Q: At 400 K oxalic acid decomposes according to the reaction: H_C,O,(9) CO.:(9)+HCOOH(g) In three…
A: The given chemical reaction is H2C2O4(g) →CO2(g) + HCOOH(g)
Q: Pt(s) | V³+ (aq, 0.210 M), V²+ (aq, 0.00600 M) || S.H.E. Ecell = V E√³+√√2+ = -0.255 V The Pt…
A:
Q: Draw a structural formula for the major ionic form of the amino acid shown below when in aqueous…
A: In amino acid when the pKa is less than pH than the proton gets deprotonated but if pKa is greator…
Q: How to create a 750 uL of XS buffer (with compositions 1% potassium ethyl xanthogenate ; 100 mM…
A: To prepare 750 μL (0.750 ml) of XS buffer with the given composition following steps are required…
Q: Calculate the pH in the titration of 100.0 mL of 0.20 M HCIO (Ka= 4.0 x 10^-8) with 0.20 M KOH after…
A: Answer :
Q: A STOCK SOLUTION containing 0.1581 g/L K2CrO4 was prepared. In order to make the CALIBRATION…
A: To calculate the ppm and molarity of K2CrO4 in the calibration standard, we first need to calculate…
Q: 5. The equilibrium constant (Kp) for the reaction: N₂O4 (9) #2 NO2 (g) at 25°C is 0.148. A certain…
A: The Gibbs free energy change (∆G) of a reaction and the corresponding reaction quotient (Q) is…
Q: PT₂ (9) 1) Hydrogen and iodine gases where placed into a 1.00 liter container, and allowed to reach…
A: We are given a reaction H2 + I2 <=> 2HI We need to find if the forward or backwards reaction…
Q: Question 3 of 5 A reaction follows the rate law of rate =k[A]²[B]³. How many times faster will the…
A:
Q: Which of the following sugars is a D-sugar? H- IIII H- H- CHO CHO -OH HO-H ОН HO-H CHO -OH H- -OH…
A: If the OH group of penultimate carbon of sugar is on right hand side than this type of sugar is…
Q: If the reaction A + B + C --> Products, follows the rate law of rate= 5.05M-5s-2[A]²[C] what is the…
A:
Q: 3. Starting with but-1-yne and any inorganic reagents required make the target molecule. Show the…
A: Synthons are hypothetical moieties that are proposed during the retrosynthesis of an organic…
Q: Which of the following is a conjugate acid-base pair? A) H3ASO4, ASO4³- B) H₂ASO4, HASO4²- C)…
A:
Q: 6-heptanal is an aldehyde that also contains a c=c double bond between carbons 6 and 7. If this…
A: A chemical reaction is the symbolic representation of the conversion of substances to new…
Q: In the mechanism of the following acid catalyzed hydration transformation, there are distinct…
A: Given that, a reaction scheme is shown below We have to give the number of intermediates and the…
Q: The pH of 300 mL solution made of 0.58 M acetic acid and 0.66 M potassium acetate is (Ka = 1.8 x…
A:
Q: at undergoes: H₂C HC H₂C- Refer to the fat structure below. Draw the possible products if the 6.…
A: Solution: We know fatty acid on saponification we get soap (alkali salt of fatty acid) and glycerine…
Q: C-OH CH₂ IUPAC name: CH₂
A: Since, Rule of IUPAC-1) Longest chain as parent chain.2) Numbering start from those side where more…
Q: Attempt 3 Calculate the pH of the resulting solution when 25.0 mL of 0.30 M HCl*O_{4} is added to…
A: A buffer is a solution which resist any change in pH on adding a small amount of acid or base . it…
Q: 2. On the basis of charge stability, rank species A-G from weakest base to strongest base. ia NO HO.…
A:
Q: Please help me write the molecular and net ionic equations for the following, including states of…
A: First write down the product of the given reaction. Then its total and net ionic equation.
Q: What type of orbital do the electrons with the highest energy reside for the ground-state Sc²+ ion…
A: Electronic configuration refers to the arrangement of electrons in an atom, molecule, or other…
Q: Calculate the equilibrium constant, Kp at 1300 ℃ for the reaction shown below if the standard…
A: We can use the Van't Hoff equation to relate the equilibrium constants at two different…
Q: A 2.490 gram sample of an organic compound containing C, H and O is analyzed by combustion analysis…
A: First calculate empirical formula using mass. Then find out molecular formula using empirical…
Q: What is the pH of a solution that contains 2.5 grams of nitric acid dissolved in 15.2 L of water?
A:
Q: A listing of a house for sale states that there are 6 bedrooms, 4000 ft² of living area, and a…
A: given:- 6 bedrooms living area= 4000 ft² dimensions = 16 x 18.5 ft
Q: The particle in a box (PiB) model can be applied to electrons which move freely in a molecule such…
A: Given: The particle in a box (PiB) model can be applied to electrons which move freely in a molecule…
Q: 1 + 7 2 3 8 4 5 9 6 10
A: Planar completely conjugated hydrocarbons will be aromatic when the ring contains (4n + 2) π…
Q: Both CaCI2 and MgCl2 react with AgNO3 to precipitate AgCI. When solutions containing equal…
A: Since both the solutions have the same mass of salts, suppose the mass of CaCl2 in the solution is…
Q: hello i need help with this question. Assuming standard conditions, and considering the table of…
A: Reducing agent is the one which reduces others and itself gets oxidised by losing electron. For an…
Q: How many unpaired electrons are in the O2+ ion
A: Pauli exclusion principle: It states that only two electrons can be present in an orbital with…
Q: QUESTION 10 In the presence of excess oxygen, methane gas burns in a constant-pressure system to…
A:
Q: 0.10 M formic acid (HCHO₂) Ka = 1.8 x 104 0.10 M sodium formate (NaCHO₂) 0.10 M acetic acid…
A: A buffer is a solution which resist any change in pH on adding a small amount of acid or base .…
Q: molecule proposed Lewis structure PO³ BH, XeO :0: | :0: [H-B-H]* :0: - | :0 Xe - I :0: .. Is this a…
A: The Lewis structure represents the valence electrons of each atom in the molecule. The bonded…
Q: Methanol (CH3OH) can be used as the fuel in a "canned heat" container that is used to keep food hot…
A:
Q: A 31.58-g sample consisting of a mixture of sodium chloride and potassium sulfate is dissolved in…
A: The reaction of lead ions with chloride ion and sulfate ion is given as= Pb2+ (aq) + 2Cl- (aq) →…
Q: To convert an alcohol to an alkene, one would use a hydrolysis reaction. Answer: FALSE, can you…
A: The given statement: Alcohols are converted to alkenes by hydrolysis reaction. The given statement…
Q: If the ?a of a monoprotic weak acid is 5.7×10^−6, what is the pH of a 0.27 M solution of this acid?…
A:
Q: Please help what are special rules or laws to predict predominant products for alcohols or et
A: Here's a chart summarizing some special rules or laws to predict predominant products for alcohols…
Q: When the concentration of H 3 O + in an aqueous solution is 5.6 x 10^-10 M, then the concentration…
A:
Q: O CH3 „NH₂ H+, heat (lose H₂O)
A: Carbonyl compounds undergoes nucleophile addition reactions takes place. Amino group acts as…
Q: The pH of a 0.67 M solution of butanoic acid (HC4H₂O₂) is measured to be 2.50. Calculate the acid…
A:
Q: 2. Using the malonic ester (diethyl malonate), synthesize 2-benzylbutanoic acid. i OH ΕΙΟ OEt
A: The question is based on organic synthesis. we need to synthesize the product from the Reactant…
Q: Decide whether the Lewis structure proposed for each molecule is reasonable or not. Note: If two or…
A: Lewis structures are the diagrams in which total valence electrons of the molecule are depicted in…
Q: Consider the following reaction. Which side is favored in equilibrium? u ů ^-^ 1 Selected answer…
A: A base abstracts proton. A nucleophile adds to the electrophilic center.
Q: Calculate the pH in the titration of 100.0 mL, of 0.20 M HCIO (Ka = 4.0 x 10^-8with 0.20 M KOH after…
A: In acid base reactions the products formed are salt and water, hence these reactions are also…
Q: balancing chemical equations: 6) N2 + O2 -> NO2 9) CuO + H2O -> Cu(OH)2 10) Cu(OH)2 + H2CO3 -> CuCO3…
A: Given unbalanced chemical reactions
Q: 2. Choose the expected major product of the following reaction sequence. Ph Ph HO. A D Ph3P=CH₂ Ph…
A: This is a multi step organic synthesis reaction This molecule under go two wittig reaction and one…
Q: Could you provide the formula of the unkown and the name :) 9. Mass Spectra 100- Relative Intensity…
A: The question is based on the concepts of Organic spectroscopy. we need to analyse and interpret the…
Q: QUESTION 9 Hydrogen peroxide decomposes to water and oxygen at constant pressure by the following…
A:
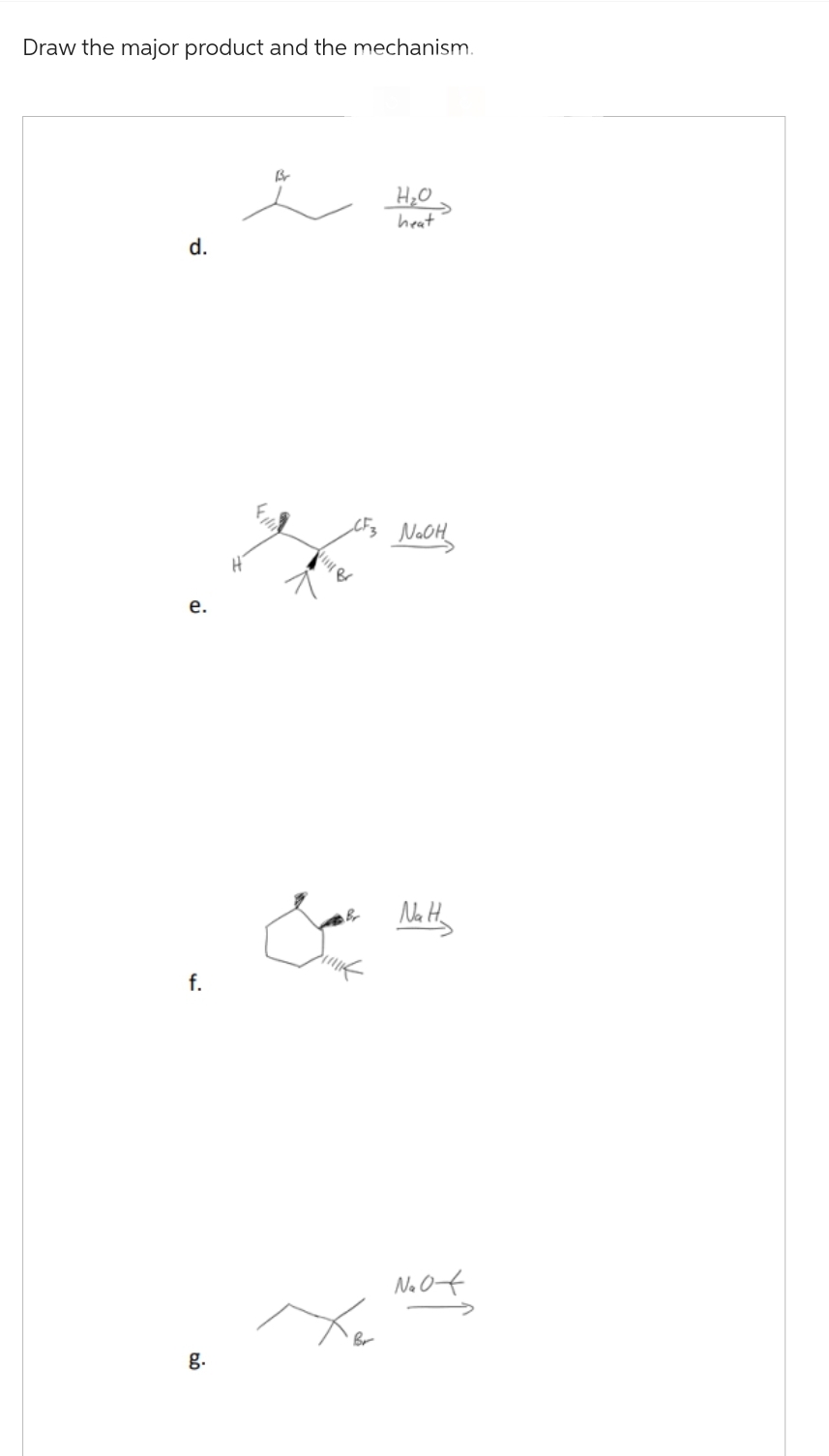
Gg.71.
Step by step
Solved in 3 steps with 3 images

- Mechanisms with major productThe iodination of acetovanillone (1-(4-hydroxy-3-methoxyphenyl)ethan-1-one) occurs in the following reaction: C9H10O3 + NaI + NaOCl --> C9H10O3I + NaCl Draw the complete reaction mechanism of acetovanillone including curved arrows.is this an E1 or E2 mechanism for this reaction? What is the major product and step by step mechanism?

