Q: Please answer this NEATLY, COMPLETELY, and CORRECTLY for an UPVOTE. Arrange the following radicals…
A: Given that : We have to arrange the following radicals in order of decreasing rate of bromination :
Q: Draw a detailed mechanism for the FeBr3@catalyzed reaction of ethylbenzene with bromine, and show…
A: The ethylbenzene on reaction with bromine in the presence of FeBr3 gives 1-Bromo-4-ethylbenzene and…
Q: Show, using the curved arrow notation, the mechanism of alcohol dehydration of 2-methylcyclohexanol…
A: Dehydration process is process where loss of water take place
Q: v.. Using curved arrow notation, propose a detailed mechanism that is plausible for the following…
A: This is an example of formation of bromonium ion followed by intramolecular SN2 reaction.
Q: Show the full curved arrow mechanism for the following reaction, clearly showing how the indicated…
A: The first step of the reaction involved the elimination of Br- (bromide) ion to generate a 2o…
Q: 2. Provide a detailed mechanism for the following reaction: Br NBS light
A: Allylic bromination of alkenes can be achieved by using N-Bromosuccinimide (NBS) in the presence of…
Q: Add curved arrows to draw the first part of the mechanism: tautomerization of a ketone to an enol.…
A: Keto-enol is a type of structural isomerism which involves a keto form (containing C=O) with alpha…
Q: Give the mechanism ? он 1. NaBHy meoH 2 H₂O
A: Note : NaBH4 is used to reduce keto to alcohol. Acid FG reacts will not react with NaBH4. ( For…
Q: Identify the conjugate base of the molecule in the box. Provide a curved arrow mechanism showing the…
A:
Q: Give a complete mechanism for the following substitution reaction. at Br ethanol
A: it is nucleophilic substitution, solvolysis reaction in which ether formation takes place.
Q: Propose a reasonable mechanism using curved arrows for the following transformation. ОН heat ОН OH
A:
Q: Draw a detailed mechanism showing the formation of all products, be sure to draw all intermediate…
A: Given reaction show mixture of ester and alkene as products. In first step of this reaction removal…
Q: Draw the curved arrow formalism showing the mechanism of the following acid-base reaction and draw…
A:
Q: Draw a detailed mechanism for the following reaction, clearly showing all electron movement NH H*,…
A: Treatment of imine with water leads to formation of ketone/aldehyde . This reaction is called…
Q: Describe the following chemical reactions as SN1, SN2, E1 and W2. Draw a curved arrow mechanism for…
A:
Q: Predict the Major Product and draw the Curved Arrow Mechanism for the following elimination…
A:
Q: Which of the following compounds (A or B) would experience a faster SN1 reaction?
A:
Q: Draw the structure of an alkyl chloride that will undergo an E2 elimination to yield only the…
A: Alkyl halide undergoes dehydrohalogenation to give the correspondence alkene. Now by adding the…
Q: Predict the mechanism of the rection below.
A: Nucleophilic substitution biomolecular reaction (SN2): The bimolecular nucleophilic substitution…
Q: Give the curved arrow mechanism for the transformation below CI H2N-NH2 NH pyridine NH
A: The answer is given as follows
Q: 1. LDA (slight excess) 2. CH3CH2I
A: LDA is a sterically hindered strong base. LDA is the best reagent to synthesize the kinetic enolate.…
Q: In both examples below the reactants shown are combined to bring about a nucleophilic substitution…
A: If weak base is used (dil.NaOH), substitution reaction takesplace Benzyl chloride undergo SN1…
Q: Add two curved arrows to the reactant side to illustrate the movement of electrons in the E2…
A:
Q: Arrange the intermediates below in order of increasing basicity:
A: The basicity is ability of species to accept proton. More the acceptance of proton, more is basic…
Q: ) Propose a detailed curved-arrow. mehunim for the following transformetion.
A: Answer
Q: 2. Draw a detailed mechanism for the following reaction.
A:
Q: Describe the following chemical reactions as SN1, SN2, E1, and E2. Draw a curved arrow mechanism for…
A:
Q: Draw a curved arrow mechanism of the following SN2' reaction. Make sure you show the intermediate…
A: In the given question we have to write following mechanism and intermediate involved in it.
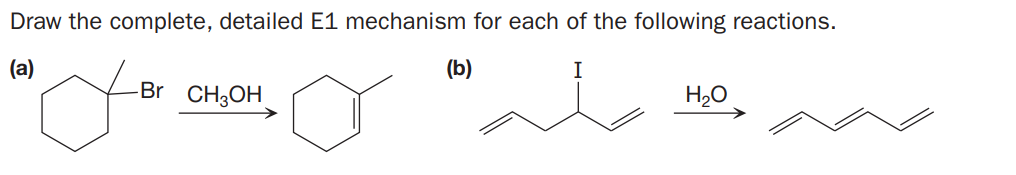
Q: Draw the complete, detailed E1 mechanism for each of the following reactions, and show all resonance…
A: E1 mechanism follows through two steps 1) In the first step by eliminating leaving group (halide…
Q: Which one favors E2 reaction? A) isopropoxide ion B) tert-butoxide ion C) ethoxide ion D)…
A:
Q: 5) Provide a complete and detailed mechanism for these transformations. OH heat, acid +
A: We have to predict the mechanism for given reaction.
Q: H₂O TfOH
A: Solution: Alkynes can be hydrated to form enols that immediately tautomerize into ketones. The…
Q: i. O3, ii. PPh3 CH2CI2 H
A: The mechanism is as follows:
Q: 3/5 5.1 each indicated reaction. Mechanisms. Using curved arrows, suggest a reasonable mechanism for…
A: The reaction of alkene with phosphoric acid leads to the formation of carbocation. The oxygen of -…
Q: nechanism for the following transformati Br2 + HBr Br CH2OH
A: The given organic reaction is the reaction between alkene and bromine followed by ethanol, results…
Q: Explain why one of the following molecules will undergo an E1 elimination faster:
A: In the E1 reaction the elimination of leaving group first and then deprotonation takes place finally…
Q: NAOH NH2 + NH, H2O
A: See mechanism below.
Q: Mechanism- Identify compound A and propose a curved arrow mechanism for the second step
A: 1. Beta- napthol acts as like acid , when it is treated with base like NaOH a salt is formed , it…
Q: Please provide a complete mechanism to account for the transformation shown below. Мео OMe Br Meo…
A:
Q: 7. Propose an efficient synthesis of for each of the following transformation: a) b)
A:
Q: Predict the most likely sites of electrophilic aromatic substitution in each of the following…
A: Predict the most likely sites of electrophilic aromatic substitution in the above molecules are…
Q: NANH, OTs NH3
A: The question is based on the concept of reaction mechanism. We have to identify what type of…
Q: Draw the mechanism of the following reaction taking into account the stereochemistry Cl2 H20
A: The mechanism can be given as follows: Step 1: The attack of the alkene on the chlorine is shown…
Q: Provide the complete mechanism using Curved Arrow Formalism for the reaction of…
A: Please find the attachment.
Q: Use curved arrows to show the movement of electrons in the attached E2 mechanism. Draw the structure…
A: E2 mechanism:- E2 refers to the elimination reaction which having bimolecular rate of reaction. In…
Q: Draw this compound in a chair form so that an E2 reaction is possible. Then, draw the mechanism and…
A: E2 reaction is possible product can be drawn as,
Q: Draw a detailed reaction mechanism for the following transformation: NaOH To obtain the complete…
A: In an organic compound when two aldehyde group is present in the same molecule, they condense and…
Q: please help with this question. thank you. Propose a mechanism for this transformation
A: We have given the organic reaction we have to find the major product of reaction.
Q: Provide an arrow pushing mechanism for the following transformation. Draw out a representation of…
A:
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

- Provide an arrow pushing mechanism for the following transformation and clearly circle the slowest (rate-determining) step in the mechanism.Draw a complete mechanism, using arrow convention to indicate electron movement, for the following transformation:Please provide a detailed mechanism (with arrows) for this reaction.
- Draw a complete curved-arrow mechanism for this reaction and predict the product.Draw the complete, detailed E1 mechanism for each of the following reactions, and show all resonance structures, where applicable.Draw a detailed mechanism for the FeBr3@catalyzed reaction of ethylbenzene with bromine, and show why the sigma complex (and the transition state leading to it) is lower in energy for substitution at the ortho and para positions than it is for substitution at the meta position.
- need a clear answer please make it straightforward!!! In the space provided below please give the detailed mechanism for the following transformation. Please be certain to use good curved arrow notation for your mechanism,Draw a detailed step-wise mechanism for the following reactions. Be sure to show all steps, formal charges, and show the movement of electrons with curved arrows.Draw a detailed arrow pushing mechanism for the following reaction. In the rate-determining step unimolecular (SN1) or bimolecular (SN2)
- Draw the product of this reaction and states its IUPAC name, and states what type of mechanism is occuring (eg. SN1, SN2, E1, E2, etc)?Draw the curly arrow mechanism for this reaction showing step by step. Q. Draw two alternative isomers of nitroacetanilide and suggest two reasons why, during this reaction, the nitro group is preferentially located in the para position....Give a clear handwritten mechanism and product for given below Sn2 reaction..a reaction shows all bonds ,lone pairs and arrows...?

