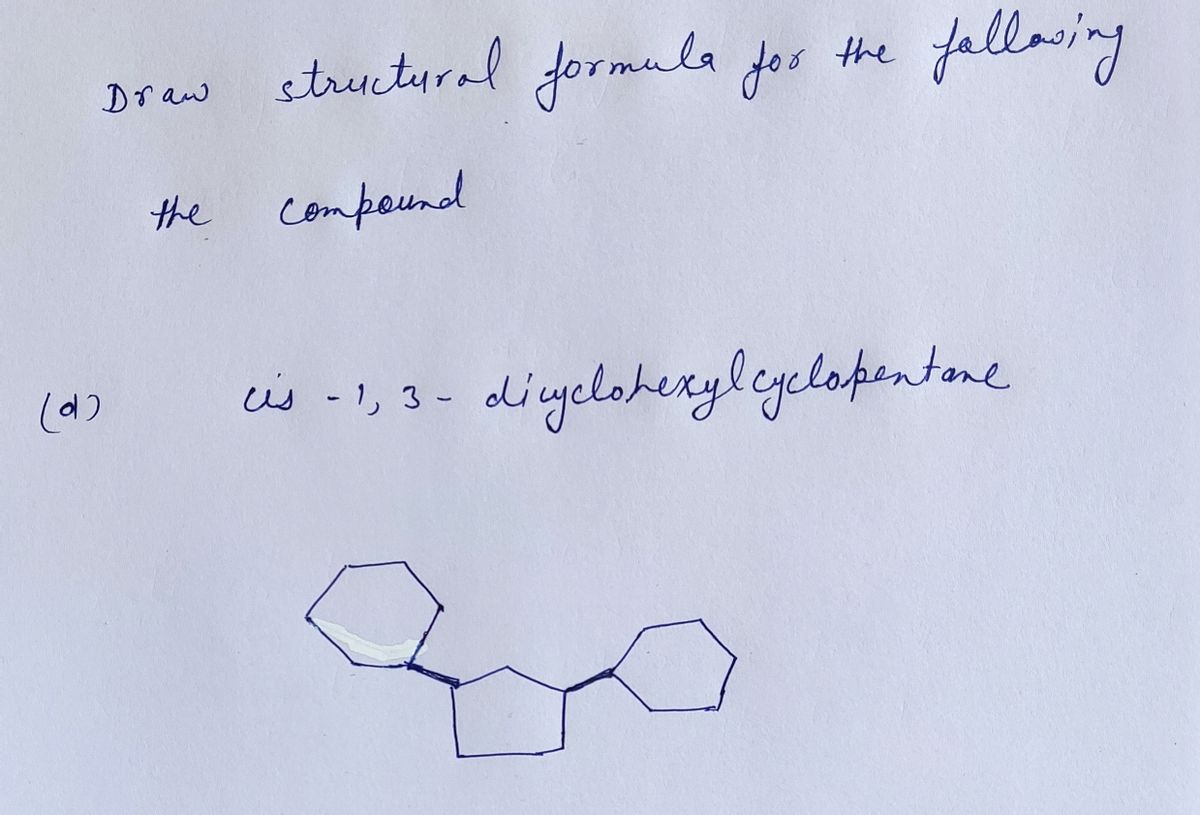
Draw structural formulas for the following the compounds: (d) cis-1,3-dicyclohexylcyclopentane (e) (Z)-2-cyclohexyl-4-methylhept-2-en-3-ol (f) 3-methoxybenzaldehyde
Q: Make the correction:
A: Calculated pH for NaOH solution is very fine and very correct hence their is no need of correction.F...
Q: The PHIVOLCS recently reported that Taal volcano emitted 15,500 tons of Sulfur Dioxide (SO2). How ma...
A:
Q: Given these initial concentrations: [SCN`] = 0.000644 M, [Fe³"] = 0.000505 M, and %3D 2+. [Fe(SCN)] ...
A:
Q: Which of the following molecules can participate in any kind of hydrogen bonding with water? H-CEN: ...
A: For hydrogen bonding, a hydrogen atom should be attached with a highly electronegative atom like F, ...
Q: Identify the 2 amino acids: Label the R groups and the alpha carbons Indicate and name the two funct...
A: Given compound :-
Q: - onder which conditions of temperature and pressure would a real gas behave most like an ideal gas?...
A: Given: To identify the conditions of temperature and pressure at which the real gas would behave mos...
Q: What causes the oscilation of the covalent bond oscillates resulting to varying electromagnetic fiel...
A: A charged particle produces an electric field. This electric field exerts a force on other charged p...
Q: Complete the following nuclear equation and identify X in each case
A: Nuclear reaction involves the formation of one or more nuclides by bombarding the nucleus with diffe...
Q: low many grams of KCI are in 125.0 mL of 0.375 M KCI? D 46.9 g D 3.49 g D 3.49 x 10g D 0.0469 g
A: The correct option is:
Q: What is the specific heat of an unknown substance if 1,462 J are required to heat 36 g of the substa...
A: Given Mass = 36 g heat = 1462 J T1 = 17°C T2 = 35°C specific heat (C) = ?
Q: The molecular diameter of helium, He, is 0.225 nm. At 0 °C and kPa, calculate the (iv) mean free pat...
A:
Q: Which of the numbered responses lists all the following stresses that would shift the equilibrium to...
A:
Q: Consider the equilibrium system C(s) + CO2(g) = 2CO(g) If C is removed, the equilibrium will and if ...
A:
Q: [이
A:
Q: For the reaction below which of the statements is correct? A+ 2B 3C The reaction is third-order over...
A: Answer: Order of reaction depends upon the molecularity of rate determining step.
Q: Which of the following molecules can only have dispersion forces as their sole intermolecular force ...
A: Given : We have to tell which of the following molecule will have dispersion forces of attraction.
Q: A chemist is studying the following equilibirum, which has the given equilibrium constant at a certa...
A:
Q: Which point represents solid-liquid-gas equilibria? 24.. 1 atm Phase I. Phasc ... P. ... ... Phase I...
A: Phase diagram: The phase diagram represents the different phases of the substance and the pressure a...
Q: A bottle of carbonated beverage has CO, concentration equal to 0.0961 Molar at a certain temperature...
A:
Q: Mixture of NH,CI, NaCl, SiO, Iron Shavings First Separation Technique First Separated Component Rema...
A: The mixture given contains ammonium chloride (NH4Cl), sodium chloride (NaCl), silicon dioxide (SiO2)...
Q: The half-life of iron-59 is 44.5 days. How much of a 3.00-mg sample will remain after 222.5 days? a ...
A: The half-life of iron-59, t1/2 = 44.5 days Time elapsed, T = 222.5 days Initial mass of the sample, ...
Q: Given these molecular weights of only nonpolar and non-ionic molecules consisting of only hydrogen a...
A: Melting point is directly proportional to number of the carbon atoms. Longer the carbon chain higher...
Q: Which of the following substances possess an internal structure of its constituent molecules that co...
A:
Q: For the first-order reaction 2N2O5→ 2N2O4 + O2 at a particular temperature, the half-life of N2Os is...
A: Given: The half-life of N2O5 = 0.90 hr Time = 2.4 hours
Q: Ultraviolet and visible molecular absorption spectroscopy is used primarily for quantitative analysi...
A: There are two statements given Ultraviolet and visible molecular absorption spectroscopy is used pr...
Q: scientists discovered that in outer space the oxygen atoms in CO2 can split off from the carbon, for...
A:
Q: find the pH.
A: This is question with multiple subparts. So, according to bartleby guidelines I need to solve only f...
Q: ", heat HO
A: Given : We have to write the reaction .
Q: (c) An ideal monoatomic gas at 1 bar and 300 K is expanded adiabatically against a constant pressure...
A:
Q: 10. Which ion has a higher ability to polarize? a) Na* or Mg* b) Mg²* or Ba?+ Explain your reasoning...
A: The polarizing power of cations depends on the charge density and size of the cation.
Q: Consider the equilibrium system C(s) + CO2 (g) = 2C0(g) If C is removed, the equilibrium will _____,...
A: C(s) + CO2(g) ⇌ 2CO(g)
Q: calculate the degree of unsaturation and draw structures that would meet the following descriptions
A: We have to find the structure of C10H10O2 which contain an aromatic ring one hydroxyl group and a ke...
Q: Compound A Compound B Compound C CI Step 1 Step 2 Circle one: Circle one: oxidation oxidation reduct...
A: The oxidation number represents the loss or gain of electrons to the respective elements of the comp...
Q: Which of the following molecules can have dipole-dipole interactions as their strongest intermolecul...
A:
Q: The above section of an NMR was taken from a student sample of this experiment. You see while the 2H...
A:
Q: Some measurements of the initial rate of a certain reaction are given in the table below. [N] H, ini...
A: The solution is as follows:
Q: 1) What is the equilibrium equation for the FeS(s) + 2 H30+ (aq) = Fe2+(aq) + H2S (aq) + 2 H20 (1)
A: Since you have posted multiple questions as per guidelines we can solve only one per session . If yo...
Q: Vanadium(V) oxide reacts with calcium according to the chemical equation below. When 20.0 moles of V...
A:
Q: Calculate the [H*] of 5.8 x 10-5 M magnesium hydroxide solution. O [H*] = 3.4X10-11 O [H*] = 3.2X10-...
A:
Q: Vanadium(V) oxide reacts with calcium according to the chemical equation below. When 20.0 moles of V...
A: • The given balanced chemical equation is, V2O5(s) + 5Ca(l) → 2V(l) + 5CaO(s) • The valu...
Q: Which one of the following is not equivalent to one mol of the substance? O 8.0 g He O 6.02 x 1023 C...
A: The correct option is:
Q: Which of the following molecules can initiate ion-dipole interactions with water? :N=N-N H. :N-F: :N...
A: Ion-dipole interaction occurs when an ion encounters a polar molecule. A cation or positive ion woul...
Q: How many moles of NaOH are present in 25.0 mL of a 0.1000 M NaOH solution? O 2.50 mol O 0.100 mol O ...
A:
Q: Using a chemical equation to find moles of product from moles ... Wine goes bad soon after opening b...
A:
Q: Which of the following (NBr3, BF3, OF2, IF3) is(are) exempted from the octet rule? Check all that ap...
A:
Q: Which of the following violates the octet rule? SF6 CO2 O AICI4 O Co3?
A: Concept is based on octet rule:
Q: It takes a minute and a half for the concentration of oxygen to decrease from 4.2 x102 M to 2.4 x103...
A: Initial concentration of oxygen gas = 4.2 x 10-2 M Final concentration of oxygen gas = 2.4 x 10-3 M ...
Q: ccording to the equation below, what is the limiting reactant if you combine 2 moles of N2 with 3 mo...
A: Given : We have to find out the limiting reagent.
Q: In each scenario, describe a step by step process in determining the unknown. Distilled water maybe ...
A: Density =mass/volume. We have to determine mass and volume separately.
Q: A piece of iron with a mass of 35 g is heated and then placed in a beaker containing 600 g of water....
A:
Draw structural formulas for the following the compounds:
(d) cis-1,3-dicyclohexylcyclopentane
(e) (Z)-2-cyclohexyl-4-methylhept-2-en-3-ol (f) 3-methoxybenzaldehyde
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Which of the structures below is (4R)-4ethyl-2,4- dimethylcyclohexan-1-one?I have a trans-3-chloro-1-methylcyclopentane. I react it with ethanol with heat. Knowing heat favors elimination, I suspect 3-methyl-cyclopentene as a product. Will I also get the substitution product 3-methoxy-1-methylcyclopentane, a pair of enantiomers and a pair of diastereomers?The cis and trans isomers of 2,3-dimethyloxirane both react with −OH to give butane-2,3diol. One stereoisomer gives a single achiral product, and one gives two chiral enantiomers. Which epoxide gives one product and which gives two?
- 1- Chloro-1,2-diphenyletane can undergo E2 elimination to give either cis- or trans-1,2-diphenylethylene (stilbene). Draw New man projections of the reactive conformations leading to both possible products, and suggest a reason why trans alkene is the major product?b. In a strongly basic solution, the starting material again converts into a molecule with the molecular formula C6H12OC6H12O. but with a completely different structure.Based on the more stable conformation (conformation A), need help providing two mechanisms for the E2 elimination of the protonated version of cis-1 using a general base (B:) to give alkenes 2 and 3. Clearly show which hydrogens meet the stereochemical requirements for E2-elimination Using Zaitsev’s rule, indicate which is favoured. Pictured is also a reaction mechanism to show what the alkenes 2 and 3 are. Thank you :)
- Q3. For the following reactions, predict the products, mechanism and name all the products obtained Catalytic hydrogenation of cycloalkenes. Reduction of cyclohexanone with Zinc amalgam (Zn/Hg alloy) in concentrated hydrochloric acid Addition of CH2I2 in an alkene in the presence of Zn-Cu 2-hexene + chlorine gas Halogenation of cyclohexane in the presence of light and catalyst Sulfonation of Cyclobutane with oleum Addition of HBr to methylcyclopropane(a) When (Z)-3-methylhex-3-ene undergoes hydroboration–oxidation, two isomericproducts are formed. Give their structures, and label each asymmetric carbon atomas (R) or (S). What is the relationship between these isomers?(b) Repeat part (a) for (E)-3-methylhex-3-ene. What is the relationship between the productsformed from (Z)-3-methylhex-3-ene and those formed from (E)-3-methylhex-3-ene?Arrange the following compounds in the increasing order of reactivity towards Conc.HNO3 & Conc.H₂SO4 1. Benzene 2. Chlorobenzene 3. phenol 4. Toluene 5. Nitrobenzene A 1.2.3.4.5 (B) 5.1,243 5.1.4.2.3 D 5.2.1.4.3
- There are a variety of isometric 1,2,3,4,5,6-hexachlorocyclohexanes, but one of them does not react with CH3CH2O- to give an elimation product. Provide the structures of this compound and briefly explain why it does not reactA chemist allows some pure (2S,3R)-3-bromo-2,3-diphenylpentane to react with a solution of sodium ethoxide(NaOCH2CH3) in ethanol. The products are two alkenes: A (cis-trans mixture) and B, a single pure isomer. Under thesame conditions, the reaction of (2S,3S)-3-bromo-2,3-diphenylpentane gives two alkenes, A (cis-trans mixture) and C.Upon catalytic hydrogenation, all three of these alkenes (A, B, and C) give 2,3-diphenylpentane. Determine the structuresof A, B, and C; give equations for their formation; and explain the stereospecificity of these reactionsWhich of the following compounds would you expect to bethe most generally reactive, and why? (Each corner in thesestructures represents a CH2 group.)



