Compound A Compound B Compound C CI Step 1 Step 2 Circle one: Circle one: oxidation oxidation reduction reduction neither neither Oxidation state of *C Oxidation state of *C Oxidation state of *C Show your work/calculations below: Compound A: Compound B: Compound C:
Compound A Compound B Compound C CI Step 1 Step 2 Circle one: Circle one: oxidation oxidation reduction reduction neither neither Oxidation state of *C Oxidation state of *C Oxidation state of *C Show your work/calculations below: Compound A: Compound B: Compound C:
Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Andrei Straumanis
Chapter19: Eas: Electrophilic Aromatic Substitution
Section: Chapter Questions
Problem 45E
Related questions
Question
100%
In the box write the oxidation state for the starred carbon. show work. circle whether its
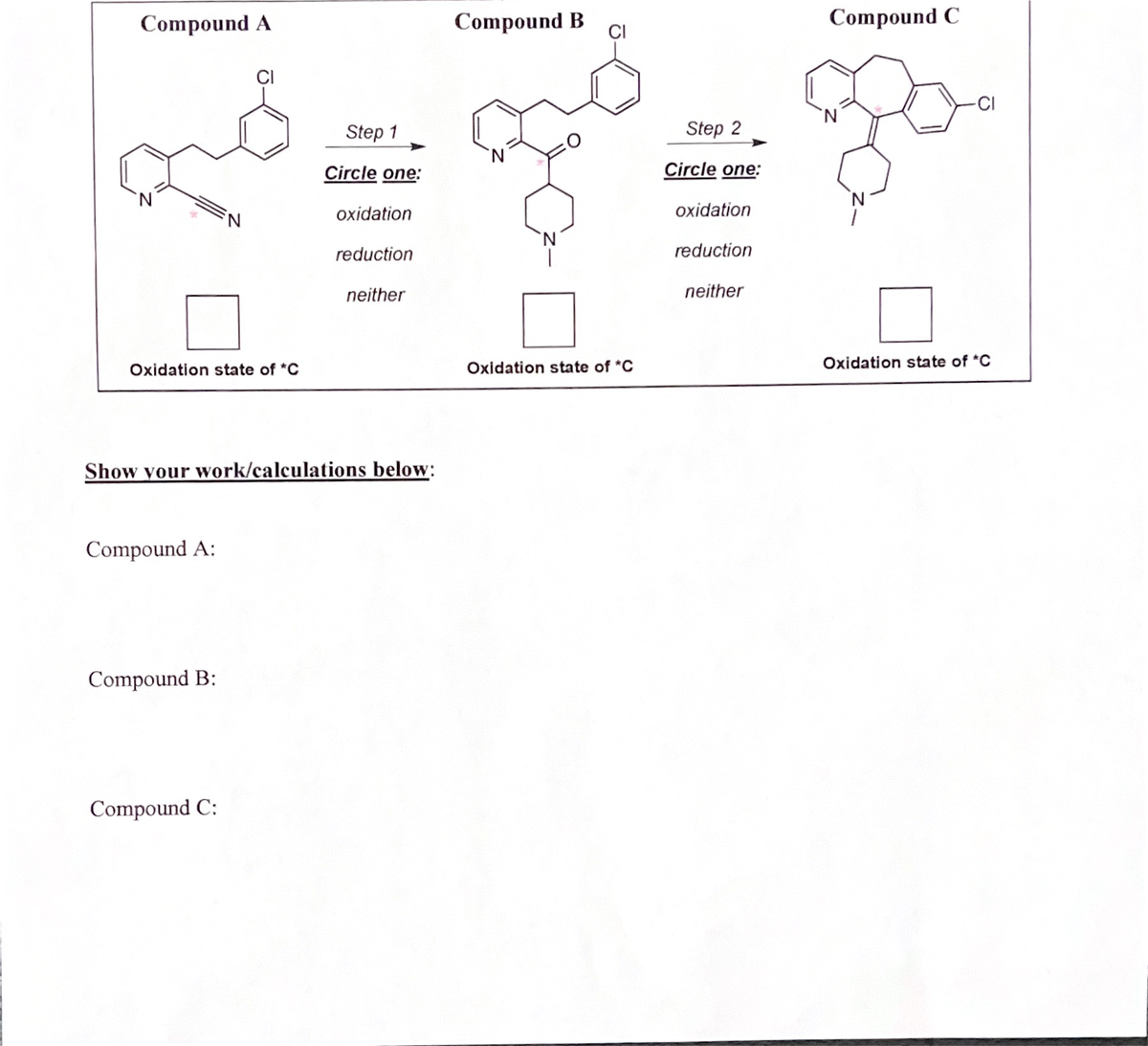
Transcribed Image Text:Compound A
Compound B
ÇI
Compound C
CI
N.
Step 1
Step 2
Circle one:
Circle one:
N.
oxidation
oxidation
reduction
reduction
neither
neither
Oxidation state of *C
Oxidation state of *C
Oxidation state of *C
Show your work/calculations below:
Compound A:
Compound B:
Compound C:
Expert Solution
Step 1
The oxidation number represents the loss or gain of electrons to the respective elements of the compound. If it has a positive charge, electrons are lost, and if it has a negative charge, electrons are gained by the element.
Step by step
Solved in 4 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning