Draw the conformation for methoxycyclohexane with the methoxy group axial and the conformation with the methoxy group equatorial, include all hydrogen atoms on cyclohexane and label each as axial or equatorial. Using the values from Table 3.2, which conformation is more stable and by how much energy? Calculate the Keq for the equilibrium between axial and equatorial cyclohexane at 298 K. Calculate the percentage of each conformation present at this temperature.
Draw the conformation for methoxycyclohexane with the methoxy group axial and the conformation with the methoxy group equatorial, include all hydrogen atoms on cyclohexane and label each as axial or equatorial. Using the values from Table 3.2, which conformation is more stable and by how much energy? Calculate the Keq for the equilibrium between axial and equatorial cyclohexane at 298 K. Calculate the percentage of each conformation present at this temperature.
Organic Chemistry
8th Edition
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Chapter2: Alkanes And Cycloalkanes
Section2.5: Conformations Of Alkanes And Cycloalkanes
Problem 2.9P: Following is a chair conformation of cyclohexane with the carbon atoms numbered 1 through 6. (a)...
Related questions
Question
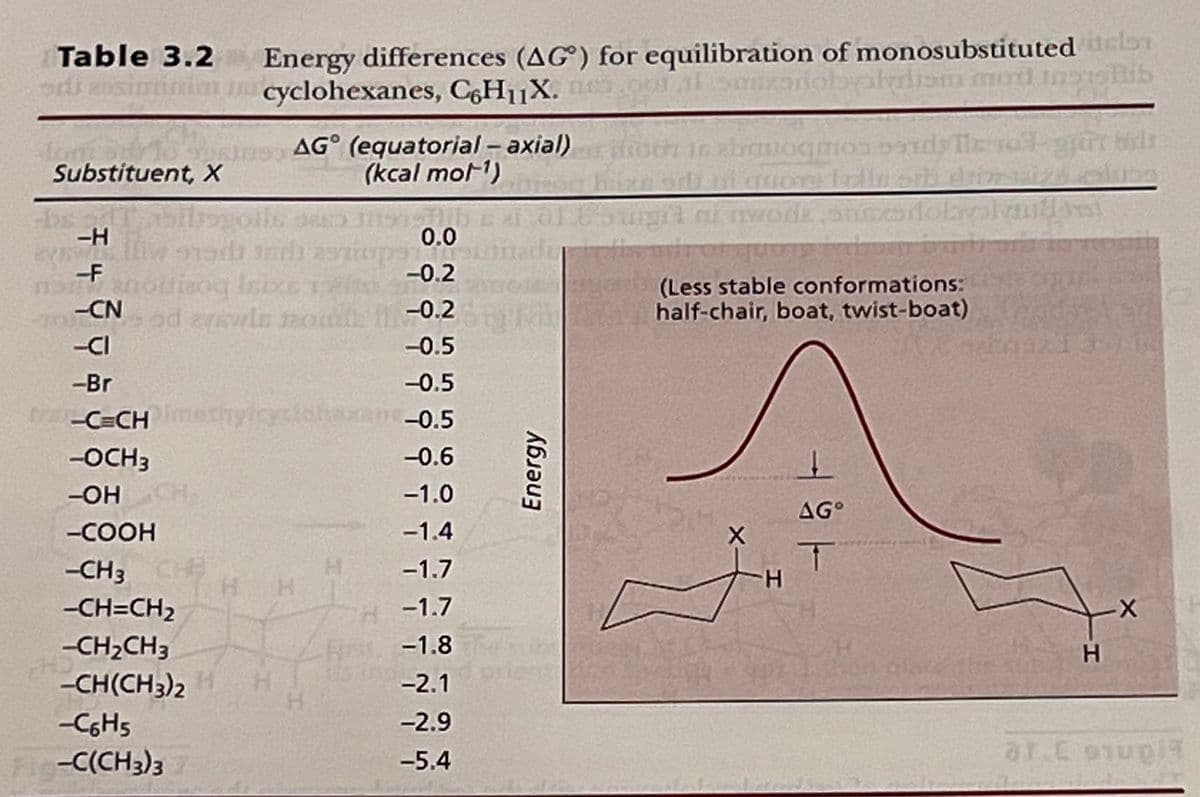
Transcribed Image Text:Table 3.2
odi ensiminimi
CVEWLE
-F
Substituent, X
bx stl
-H lliw sadi sah asupsto
toutieng Isixs
-0.2
od 24swin modis -0.2
-0.5
-0.5
11
-CN
-CI
-Br
1015
Energy differences (AG) for equilibration of monosubstituted cl
cyclohexanes, C6H₁X
zadobadiam mord inprobib
a
abruogmos.sondy the 101-gur sel
-OCH3
-OH
-COOH
-CH3 CH#
-CH=CH₂
-CH₂CH3
-CH(CH3)2
AG (equatorial-axial) moc
(kcal mol-1)
-C6H5
-C(CH3)3
-C=CHDimethylcycloban-0.5
goils, bars in caf
H H
-0.6
-1.0
-1.4
-1.7
-1.7
-1.8 T
-2.1
-2.9
-5.4
Energy
vode
Joy wibor b
folavalvulad
(Less stable conformations:
half-chair, boat, twist-boat)
H
+
AGⓇ
314
H
-X
at E sugia

Transcribed Image Text:Draw the conformation for methoxycyclohexane with the methoxy group axial and the
conformation with the methoxy group equatorial, include all hydrogen atoms on cyclohexane
and label each as axial or equatorial. Using the values from Table 3.2, which conformation is
more stable and by how much energy? Calculate the Keq for the equilibrium between axial
and equatorial cyclohexane at 298 K. Calculate the percentage of each conformation present
at this temperature.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 4 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning