Q: An empty steel container is filled with 0.0410 atm of HF. The system is allowed to reach…
A:
Q: 10-³ M NaCHO₂ (sodium formate) added to water
A:
Q: 4. Write a balanced nuclear equation for each of the following: a) b) 20 Tl(d,p) 216 85 At(d, a) c)…
A: #4: We are given the shorthand notation of nuclear reactions and we have to write the complete…
Q: How many signals would be present in the ¹HNMR? O 4 02
A: 1HNMR is also known as proton NMR. NMR is nuclear magnetic resonance. It is a technique that is use…
Q: 7. Which of the following choices is a diastereomer of the first structure shown. OH OH C. OH OH B.…
A: Since you asked multiple questions so as per Q&A guidelines of portal I solved first question…
Q: A Chemistry student at Agora is calculating the average atomic mass of Potassium using the following…
A:
Q: 2. Select all of the answers that describe an unstable nucleus: (this is a multiple select question,…
A: Unstable nucleus is define as .
Q: ound on pg.805 to Identify the products produced at the anode and cathode for the Electrolysis of…
A: Here we to predict the products obtained at respective electrodes for the electrolysis of KI in…
Q: 1,1-dimethylcyclopentane (draw condensed structural diagram)
A: Condensed structure: It show the order of atoms like a structural formula which is written in a…
Q: a) the following compour d) CH3 CHCHCH₂CH3 OH Br CH₂CH₂CH3 -OH b) CH3C(CH3)2CH(OH)CH3 C) e) OH OH
A: we have to write the IUPAC names of the structures given
Q: e e following compounds is most basic? se I II III IV O
A: The base which form a stable conjugate acid that is more basic . -> conjugate acid is formed when…
Q: ,Chlorine gas can be prepared in the laboratory by the reaction of hydrochloric acid with manganese…
A:
Q: What is the major organic product obtained from the following reaction? H a. butyrophenone,…
A:
Q: The following two drawings are resonance structures of one compound [K But the following two…
A: Delocalisation of pi electrons leads to resonance structures.
Q: The air pressure in the tires of an automobile is adjusted to 36.0 psi at a gas station in San…
A: Initial pressure - 36.0 psi Initial temperature - 68 oF Final temperature - 172 oF
Q: IIA. Balance the following chemical reactions (1-10) IIB Identify the type of chemical reaction…
A: According to law of mass conservation we know that in a chemical reaction mass is neither created…
Q: In the circled atom in this structure. CH3 A) four sigma bonds B) three sigma bonds, one pi bond and…
A:
Q: 1. The cathode in a galvanic and electrolytic cells is: negative in certain type of cells the site…
A:
Q: Consider the reaction: CaCl₂(aq) + 2AgNO₃(aq) → Ca(NO₃)₂(aq) + 2AgCl(s) How many grams of CaCl₂…
A:
Q: Which of these would have a double covalent bond? O C3H8 O C₂H4 O CH4 O C₂H₂ O C₂H6
A:
Q: An IR spectrum of p-cresol is acquired. Select the region of the spectrum where the C-O stretching…
A: P cresol is also caled as 4-methyl phenol.
Q: co₂²- ppt or NR? Description CI™ ppt or NR? Description OH ppt or NR? Description NO₂ ppt or NR?…
A: The formation of a precipitate is an indication of chemical reaction called a precipitation…
Q: Determine pCl and pAg if 20 mL of a 0.1 M AgNO3 solution is titrated with 6 mL of 0.2 M HCl.…
A: The reaction taking place is - AgNO3(aq)+ HCl (aq)→AgCl (s)+ HNO3 (Aq) The net reaction is-…
Q: Use the table to determine which of the following species will best convert Cd +² to Cdº Fe+3 Pb+2…
A: Cd2+ to Cd0 The species which is best reducing agent that reduce Cd2+ to Cd . Best reducing agent…
Q: Draw the skeletal line bond structure of 1-methyl-4-nitrobenzene
A:
Q: What mass is needed to obtain 4 mmol of sodium borohydride?
A: Mole is equal to weight of substance divide by it's molar mass. Hence.…
Q: Write the structural formula for each of the following compounds 2,2-dimethyl-1-butanol a) b)…
A:
Q: Curved arrows are used to illustrate the flow of electrons. Follow the curved arrows and draw the…
A: Given : structure of reactant.
Q: Determine the radius "r" of an impurity atom that will just fit into a tetrahedral site of BCC iron…
A: Given, Iron has BCC crystal structure
Q: 3. Drawn below is the structure of Crestor® (rosuvastatin), a medication used to reduce cholesterol.…
A:
Q: Determine the type of each chemical equation describing a precipitation reaction. - CaBr2 (aq) +…
A: Precipitation reaction : It is a type of chemical reaction ,where two ionic compounds dissociates in…
Q: 20 ml of 1M NaOH solution and 20 ml of 1M HCl solution both at room temperature were made to react…
A: This problem is based on heat of neutralization of acids and bases. We know that when 1gram…
Q: 1. Predict the major product or products of the following reactions: H+ CH3OH Me Et H Me
A:
Q: 7. A group of Colby students discovered a hydrolase that cleaves cellulose. After purifying the…
A:
Q: The atomic number of an element changes for all of the following radiation processes except:…
A: Radioactive decay: Radioactive decay is the emission of energy in the form of ionizing radiation...
Q: Explain how the molar volume of a gas would change as the temperature increases.
A: Please find your solution below : Molar volume of a gas is the volume occupied by one mole of gas at…
Q: What statements are always true about limiting reactants? Select one or more: a. There will be an…
A: Limiting reactants : It is the reactant in the chemical reaction which is completely consumed for…
Q: 30.0 mL of 0.200M H2SO3, a diprotic acid is titrated with 0.100M NaOH. a) Calculate the volume…
A: The reaction taking place between H2SO3 and NaOH is- H2SO3 + NaOH→NaHSO3 + H2ONaHSO3 + NaOH→Na2SO3 +…
Q: An atom with 15 protons and 18 neutrons would be an isotope of: arsenic Ophosphorus argon sulfur
A: Phosphorus ---> option(b)
Q: ? MeOOC
A:
Q: Using the Bohr model, determine the wavelength when an electron in n = 3 is excited to n = 7.
A:
Q: Exercise : Consider the following potentiometric titration data for acetic acid solution against…
A: a)
Q: Be sure your chemical equation is balanced! ZnS(s) +O2(g) ->
A: We have to write products of the reaction ZnS(s) +O2(g) ->
Q: 1) LiAlH, in Et₂O 2) H₂O*
A:
Q: please answer this spec question! answer choices are also given, except tetrahydrofuran and toluene…
A: Infrared spectroscopy deals with the study of interaction of compounds/ organic molecules with…
Q: The pH of a basic solution is 12.99. What is [H⁺]?
A: The value of [H+] is = 1.023 x 10-13 M
Q: 2. The following questions are designed to help acquaint you with identifying and drawing…
A: #(a): Enantiomers are non superimposable mirror images that can be drawn by plotting an exact…
Q: A gas expands and does work on the surroundings equal to 0.50 BTU. At the same time, it absorbs 0.21…
A:
Q: 1. A gas expands from 250 to 1000. mL at constant temperature. Calculate the worl done in joules…
A:
Q: CH3 A) ion-ion B) dipole-dipole C) hydrogen bonding D) dipole-induced dipole E) London dispersion…
A: The force of attractions between two molecules are called inter molecular attractions.
See image below
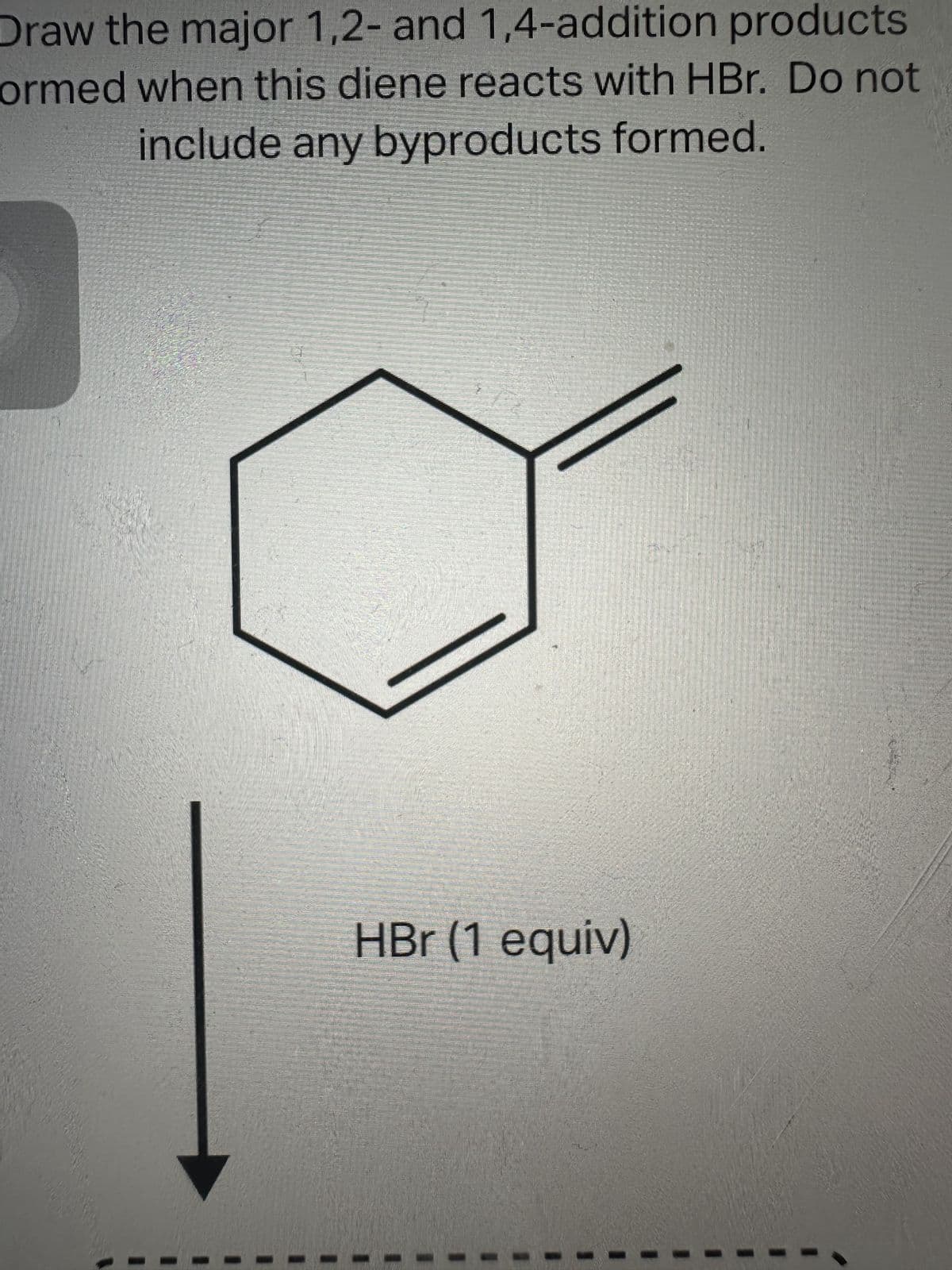
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- Which alkyl bromide(s) can form the alkene under E2 elimination conditions.Elimination occurs when (Z)-3-bromohex-3-ene is treated with NaNH2. Under the same conditions, 1-bromocyclohexeneundergoes elimination much more sluggishly. Explain why(1) Which is an isolated diene? (2) Which is an alkene with possible rearrangement product (1,2-H shift) with reaction to HI? (3) Whixh isa diene capable of having 1,2 and 1,4-addition products? (4) Which is a conjugated diene?
- 1. Which among these would be the most stable carbocation?a. Allylicb. Vinylicc. Primaryd. Secondary2. What is the primary reason for the stability of tertiary carbocations?a. Free rotationb. Resonancec. HyperconjugationArrange the alkyl halides in order of increasing reactivity in an SN2 reaction with KI in acetone (least first). I, IV, III, II II, III, I, IV IV, I, III, II III, II, IV, IThere are two isomeric cyclohexa-1,4-diene products when toluene undergoes the Birch reduction (see Problem 25.24). (a) Draw the mechanism that leads to the formation of the major product. (b) Will the Birchreduction of toluene occur faster or slower than the Birch reduction of benzene itself? Hint: Is –CH3 an electron-donating or an electron-withdrawing group?
- Please identify priorities of functional groups and name following molecules based on thier stereochemistry as R or SA8 Is there anyone who can provide this pericyclic rxn mechanism?When Br2 is added to buta-1,3-diene at -15 °C, the product mixture contains 60% ofproduct A and 40% of product B. When the same reaction takes place at 60 °C, theproduct ratio is 10% A and 90% B.Show why A predominates at -15 °C and B predominates at 60 °C.
- a) What product is formed from the [1,7] sigmatropic rearrangement of a deuterium in the following triene? (b) Does this reaction proceed in a suprafacial or antarafacial manner under thermal conditions? (c) Does this reaction proceed in a suprafacial or antarafacial manner under photochemical conditions?When Br2 is added to buta-1,3-diene at -15 °C, the product mixture contains 60% ofproduct A and 40% of product B. When the same reaction takes place at 60 °C, theproduct ratio is 10% A and 90% B.) Propose a mechanism to account for formation of both A and B[VI9] Instructions: Provide the missing reagent/s, substrate/s, or product/s for each reaction. (refer to the photo below)

