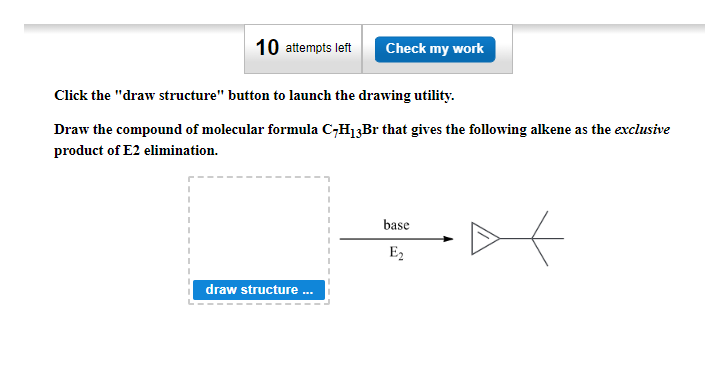
Draw the compound of molecular formula C-H₁3Br that gives the following alkene as the exclusive product of E2 elimination. draw structure base E₂
Q: Please explain: When experienting, what is the purpose of adding hydroxylamine hydrochloride with…
A: The combination of hydroxylamine hydrochloride and iron-phenanthroline is often used in analytical…
Q: Octane, C8H18, is a hydrocarbon that is present in gasoline. It is produced using the following…
A: l
Q: Read pages 181-183 and 188-191 in Zubrick. Suppose you wanted the Rf of your benzoic acid to be ~0.5…
A: We must obtain the benzoic acid's Rf as 0.5 while performing TLC. We must suggest whether ethyl…
Q: Dehydrohalogenation of 2-bromo-2,3-dimethylbutane with a strong base can lead to multiple products…
A: Zaitsev rule: The rule states that the more substituted alkene is the major product in an…
Q: interpret/identify the peaks and their characteristics. (ex. C=O, 4H, doublet) compound name:…
A: Molecular formula = C7H14O2 HDI =(2C+2+N-H-X)2HDI = 2x7+2+0-14-02=1 From HDI =1 given compound may…
Q: At 25 °C, the following concentrations were found for the gases in an equilibrium mixture for the…
A: Given that, an equilibrium reaction is N2Og + NO2g ⇌ 3NOg. The equilibrium concentrations are [NO2]…
Q: Example #1 BaCO3 Write the Ksp equation AND the chemical equation for each of the following Ksp =…
A: Solubility product constant (Ksp): When a slightly soluble ionic compound is mixed with water,…
Q: Predict the reactants of this chemical reaction. That is, fill in the left side of the chemical…
A: A balanced chemical equation is an equation which contains same elements in same number on both the…
Q: Question 12 What is the number corresponding to the stationary state located between n = 3 and n =…
A: Since, For representation of electron in an orbital we use four type of quantum number-- 1)Principle…
Q: [Review Topics] [References] Use the References to access important values if needed for this…
A:
Q: Give detailed Solution with explanation needed
A:
Q: H₂(9)+Cl₂(9) 2 HCl(9) 4 At the temperature the engineer picks, the equilibrium constant K, for this…
A: Answer: Relation between Reaction quotient QP and equilibrium constant KP is shown below: 1. When…
Q: What is regioselectivity due to? a) On the stability differences of carbocations b) On the…
A: This question belongs to Stereochemistry.
Q: A good recrystallization solvent is one in which the crystals don't dissolve when the solvent is…
A: Recrystallization is a purification technique used to obtain pure crystals from a mixture of solids,…
Q: Upon reaction of 2.548 g of copper sulfate with 3.094 g of iron metal, 0.784 g copper metal was…
A:
Q: Identify whether each species functions as a Brønsted- Lowry acid or a Brønsted-Lowry base in this…
A: We have to identify the Bronsted Lowry acid and base in the given reaction
Q: How many electrons are exchanged in the completed reaction Ba (s) + Al(NO3)3(aq)
A: Oxidation reduction reaction (Redox reaction) It can be defined as the chemical reaction in which…
Q: For the reaction: 2 SO2 (g) + O₂(g) → 2 SO 3 (g) Kp = 6.2 x 10⁹. What is the position of the…
A:
Q: Which of the following are ionic bonds, polar covalent bonds, and nonpolar bonds? A) I - I B) C - O…
A: Given are some molecules, we have to tell which one is ionic or covalent. As per guidelines, only…
Q: 19. Which of the following sets of quantum numbers has a mistake? n = 1 1 = 0 m₁ = 0 ms = 1/2 n = 3…
A:
Q: Calculate the Gibbs free energy of the reaction from mixing 50 mL each of 0.50 M Ag*(aq) solution…
A: The spontaneity of any reaction can be determined by ∆G value and it's sign.
Q: B. Determine the pressure, volume, or temperature of a gas under a certain set of changing…
A: Given, initial volume = 150 mL initial pressure = 760 torr final pressure = 740 torr to…
Q: Studying the reaction A + B → Products, you gather the following information: [A] 0.10 0.30 0.40 [B]…
A:
Q: 5. All of the following word equations represent double displacement reactions. Complete the word…
A: Double displacement reactions are those in which a part of one reactant is displaced by other…
Q: Calculate the volume in liters of a 1.87 × 105 mol/L zinc oxalate solution that contains 700. mg of…
A:
Q: Use the solubility table to predict how you would test for the following ions in solution. Write a…
A: The question is based on qualitative analysis. we need to write the proper tests for the presence of…
Q: 9.91 g of MgSO₄ is placed into 100.0 mL of water. The water's temperature increases by 6.70 °C.…
A: Please find your solution below : Enthalpy of dissolution is defined as the change in enthalpy…
Q: The overall reaction in a commercial heat pack can be represented as 4Fe(s) +30₂(g) →2Fe₂O3(s) AH =…
A: The overall reaction in a commercial heat pack is: 4Fe(s)+3O2(g)→2Fe2O3(s) , ∆H=-1652 kJ From the…
Q: Consider the equilibrium 2NaHSO3(s) Na₂SO3(s) + H₂O(g) + SO₂(g) How will the position of equilibrium…
A: Le Chateliers principles states that, when the dynamic equilibrium (Pressure, concentration etc) is…
Q: Draw both (i) A and (ii) A isomers of the [Ru(en)3]2+.
A: Chiral octahedral complexes chelating ligands can have ∆ and ∧ configurations. delta(∆) is used to…
Q: Will the following reaction proceed as written? Select an answer and submit. For keyboard…
A: To draw the product of the given organic reaction.
Q: The chemical formulae of some acids are listed in the first column of the table below, and in the…
A: we have to identify the major species present when the given acids are dissolved in water
Q: How many joules of heat does it take to melt 128 grams of ice that has an initial temperature of -18…
A: To solve this problem, I used the concepts of specific heat, heat of fusion, and the calculation of…
Q: Calculate the pH of a buffer solution containing 0.95 M NaF and 0.78 M HF. The pKa of HF is 3.14.
A:
Q: A 8.00 mL ampule of a 0.150 M solution of naphthalene in hexane is excited with a flash of light.…
A: we have to calculate the percentage of naphthalene molecules that emit photon
Q: Determine if the following salt is neutral, acidic or basic. If acidic or basic, write the…
A: Salts of strong acids and strong bases are neutral in nature. Salts of strong acids and weak bases…
Q: What is the energy change when the temperature of 14.0 grams of gaseous helium is decreased from…
A:
Q: Dimensional analysis & conversions. If you need scientific notation in you answer, copy this for the…
A: Given, mass of Sn(OH)2 = 10.44 g mass of tin (Sn) = ? Note: Molar mass of Sn(OH)2 = 152.73 g/mol…
Q: Question 4 Calculate the frequency of the light emitted by a hydrogen atom during a transition of…
A: Please find your solution below : Planck's equation is an expression used to calculate energy of a…
Q: The image below represents a gaseous equilibrium system for two reactive gases, R and P. The spheres…
A: Given that, an equilibrium reaction system of R → P is shown below Also, given that, the enthalpy…
Q: any help? ( Please type answer note write by hend )
A: Solution- The partition function for a two-state system is given by: q=e-E1kT+e-E2kT where E1 and E2…
Q: 2. Use the activity series to predict if the following reactions will occur. If not, write NR. If…
A: ♦ The metal activity series is used to predict whether a metal displacement reaction will occur or…
Q: If an electron is in a d subshell, that means / = to Drag and drop your selection from the following…
A:
Q: nemist adds 240.0 mL of a 6.5M silver nitrate (AgNO3) solution to a reaction flask. Calculate the…
A: Molarity is defined as number of mole of substance divide by volume of solution in liter
Q: The rate constant k for a certain reaction is measured at two different temperatures: temperature k…
A: Activation energy is the difference in energy content between atoms in an activated or…
Q: The pH of a 0.013M perchloric acid is:
A:
Q: Using the concentrations found in the experiment for the iron cation and thiocyanate anion determine…
A: A = εlc where A is the absorbance, ε is the molar absorptivity (in L/mol cm), l is the path length…
Q: what is the size of bond angle H-C-C in cis-1,2-dichloroethene and what is the size of Cl-C-C?…
A: In cis-1,2-dichloroethene, the H-C-C bond angle is roughly 117 degrees. In cis-1,2-dichloroethene,…
Q: A 750 mL aqueous solution containing 6.31 g of nitrous acid, HNO2 (47.0135), is about 58.7 %…
A:
Q: H 100 Br H ** CH3 NH₂
A: Given that, a synthesis scheme is shown below We have to carry out the above synthesis in two…
Step by step
Solved in 3 steps with 2 images

- 1. Which of the following is the strongest nucleophile? Refer to the diagram below with an MeOH, MeSH, etc. 2. Which of the following is true about the stereochemistry of SN1 reaction?a.retention of configuration at the electrophilic centerb.50:50 mixture of retention and inversion of configuration at the electrophilic centerc.inversion of configuration at the electrophilic centerd. slightly more inversion than retention at the electrophilic center1. Draw a reasonable arrow-pushing mechanism for the transformation shown along. 2. Identify nucleophiles and electrophiles 3. Name any type of reactions taking place like E2 or E1 4. Account for any regio- or stereoselectivitySupply the missing reagent in the following reaction: (see attachment) A. Lindlar, H2, mCPBA B. O3 or KMnO4 C. fused KOH D. NH3 E. NaNH2
- Based on your answer to parts A - C, draw the correct regio- and stereoisomer of the product in the space provided. Provide a systematic name of the substrate. Provide a systematic name of the product (see section 8.3 for alkene nomenclature). Draw a transition state structure of the reaction with partial bonds and partial charges indicated. Draw an energy diagram for the E2 reaction on the previous page. Your reaction coordinate diagram should: Include structures of starting materials, products, and transition states at correct relative energies. Indicate the activation energy of the rate determining step and whether the reaction is endothermic or exothermic.For which reaction mechanisms—SN1, SN2, E1, or E2—of thefollowing statement true? A statement may be true for one or moremechanisms. Tertiary (3°) alkyl halides react faster than 2° or 1° alkyl halides.For the following starting material A, draw the products B and C that you expect to form under the following reaction conditions.
- which newman projection displays the proper antiperiplanar orientation for an E2 elimination of the reaction sequence belowOf the two molecules below select which molecule would be predicted to react faster via an E2 elimination. (hint: stereoelectronic arguments can help)Draw the major organic product of this E1 elimination reaction. Ignore byproducts.
- With the choice of thumbnails labeled A to P below, construct a comic strip depicting the complete mechanism of cyclopentane monobromination. List all the steps and in the correct order. Enter your answer as follows: ABCDEF (sequence of capital letters without spaces). N.B. There are more thumbnails than necessary.Please help with the following ochem mechanisms.... 1. Provide the stepwise mechanisms for the following reactions (see attached picture)For the dehydrohalogenation (E2) reaction, draw the Zaitsev product, showing the stereochemistry clearly. You might find it helpful to make a model of the starting material to determine the correct conformation.

