Q: Draw a resonance structure, complete with all formal charges and lone (unshared) electron pairs,…
A: In this question, we will draw the structure of the resonance structure for aniline after given the…
Q: Convert between moles and numbers of atoms. A sample of lithium contains 2.24x1025 Li atoms. What…
A:
Q: A+B+C ⇒ D Exp. Initial B Initial A (mol/dm³) Init. Rate of Formation of D (mol/dm³ min-¹) (mol/dm³)…
A: k = Rate constant = 0.75 (mol/L)-3 s-1
Q: Use Bronted-Lowry acid base theory to write balanced net ionic equation for the following reaction.…
A:
Q: Calculate the approximate temperature of a 0.20 mol sample of gas at 750 mm Hg and a volume of 9 L
A:
Q: Which labeled atom will most likely react when acetic acid is treated with sodium hydroxide? Ο Α
A:
Q: 26. The e.m.f of the cell: Ni/Ni² (1.0 M) // Au (0.1 M)/ Au (E° Ni³/Ni - -0.25 V; E° Au¹/Au-1.5 V)…
A: Given, Ni/Ni2+(1.0 M) // Au3+(0.1 M)/Au Standard reduction potential E°Ni2+/Ni = -0.25 V E°Au3+/Au…
Q: What is the mol fraction of ethanol, C 2H 5OH, in a solution of 75.3g of ethanol and 525g of water?…
A: Number of moles is calculated by dividing the given mass with molecular mass.
Q: Which of the following statements is true? A catalyst raises the activation energy of a…
A: The catalyst is a chemical species that increases the rate of a chemical reaction and it does not…
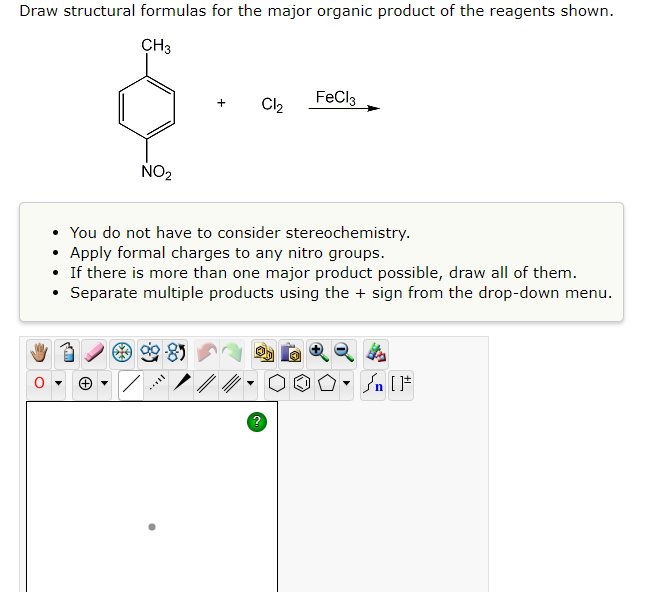
Q: CH,CH,COOH Carboxylic Acid SOCI₂ H₂O Acid anhydride CH₂CH₂COO-Na Acid chloride CH,OH Ester CH,CH,NH,…
A: Here we are required to predict the product of the reaction
Q: شاہد i) j) لا 1) ix II *** رو له s) سعد t)
A: The Name of compound is done using IUPAC rules .
Q: Please solve this
A:
Q: A membrane is made up of an alumina support (Al2O3) and a selective layer of a polymer called Naflón…
A: The membrane given is made up of an alumina and selective layer of polymer called Nafion.
Q: Choose the correct structure for the aldol condensation product of the reaction below: O O OH, H₂O…
A:
Q: 24. In a globular protein dissolved in water: a) polar amino acids are located towards the interior…
A: We have to determine the correct statement regarding globular proteins
Q: QUESTION 14 The (+) enantiomer of Lampent has a specific rotation of +31.5. What is the percent of…
A:
Q: 1) 2,4,6-Trimethylpyridine (also called 2,4,6-collidine) (shown in figure 1) consists of a pyridine…
A: Desired pH of the buffer solution = 7.25 Total volume of the buffer prepared = 250 mL Total…
Q: In the following: Al³+ (aq) + 3 e Al(s) E° = -1.66 V Cr³+ (aq) + 3e →→ Cr(s) E° = -0.74 V What is…
A:
Q: (dehydration). A 48.43 gram sample of a hydrate of CaSO4 was heated thoroughly in a porcelain…
A: Weight of hydrated CaSO4 = 48.43 gm Weight of anhydrous CaSO4 = 38.30 gm
Q: Antifreeze is often about 50.% ethylene glycol, C₂H6O2, in water. What is the molality of the…
A: Given - Ethylene glycol (C2H6O2) is present in water =50% Molar mass of ethylene…
Q: What is the pH of a buffer that is 0.08 M NaAc and 0.1M HAc. The Ka of HAC= 1.0 x 10^-4. O2.16 O…
A: Buffer solution: The solution that resists the change in pH is known as a buffer solution. The…
Q: Which is the correct net ionic equation for the following reaction MgSO4 (aq) + Cu(s) → Mg(s) +CuSO4…
A: A chemical equation that shows only those ion element compounds that directly take participate in…
Q: Name the following compounds. I just need part CC and DD.
A: Some important rules for IUPAC nomenclature Select longest possible continuous carbon chain with…
Q: Determine the pH of the solutions when Eu(OH)3(s) (Ksp = 9.38x1027) is mixed with each of the…
A:
Q: Need help with this one: Give at least three (3) specific reactions (ie. with organic reactant's and…
A:
Q: Calculateur (298.15 K, 1 bar) for nitrogen in air, assuming that the mole fraction of N₂ in air is…
A: Temperature = 298.15 K Pressure = 1 bar
Q: 12. What is the expected limiting molar conductivity (mS m² mol¹) for Ca(NO3)2? a) 25.012 b) 25.662…
A:
Q: What starting ketone will yield the following acetal? O Cyclohexanone O Propane-2,2-dione O…
A:
Q: In the molecule shown, the portion highlighted in red is called the ( ) group. o alkoxy O alkyl o…
A: The ester is formed from alcohol and carboxylic acid. The name of an ester is written as an alkyl…
Q: According to valence bond theory, what orbital contains the nitrogen lone pair? o sp op sp3 sp2 S
A: The correct answer is given below
Q: For each of the following substances: 2. 1. Classify the substance as a weak acid, weak base, strong…
A: Sodium oxalate, Na2OOCCOO(aq) or C2O42-(aq) is a fully deprotonated form of oxalic acid, which is a…
Q: Be sure to answer all parts. Draw the structure of a naturally occurring amino acid that is an…
A: We have to draw the structures of the amino acids
Q: 29.06 g butane, C4H10, is added to 225 g CCl4. Assume the components are nonvolatile and that the…
A:
Q: The reaction of the compound below with 1 equivalent of methanol yields: O 3-methylbutane-2,2-diol o…
A:
Q: O Na₂Cr₂O7 in H₂SO4 O NaOH in H₂O O NaBH4 O H₂O in pyridine O NaNO₂ in HCI NH₂
A: When aniline is treated with NaNO2 in acidic medium, diazonium salt is formed.
Q: When a metal leaf electroscope is charged by contact with a negatively charged rod: Oprotons move…
A: An atom consists of protons and neutrons present inside the nucleus and electrons revolve around…
Q: Q7. What is the mole fraction of B at equilibrium when 0.16 mole of A and 0.18 mole of B are placed…
A:
Q: Which of the following solutions is the least concentrated? A. 9.0 grams of NaCl in 100 mL of water…
A:
Q: A competitive high school swimmer takes 57.9 seconds to swim 100. yards. What is his rate in m/min?
A: Given that, length= 100 yard time=57.9 seconds then,
Q: Propose a structure for the starting material of the below reaction scheme, and answer any follow-up…
A: Unsaturated compound undergoes ozonolysis reaction in presence of ozone gas and gives carbonyl…
Q: Page 1 of 2 PRACTICE PROBLEMS 5A. For chains, draw Newman projections of all conformations around a…
A: Here we have to write Newman projection of the following given compounds.
Q: Which of these alcohols will react the fastest in hot conc. sulfuric acid ? ОН О OH ОН ОН ОН
A: Alcohol gives dehydration reactions when they react with a strong acid like H2SO4 or H3PO4 to form…
Q: This structure packages and distributes proteins throughout the cell. O golgi body Oribosomes O…
A: The correct answer is given below
Q: 9. Indicate type of intermolecular forces (London-dispersion, dipole-dipole, and/or hydrogen…
A: Here we have to predict the types of intermolecular forces of attraction present in the following…
Q: Find the difference in Second and first excited state energies 3 3-D (AE) of Particle in @1-0 2-D…
A: Answer is below:
Q: According to valence bond theory, what orbitals are overlapping to form either of the C-O sp3 on O…
A:
Q: Draw the most stable cheir conformation of the following cyclohexane.
A: Stereo chemistry is branch of chemistry in which we deal with the three dimensional arrangement of…
Q: A chromatogram from a certain column has a peak with a w1/ 2 of 12 mm and a tr of 650 mm as…
A: Given, Width of half peak height , w12= 12 mm retention time , tr = 650 mm
Q: If during a chemical reaction, carbon changes from an oxidation number of +2 to –2, what happened to…
A:
Q: Complete the following reaction schemes by choosing the correct product, starting material, and/or…
A:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- C6H5CO2CH2C6H5 can be synthesized by an SN2 reaction. Draw the structures of the alkyl chloride and nucleophile that will give this compound in highest yield. Use the wedge/hash bond tools to indicate stereochemistry where it exists. Include all valence lone pairs in your answer. Separate multiple reactants using the + sign from the drop-down menu. When a metal counterion is needed, use Na+, but draw it in its own sketcher.Draw every stereoisomer for 1‑bromo‑2‑chloro‑1,2‑difluorocyclopentane. Use wedge‑and‑dash bonds for the substituent groups, and be sure that they are drawn on the outside of the ring, adjacent to each other. Clearly show stereochemistry by drawing the wedge‑and‑dashed bonds like the example. You should replace the X with the appropriate halide.Drawing on what you know about the stereochemistry of alkene addition reactions, a. write the mechanism for the reaction of 2-butyne with one equivalent of Br2. A. predict the configuration of the product of the reaction.
- 1) What is the major product for the reaction of 3-bromo1-butene and HBr? Give the name of the product. 2) For this product, how many chiral carbon(s) are there in the structure?3) How many stereoisomers could we write for this product? 4) Write the Fischer Projections of all stereoisomers. (attach a file or other ways you could show your work)How many mono-chlorination products are possible for 3-methylpentane? Draw them out.MODIFIED TRUE OR FALSE 1. When hydrocarbons are burned, black smoke is produced, indicating incomplete combustion and a [low] C to H ratio in the sample. 2. The desired ingredient decomposes and is obtained at a temperature [higher] than its actual boiling point in steam distillation. 3. The [Baeyer's Test] couldn't differentiate 2,3-dimethylheptane and 2,3-dimethylhepta-1,5-diene .
- A chemist allows some pure (2S,3R)-3-bromo-2,3-diphenylpentane to react with a solution of sodium ethoxide(NaOCH2 CH3) in ethanol. The products are two alkenes: A (cis-trans mixture) and B, a single pure isomer. Under the same conditions, the reaction of (2S,3S)-3-bromo-2,3-diphenylpentane gives two alkenes, A (cis-trans mixture) and C. Upon catalytic hydrogenation, all three of these alkenes (A, B, and C) give 2,3-diphenylpentane. Determine the structures of A, B, and C; give equations for their formation; and explain the stereospecificity of these reactions.Name the alkene below.Use only E/Z designators to indicate stereochemistry.what are the physical porpeties of 1,3,5-cyclohexene thank you
- I put in the answer for compound A, (dimethyl‐lambda4‐sulfanylidene)cyclopropane, and the product 9‐oxadispiro[2.0.44.13]nonane, in the boxes, but it says it's incorrect. What am I not getting?Thank you for your help!Compound X is insoluble in cold KMnO4, Br2 in CCl4, and conc. H2SO4. Compound X is most likely: a. an alkane b. none of these c. an alkene d. an alcohol e. an alkyl halide Indicate which of the ff. statements regarding nucleophilicity is incorrect. F- is more nucleophilic, hence, more reactive towards methyl iodide than Cl-. Second row elements are more nucleophilic than first row elements of comparable basicity. The rate of SN2 reaction may be markedly affected by the nucleophilicity of the attacking atom. Nucleophilicity is the affinity of a nucleophile to an electrophilic carbon Which of the following alkynes can be deprotonated by NaNH2 in liquid NH3? 3-Methylhex-2-yne Pent-2-yne 3-Methylbutyne none of these Hex-3-ynePlease help with the following... Draw the bond line structures for the reactants and the products obtained in the following reactions: 1. m-chlorocumene +NaNiPr2, HNiPr2 2. 4-tertbutyl-3-methyl anisole + HNO3, H2SO4 3. o-isopropyl acetophenone + PhCH2COCl, AlCl3

