Draw major resonance structures, B and C, and one minor resonance structure, D. Be sure to include the formal charge, lone pairs, and hydrogens on nitrogen for structures B, C, and D. H. N" H. H. C major resonance structure #1 major resonance structure #2 H. H. minor resonance structure
Draw major resonance structures, B and C, and one minor resonance structure, D. Be sure to include the formal charge, lone pairs, and hydrogens on nitrogen for structures B, C, and D. H. N" H. H. C major resonance structure #1 major resonance structure #2 H. H. minor resonance structure
Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Andrei Straumanis
Chapter21: Nas: Nucleophilic Aromatic Substitution
Section: Chapter Questions
Problem 1E
Related questions
Question
Draw the major resonance structure structure for b,c, d
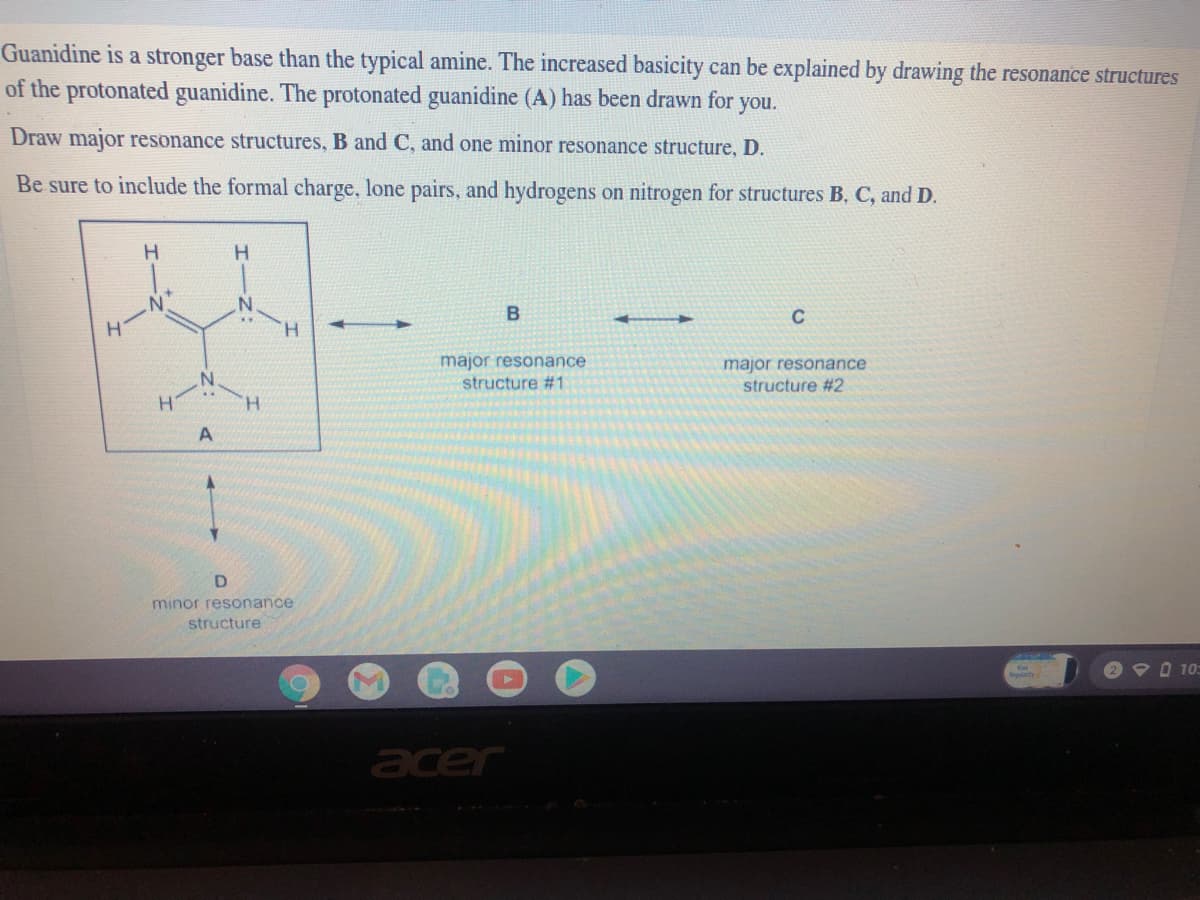
Transcribed Image Text:Guanidine is a stronger base than the typical amine. The increased basicity can be explained by drawing the resonance structures
of the protonated guanidine. The protonated guanidine (A) has been drawn for you.
Draw major resonance structures, B and C, and one minor resonance structure, D.
Be sure to include the formal charge, lone pairs, and hydrogens on nitrogen for structures B, C, and D.
H.
N*
H.
C
major resonance
major resonance
structure #2
structure #1
H.
H.
minor resonance
structure
9O 10:
acer
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning