly strong base i.e NaOEt abstract the acidic proton to forms enolate anion which…
Q: U U U CI Recall that the Friedel-Crafts alkylation reaction forms a new C-C bond between an aromatic…
A: The objective of the question is to identify which of the given groups (OH, Cl, +a, NO2, Cl as the…
Q: 5) What series of synthetic steps could be used to carry out the transformation shown below? Draw…
A: Question 5(a)Detailed explanation: Key references:
Q: →> CH3CH₂OH HCl n + 0 OH CH CH OH 3 HCI
A: Step 1:
Q: Question 25 Predict the FINAL (?) product for each of the following reaction or synthetic chain: 1.…
A: Step 1:
Q: A chemistry graduate student is given 500. mL of a 0.50M chlorous acid (HCIO) solution. Chlorous…
A:
Q: What is the IUPAC name for the compound number E shown in the figure?
A:
Q: Draw Walsh diagram for a [IrCl 4]3-in Td symmetry undergoing Jahn - Teller distortion to [IrCl 4]3-…
A: The objective of the question is to understand the Jahn-Teller distortion of the [IrCl4]3- complex…
Q: How many grams of solid ammonium bromide should be added to 1.50 L of a 0.0879 M ammonia solution to…
A: The objective of this question is to determine the amount of solid ammonium bromide that needs to be…
Q: A small piece of papyrus is to be 14C dated by a 10 minute run of the detector set to record 14C…
A: The objective of the given data is to determine the required values of the papyrus sample that is…
Q: Chemistry
A:
Q: 2. Devise a synthetic route to accomplish the following transformations. (These transformations may…
A: Approach to solving the question: Detailed explanation:
Q: Step by step full solution
A: These values are very small, reflecting the low concentrations and the small sample volume assumed.…
Q: The glass floor of the CN Tower’s observation deck can withstand a pressure of 14 000 kPa. How many…
A: The objective of this question is to convert a pressure value from kilopascals (kPa) to pounds per…
Q: A small amount of strong acid is added to a 0.15 M HF and 0.15 M F buffer solution. Which species…
A: The objective of the question is to identify which species in a buffer solution of HF and F- will…
Q: The surface of the eye is wet by tears, and to minimize evaporation, the tear layer is coated with…
A: The reaction described involved the condensation of the carbonyl group of oleic acid with the…
Q: III O Chemical Reactions Determining the volume of base needed to titrate a given mass of acid 1/5 A…
A: To calculate the volume of NaOH solution needed to reach the equivalence point, we first need to…
Q: 5. What is the major product of the following reaction? Explain. Br i NaOH ?
A: Step 1: Step 2: Step 3: Step 4:
Q: None
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: Draw the mechanism for the following reaction. OH + H2N H3C H3C Н* butanal glycine Imine OH
A: Thank you.
Q: Problem 34 of 50 Submit Draw the product that could be formed when 1,3-butadiene reacts with…
A: Step 1:Mechanism Diels-Alder Reaction:It's a pericyclic reaction, meaning all bond breaking and…
Q: Give the product of the reaction shown below. Would the enantiomer or a diastereomer of the molecule…
A:
Q: A 3.00 mol portion of gas initially at a pressure of 1.00 atm and a temperature of 100. °C is…
A: To solve this problem, we can use the combined gas law equation, P1V1/T1=P2V2/T2, where P is…
Q: one: Topic Molecular Spectroscopy (16 marks) You were given an unknown mixture that contains two…
A: Step 1: Calculate the absorbance of the mixture at each wavelength (510 nm for salt A and 575 nm for…
Q: 7. Balancing Equation Practice ections: Balance the following chemical equations. Write final…
A: Please see the attached image for the solution. If there are queries, please do not hesitate to ask.…
Q: The first enzyme to rise in acute pancreatitis is ________________, and the enzyme which stays…
A: The objective of the question is to identify the enzymes that are affected during acute…
Q: 2. Indicate whether you would use NaOEt or t-BuOK to achieve each following transformations. (a) Br…
A: Step 1:
Q: 9. For a spherical harmonic given by (a) What is the value of 1? (b)What is the value of my ? 3 Y…
A: Step 1:Ans:In spherical harmonic case the value of l and m decide in simply way we say that (1) The…
Q: What is calibration and why is it essential in relation to food analysis? Provide examples.
A: What is calibration ? Calibration is comparing the measurements of an instrument or piece of…
Q: Question 1 In carbon NMR, the carbon atom of the carbonyl group in aldehydes and ketones has a…
A: In carbon NMR spectroscopy, the chemical shift refers to the position of a carbon atom's resonance…
Q: 3. What is the change in entropy for the process where the energy is initially associated with…
A: Step 1: Step 2: Step 3: Step 4:
Q: What's the reaction of zinc phosphate?
A:
Q: For benzene, the ∆H° of vaporization is 30.72 kJ/mol and the ∆S° of vaporization is 86.97 J/mol・K.…
A: Step 1: Step 2: Step 3: Step 4:
Q: For the electrochemical cell represented by this shorthand notation, which statement below is true?…
A: The objective of the question is to identify the correct statement about the electrochemical cell…
Q: provide A and B both answer with explanation
A: In case of any query feel free to ask.
Q: help question 8
A:
Q: show me full curly arrow mechanism for electrophilic substitution reaction of droperidol where…
A: ### Step 1: Formation of the ElectrophileIn many electrophilic aromatic substitution reactions…
Q: A solution is prepared at 25 °C that is initially 0.92M in ammonia (NH3), a weak base with K₂ = 1.8…
A: 1. Given: [NH3]=0.92M;[NH4+]=1.5M;Kb=1.8x10−5Step 1: Write the Henderson-Hasselbach…
Q: What are the products for the reaction of magnesium hydroxide with water? Mg2+ (aq) + 2 OH¯ (aq)…
A:
Q: None
A:
Q: O Chemical Reactions Standardizing a base solution by titration 3/5 Bis A chemistry student needs to…
A:
Q: Calculate the pH of solution of the following weak acid. Use Table 14.2 and Appendix H in your…
A: Step 1:Amphiprotic ion is a specie that can accept as well as donate the H+ ion. It means it can act…
Q: A buffer solution contains 0.361 M ammonium bromide and 0.470 M ammonia.If 0.0565 moles of…
A: The objective of this question is to calculate the pH of a buffer solution after the addition of a…
Q: Convert from rectangular to spherical coordinates. (Use symbolic notation and fractions where…
A: The objective of the question is to convert the given rectangular coordinates (6,-6√3,12√3) into…
Q: Nitrous acid, HNO2, is a weak acid. What is the reaction of nitrous acid in water? HNO2 (aq) + H2O…
A: The question is asking for the correct reaction of nitrous acid (HNO2) in water. Nitrous acid is a…
Step by step
Solved in 2 steps with 1 images

- For the reaction shown, draw the product of one mole of reagent adding across the triple bond, and then draw the product of a second mole of reagent.Add curved arrows to each step in the attached reaction sequence.Can you please draw out a complete curve arrow mechanism with the reagent and intermediate steps?

