Q: Draw the products from the following transformations. Your file should have your name clearly…
A: Given are organic synthesis reactions
Q: A Substance that is responsible for UV-vis absorption is? A. Chromophores B. Overtones C. Photons D.…
A: A Substance that is responsible for UV-vis absorption is?
Q: Question 18 For the following endothermic reaction: X(g) + Y(s) = 2Z(g). Circle which direction the…
A: According to Lechatellier principle in a equllibrium system if you change temperature, pressure ,…
Q: 3:18 For each of the following, carry out the mathematical operation and report answers in…
A: Answer can be written in lowest significant digit present in the question.
Q: 5. Which of the following compounds displays besides other signals - a nonet (a signal that is split…
A:
Q: Calculate the vapor pressure (in mmHg) of a solution made by dissolving 69.9 g of organic compound A…
A: Given :- the vapor pressure (in mmHg) of a solution made by dissolving 69.9 g of organic compound A…
Q: Calculate the mass % of a solution of KCl prepared by dissolving 12.75 g KCl in 54 g H2O?
A: Given : Mass of water = 54 g Mass of KCl = 12.75 g To calculate : mass % of solution
Q: Which terms describe the relationship between the following molecules? "CI CI I. Enantiomers…
A: In this question we have to tell the relationship between the molecules.
Q: 2NaOH + H₂SO4 → 2H₂O + Na2SO4 AH = -112 kJ 50.0 mL of 0.50 M H2SO4 is added to 60.0 mL of 0.80 M…
A:
Q: Very different concentrations of Na+ ions exist in red blood cells (12 mM) and the blood plasma (165…
A: Given: Concentrations of Na+ ions exist in red blood cells =12 mM Concentrations of Na+ ions exist…
Q: •Br 1er : Bri 1 : Br :: G- 1 Br: 6. square planar 7. square pyramidal trigonal planar 9. trigonal…
A: Molecular geometry takes into account only the bond pairs around the central atom.
Q: The pH at the equivalence point of a strong acid strong base titration is.... Select one: a. equal…
A: ->If solution is acidic then pH<7. ->If Solution is basic then pH>7. ->If Solution is…
Q: 1. Calculate the cell potential, AG (J), and the equilibrium constant Keq for the following…
A: Calculation of Cell potential of the cell
Q: What would be the pH of 0.00551 M H2SO4 solution?
A:
Q: The following plot shows a titration curve for the titration of 1.00 L of 1.00 M diprotic acid H 2A…
A: Answer: Buffer is a type of solution that resists the change in its pH on adding small quantity of…
Q: Suggest a synthesis for the following compound from benzene
A: Given is organic molecule.
Q: The combustion of how many moles of ethane (C2H6) would be required to heat 561 g of water from…
A:
Q: -4 For a certain chemical reaction, the equilibrium constant K = 1.9 × 10 at 5.00 °C. Calculate the…
A: Given that, K=1.9×10-4 T=5.00℃ = (5.00+273) K = 278 K ∆G°= ?
Q: How many hydrogen atoms are in the molecule below?
A:
Q: When the following equation is balanced properly under acidic conditions, what are the coefficients…
A:
Q: Explain why p-nitrophenol (the left molecule) is more acidic than p-methylphenol (the right…
A: Why p-nitrophenol is more acidic than p-methylphenol ?
Q: Show all the mechanistic steps in the following assignment. 1) Roll two die to determine the number…
A: In this question we will take a primary bromoalkane and react with Mg/dry ether followed by CO2 to…
Q: What product would be formed in the following Claisen condensation reaction? base ? CH₂O OCH 3 H H
A: Claisen Condensation Reaction :
Q: 20) Which of the following reagents would be suitable for making "ethyl-benzene" from "acetophenone"…
A: We are authorised to solve only one question at a time. Please post rest of the questions…
Q: Avogadro's Number: 6.022 x 1023 M = mol/L Calculate the molarity of 2.50 grams of Compound XX (molar…
A:
Q: Complete and balance the following half-reaction in acidic solution. Be sure to include the proper…
A: Complete and balance the following half-reaction in acidic solution---
Q: love. How many molecules of CO₂, FADth and NADH are respectively made por each of…
A: Q1)A Q2 ) C
Q: A sample of a gas in an 11.2L11.2L container is at 1.0atm1.0atm and 0°C0°C, as shown. The mass of…
A: Given : Volume = 11.2 L Pressure = 1 atm Temperature = 0°C = 273 K Mass of sample = 16 g
Q: Your picture has labels in red above specific bonds. Choose the answer that places those bonds in…
A: In this question, we have to choose the option in which bond length is arranged in decreasing order…
Q: Match the molecule with appropriate molecular geometry H20 ✓[Choose] bent trigonal planar trigonal…
A: Determine molecular geometry of the given compound ----
Q: Limitations/obstacles of green chemistry
A:
Q: ve the major products for each of the following reactions OH PCC CH₂Cl2 CrO3, H₂SO4 1. LiAlH4, THF…
A:
Q: 3. A solution has [Pb2+] = 0.0012 M and [CI] = 0.010 M. Will PbCl2 precipitate form? Ksp for PbCl₂ =…
A:
Q: A student made measurements on some electrochemical cells and calculated three quantities: • The…
A:
Q: A chemist fills a reaction vessel with 0.999 g calcium phosphate (Ca3(PO4)2) solid, 0.689 M calcium…
A:
Q: 5 please
A: You made acetaminophen analogue using 3-amino phenol and acetyl chloride (see above). You mistakenly…
Q: 4.00 mL of H2O, 3.25 mL of an aqueous 0.030 M SCN- solution, and 11.00 mL of an aqueous 0.020 M Fe3+…
A: Given data,Molarity of SCN-=0.030 MVolume of SCN-=3.25 mL=0.00325 LMolarity of Fe3+=0.020 MVolume of…
Q: arrange the following ionic compounds in order of decreasing amount 10 INT
A: Arrange the following ionic compounds in order of decreasing amount of energy released in lattice…
Q: The heat of fusion AH, of water (H₂O) is 6.0 kJ/mol. Calculate the change in entropy AS when 2.4 g…
A:
Q: Using the thermodynamic information in the ALEKS Data tab, calculate the standard reaction entropy…
A: Calculate standard entropy change of the given reaction--
Q: 5. Do the following synthesis problems. You may use any reagents necessary as long as they are real…
A:
Q: How many stereoisomers does the following compound have? CH3CHCICH₂CHCICH 3 Select one: OA. O OB. 1…
A:
Q: What would be the major product formed from the reaction of 1-phenyl-2- butene with NBS?
A: In this question we have to tell the major product of the reaction.
Q: Calculate the standard Gibbs energy in kJ/mol change for the following voltaic cell A(s) | A3+(aq)…
A:
Q: If the following Aspartame was hydrolyzed completely what will it be broken down into? Show ALL…
A: We have to give the hydrolysis products of the aspartame.
Q: What is the pH of a solution made by mixing 30.00 mL of 0.10 M HI with 40.00 mL of 0.10 M NaOH?…
A:
Q: Determine the osmotic pressure in atm at 45.8 °C of an aqueous solution that is 0.502 M NaNO3. (R =…
A: Given :- concentration of NaNO3 solution = 0.502 M R = 0.0821 atm.L / K.mol T = 45.8°C To…
Q: consider the titration of 100.0ml of 0.200M HONH2 by 0.100M HCl (Kb for HONH2 = 1.1x10^-8) a.…
A: NH2OH is a weak base with Kb = 1.1*10-8 Given the initial concentration of NH2OH = 0.200 M
Q: oh. What is this formula molecular structure, number of carbon sites how many hydrogen sites…
A: Please open the attachment.
Q: Provide the structure of the product(s) with stereochemisty if appropriate. If multiple products…
A:
Give a clear handwritten answer with explanation
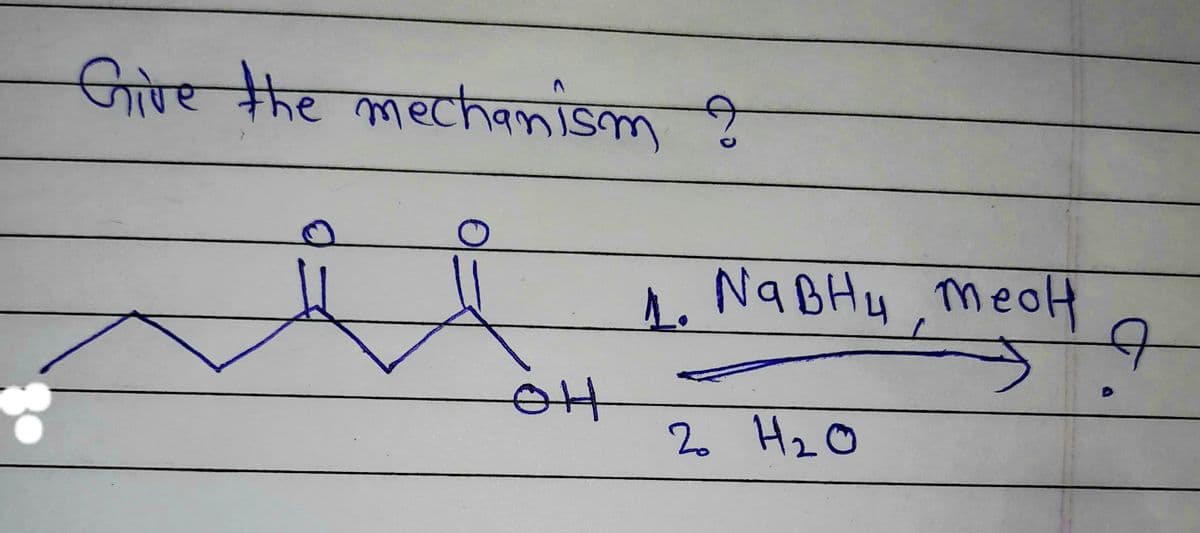
Step by step
Solved in 2 steps with 1 images

- Don't provied handwriting solution. 0Can you please explain the reasonin behind why we split the molecule the way we do to obtain the desired product in addition to showing the mechanism? Thank you!In the following enantioselective reaction find the product. A positive non-linear effect (NOE) was observed, Explain with reaction pathway.Can show the mechanism for this with explaination and say what's the role of n-Bu4NF including AcOH

