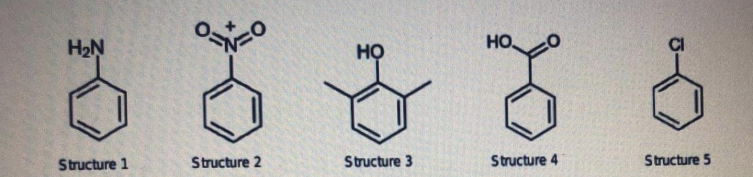
Which structure has the greatest number of pi electrons? A, Structure 1 B. Structure 3 C. Structure 4 D. Structure 5 Which molecule has the lowest resonance stability? A. Structure 1 B. Structure 2 C. Structure 3 D. Structure 4 . Which molecule has the least ability to form hydrogen bonds with water? A. Structure 1 B. Structure 2 C. Structure 4 D. Structure 5 Which aromatic molecules acts as both hydrogen bond donor and hydrogen bond acceptor? A. Structures 1, 2 and 3 B. Structures 2, 4 and 5 C. Structures 1, 3 and 4 D. Structures 3, 4 and 5
Which structure has the greatest number of pi electrons?
A, Structure 1
B. Structure 3
C. Structure 4
D. Structure 5
Which molecule has the lowest resonance stability?
A. Structure 1
B. Structure 2
C. Structure 3
D. Structure 4
. Which molecule has the least ability to form hydrogen bonds with water?
A. Structure 1
B. Structure 2
C. Structure 4
D. Structure 5
Which
acceptor?
A. Structures 1, 2 and 3
B. Structures 2, 4 and 5
C. Structures 1, 3 and 4
D. Structures 3, 4 and 5
Which among the five aromatic molecules will have the least ability to undergo molecular
stacking?
A. Structure 1
B. Structure 2
C. Structure 3
D. Structure
Which aromatic structures contains functional groups which are electron-withdrawing?
A. Structures 1, 2 and 4
B. Structures 2, 3 and 5
C. Structures 2, 4, and 5
D. Structures 1, 3, and 4
Which aromatic structures contain functional groups which are electron-donating?
A. Structures 1 and 3
B. Structures 1 and 4
C. Structures 3 and 5
D. Structures 3 and 4
In which structure do you expect two products after alkylation?
A. Structure 1
B. Structure 2
C. Structure 3
D. Structure 4
In which carbon position will an incoming halogen be located in Structure 3, relative to
the OH group?
A. Ortho position
B. Meta position
C. Para position
D. Ortho and para position
In which aromatic molecule do you expect to have a carbocation form during resonance
stabilization?
A. Structures 2, 3 and 4
B. Structures 2, 4 and 5
C. Structures 2, 3 and 5
D. Structures 1, 3 and 4
Step by step
Solved in 2 steps with 2 images







