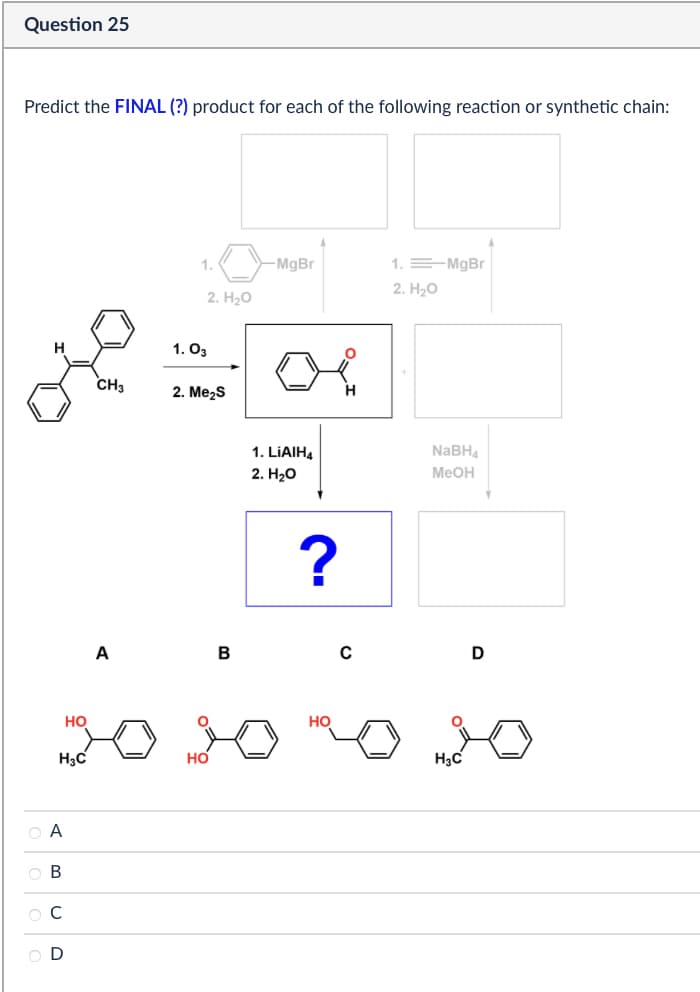
Question 25 Predict the FINAL (?) product for each of the following reaction or synthetic chain: 1. 2. H₂O CH3 1. 03 2. Me₂S -MgBr 1. MgBr 2. H₂O 1. LiAlH4 2. H₂O ? H NaBH4 MeOH A B C D HO H3C HO ABCD ов OD HO H3C
Q: These disorders are associated with inborn errors of metabolism: Question 13 options:…
A: Option a: This option is incorrect because Types 1 and 2 diabetes mellitus can impact metabolism,…
Q: Which of the following is a correct net ionic equation for the reaction of a metal and an acid? ○ Mg…
A: The objective of the question is to identify the correct net ionic equation for the reaction of a…
Q: Question 13 Choose the correct chemical structure for every single acronym used below: i-BuBr ? A B…
A: Step 1:Step 1:The acronym "i-BuBr" stands for "isobutyl bromide." Explanation:- 1. "i-" Prefix:…
Q: Which of the following aqueous solutions are good buffer systems? (Select all that apply.) 0.20 M…
A: The objective of this question is to identify which of the given aqueous solutions are good buffer…
Q: A prepared buffer solution contains a mixture of H2PO4 and HPO ions. Which species will react with…
A: The objective of the question is to identify which species in a buffer solution will react with any…
Q: Suppose all the hydrogen atoms in this molecule are replaced by chlorine atoms: དཡིག་བྱ། །ད Draw a…
A: Thank you.
Q: 4. For each reaction, look at all the reagents and conditions, and determine if the reaction will…
A:
Q: In a titration, 33.1 mL of 0.82 M KOH were needed to neutralize 25.0 mL of HCl. How many moles of…
A: The objective of this question is to determine the number of moles of HCl present in the solution…
Q: Incorrect Your answer is wrong. In addition to checking your math, check that you used the right…
A: Step 1:Step 2:
Q: What are the formal charges on terminal N, central N, and O atoms in the following molecule? OA-2,…
A: Step 1: Formula for calculating formal charge on an atom : Formal charge = (Numbers of valence…
Q: The acid dissociation K of acetic acid (HCH3CO2) is 1.8 × 105. -4 Calculate the pH of a 4.1 × 10 M…
A: The objective of this question is to calculate the pH of an acetic acid solution given its molarity…
Q: What type of compound is H2CO3? a base a diprotic acid a monoprotic acid a triprotic acid
A: The objective of the question is to identify the type of compound H2CO3 is among the given options.
Q: Please correct answer and don't use hend raiting
A: Step 1: Step 2: Step 3: Step 4:
Q: None
A: Step 1: Step 2: Step 3: Step 4:
Q: Question 34 Predict the FINAL (?) product for each of the following reaction or synthetic chain:…
A: Step 1: Step 2:Mechanism Step 3: Step 4:
Q: U U U CI Recall that the Friedel-Crafts alkylation reaction forms a new C-C bond between an aromatic…
A: The objective of the question is to identify which of the given groups (OH, Cl, +a, NO2, Cl as the…
Q: Draw the major product of the aldol addition reaction. Ignore inorganic byproducts. I H H Q 1. NaOH…
A:
Q: Question 36 Propose a synthesis and identify the the FINAL (?) product: Hint 1: Zaitsev product is…
A: Step 1:Step 2:Hence option A is correct.Step 3: Step 4:
Q: 1. Calculate the density of oxygen, O2, under each of the following conditions: STP 1.00 atmatm and…
A: The objective of the first part of the question is to calculate the density of oxygen under two…
Q: What's the value of solubility product constant for zinc phosphate?
A: Here are some of the references for the solubility product constant of zinc phosphate at 25oC:…
Q: Starting from cyclohexane make the following compounds. You must start with cyclohexane, but then…
A: Step 1:1) In this preparation, • firstly cyclohexane converted to bromo cyclohexane in…
Q: What is the IUPAC name for the compound shown in the figure? Br CI
A:
Q: An air-gap parallel plate capacitor whose plates have an area of A=0,38m² are separated by a…
A: Step 1:Step 2:
Q: Draw the major product of this aldol condensation reaction. Ignore inorganic byproducts. + H a H Q…
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: What is the pH of a solution made by dissolving 4.61 grams of calcium fluoride in enough water to…
A: Calculate the moles of CaF2: The molar mass of CaF2 is approximately 78.08 g/mol. So, the moles of…
Q: What are some characteristics of proteins? Question 27 options: Average…
A: The objective of the question is to identify the correct characteristics of proteins from the given…
Q: Question 2 2 pts Question 3 1 pt Question 4 2 pts Question 5 1 pt Question 6 2 pts Question 7 1 pt…
A: pH is the measure of acidity in aqueous solution. The small concentrations of protons are expressed…
Q: A solution has a pOH of 5.60. What is the hydroxide ion concentration?
A: The objective of this question is to find the hydroxide ion concentration of a solution given its…
Q: Question 10 Please predict the products for each of the following reactions: 1. Na 2. PrBr เวรเ 10…
A: Step 1:At first presence of Na metal ,it released one electron , which abstract alcoholic proton and…
Q: A solution contains 0.0220 M Pb2+(aq) and 0.0220 M Sr²+(aq). If you add SO2(aq), what will be the…
A: Step 1: SrSO4 begins to precipitate - [Sr2+] = 0.0220 M We know at 250 C Ksp of SrSO4 is…
Q: 2:15 AM Sun Apr 14 5% Question 4 of 8 One of the commercial uses of sulfuric acid is the production…
A:
Q: Formula Lewis structure VSEPR model Geometry name AB(E) notation 1.C2H6 H H-C ·C-H H H 2. C2H4 3.…
A: The objective of the question is to determine the Lewis structure, VSEPR model, Geometry name, AB(E)…
Q: :$:$;);$):):$$:$;$;$
A:
Q: Give the IUPAC name of the molecule.
A: Step 1:name of the molecule is "3-tert-butylcyclopentane-1-carboxylic acid"Explanation 1. Select…
Q: Why is appropriate sampling important in relation to food?
A: Approach to solving the question:SamplingDetailed explanation:Appropriate sampling is crucial in…
Q: Sodium hydroxide is a substance that causes severe burns. It is used in many educational laboratory…
A: The objective of the question is to identify the correct statements about the nature of sodium…
Q: A solution has a hydroxide ion concentration of 7.3×10−5M. What is the pOH of the solution?
A: The objective of this question is to calculate the pOH of a solution given the concentration of…
Q: Calculate the K, and AG° for the following reaction at 25 °C: 2 AgNO, (aq) + Zn(s) Zn (NO,), (aq) +…
A: Step 1 The given reaction is: 2AgNO3 aq + Zns ⇌ Zn(NO3)2 aq+2Ags Equilibrium constant (Kc) of a…
Q: Can you explain the problem? What if it was a base?
A:
Q: יוון UIPAC name of this compound?
A: The IUPAC nomenclature of organic compounds follows this naming format:Writing the name of organic…
Q: None
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: 20. What would be the major product of the following reaction? Br₂, CCI C₂H5 C2H5 C2H5 C₂H5 H3C Br…
A: Anti addition of bromination of alkene in presence of carbon tetrachloride ,. This bromine atom will…
Q: High serum total protein with high levels of both albumin and globulins is usually seen in…
A: In Waldenström's macroglobulinemia:1. Malignant B-lymphocytes proliferate and overproduce monoclonal…
Q: Which one shows aromacity? OA. Cycloheptatrienyl anion. B. Cyclopentadienyl anion OC…
A: The objective of the question is to identify which among the given options exhibits aromaticity.…
Q: 1. Below are two potential methods for preparing the same ether, but only one of them is successful.…
A:
Q: Determine the density of CO2 at 745 mmHg and 65 degrees C?
A: The objective of this question is to calculate the density of carbon dioxide (CO2) under specific…
Q: Br OH H₂O HO (14 pts) Provide a complete mechanism for the reaction in the box. Include all…
A: Step 1:Loss of Br- to form a less stable 2o carbocation. Step 2:Hydride (H-) shift to form more…
Q: None
A: Part 2: Explanation:Step 1: Determine the moles of NaOH used in each trial:The moles of NaOH can be…
Q: Macmillan Learning An 80.0 g sample of a gas was heated from 25 °C to 225 °C. During this process,…
A: The objective of the question is to calculate the specific heat of the gas. The specific heat of a…
Q: Please fast expert answer solutions and dwor computer figure
A: In order to determine an aromatic compound, it must be: cyclicplanarComplete delocalization of π…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- mmol of 1,3-dibenzoylpropane used to 4 sig. fig Used: 405 mg molecular weight: 252.31 g/molQuestion A wittig reaction experiment: Week 1 -》 preparation of phosphonium salt Materials : triphenylphosphine (5.3g),methyl Bromoacetate 3.36g (2.1ml) , ethanol 30ml . The yield obtained was 8.93g . The percentage yield obtained was 106.4% Week 2 -》 Formation of the ylide and wittig reaction Materials :Phosphonium salt (5g), napthyl-2- carboxwaldehyde( 2.65g), 20 ml of water and 5 nl of Nahco3. Yield obtained was 0.51g .Calculate the percentage yield Week 3: Solvent free wittig reaction Materials: Benzyltriphenylphosphonium chloride (0.5g ), 4 - Bromoabenzaldehyde (0.24g), Potassium phosphate (tribasi c) 0.275g .The yield obtained was 1.21g. Calculate the percentage yieldNeed help on last question. The reaction is a fisher esterification. My unknown ester is pentyl acetate (amyl acetate) and my unknown alcohol is 1-pentanol. 1.5 mL of 1-pentanol used giving us 0.013806 moles used. 3.0 mL of acetic acid used giving us 0.052604 moles used. Please show all work (including identification of limiting reactant)
- an alkene when acting with HCl shows a weight increase of 52.14% .What is the molecular formula of the alkene?question 1/ 1.0 g of a mixture containing 80% CuSO4.5H2O is taken and heated to 120 oC and 160 oC. According to this, find the weight reductions percentages a) at 120 oC, b) at 160 oCCompounds Mass 3-nitrophthalic acid used 200 mg = 0.2g 8% aqueous hydrazine used 0.4mL 3-nitrophthalhydrazide obtained 130 mg = 0.13 g sodium hydrosulfite dihydrate 0.6 g luminol obtained 70 mg = 0.07 g compute yield for nitrophthalhydrazide in the first step (assume nitrophthalic acid is limiting reagent) compute yield for luminol in the second step (using nitrophthalhydrazide as limiting reagent) compute yield for the overall reaction
- For 20.7 the solutions for part c and d dont seem right. Shouldn't reacting with 1 eq of H2 just take away the C=C bond and not the C=O bond. (H2 reduces C=C selectively to form a ketone according to page 738. Also the both the C=C and C=O bond should be reduced if excess H2 is usedAcetanilide (C6H5NHCOCH3, M = 135.2) can beprepared from aniline (C6H5NH2, M = 93.1) and aceticanhydride ([CH3CO]2O, M = 102.1) according to thebalanced equation below. C6H5NH2 + (CH3CO)2O →C6H5NHCOCH3 + CH3COOH If 10.0 g acetanilide is isolated from the reaction of 10.0g aniline with 10.0 g acetic anhydride, what is the percentyield of the reaction?An unkown solvent has a formula of C4H8O and molecular weight of 72.11 g/mol. Determine the ff: a.) index of hydrohgen deficiency b.) all possible functional group present base on IHD
- in a lab going from benzil to hydrobenzoin (using NaBH and ETOH, i received a 152% percent yield (1g of benzoin at the beginning and ended with 1.52g of hydrobenzoin) and i was just wondering what could've went wrong?IR given is furoin the second picture shows the structure of furoin, can you label the peaks given I'm bad at IRCase #1: Someone stole the net from the High Wire act at the Crazy Circus! Without it, the show can't go on! The CSI team found a liquid at the crime scene. When analyzed, it was found to contain 5.94% H and 94.06% O. Who committed the heinous act? Whodunnit? Cases: Suspect 1: George Norman Suspect 2: Becky Baker George Norman recently had an accident where he cut himself with a knife. He cleaned the wound with rubbing alcohol. The formula for rubbing alcohol (isopropanol) is C3H8O. Becky Baker is a world famous pastry chef. She had just finished baking a cake at the time of the crime. The recipe called for vanilla. The formula for vanilla is C8H8O3. Suspect 3: Liz Lemon Suspect 4: Ben Linus Liz Lemon just had her nails done and she still has traces of nail polish remover on her hands. Nail polish remover contains acetone. The formula for acetone is C3H6O. Ben Linus had been walking around in the rain for about an hour before the…



