Q: Он H OH H2, Lindlar's catalyst unctional groups such as alkynes react the same in complex molecules…
A: Role of H2, Lindlar's catalyst selectively reduces alkyne into cis alkene
Q: The triene has a non bonded molecular orbital (a) true (b) false (c) cannot be determined R P
A: The triene is given with three double bonds.
Q: Please help synthesize B. B,
A: Given is organic synthesis reaction.The given starting compound is ketone.
Q: Suppose 7.5 g of Compound A are consumed in a reaction with 3.5 g of Compound B that produces only…
A:
Q: Please predict the products for each of the following reactions. Clearly show the regiochemistry…
A: The reactant is an alkene since it has a carbon-carbon double bond.
Q: Draw the major product of this aldol addition reaction. Ignore inorganic byproducts. H 1. LDA 2.…
A: “Since you have posted multiple questions, we will provide the solution only to the first question…
Q: Oxyacetylene welding torches reach temperatures of -3,200 °C using the following reaction: 2 C2H2(g)…
A:
Q: 4L of 0.02 M aqueous solution of NaCl was diluted by adding one litre of water. The molality of the…
A:
Q: 2. Synthesis questions a. Η b. from from ethyl bromide (the methyl groups in the product have to…
A: The question is based on the concept of organic synthesis.We need to synthesize the product using…
Q: For each of the following interactions between orbitals of two different atoms, choose the correct…
A: Lus now see what types of molecular orbitals are formed by combination of different types of atomic…
Q: Select correct and most relevant mechanism for each of the following reaction. Hint: Review Chapter…
A: Product of following reaction can be made by applying appropriate reaction mechanism.
Q: SC What is the correct IUPAC name for the following compound? O 6-cyclopropyl-5-hexyne O…
A: I hope this helps you! Thank you! Explanation:Step 1: Rule for IUPAC nomenclature Rule of IUPAC-1)…
Q: A solution contains 1.49×10-2 M iron(III) nitrate and 1.26×10-2 M zinc acetate. Solid ammonium…
A:
Q: Synthesize the following compound from benzonitrile (C6H5CN) as the only organic starting material
A: In this reaction, firstly benzonitrile is converted into aldehyde compound by converting into…
Q: Curved arrows are used to illustrate the flow of electrons. Use the reaction conditions provided and…
A: Final answer is given in explanation please see from there.Explanation:Approach to solving the…
Q: Draw the major product of this reaction. Ignore inorganic byproducts. 1. BuLi, -78 °C 2.…
A: answer attached belowExplanation:Step 1: Step 2: Step 3: Step 4:
Q: D Question 10 Choose the correct reagent(s) to perform the following transformation. 1. Choose the…
A: 1. MeMgBr2. H3O+Explanation:Initially, a Grignard reagent is formed by reacting the specified…
Q: Do not use chatgpt. Thank you!
A: The product are discussed in explanation step in below;Explanation:Step 1: Step 2: Step 3: Step 4:
Q: Piperazinone derivatives, such as compound 4, are being investigated for their potential use in the…
A: We have to draw the structure of compound 3.
Q: Complete each trial using the substances and conditions indicated in the table below. Trail Number…
A: According to Bartleby guidelines I will be solving first 3 questions. Please upload other questions…
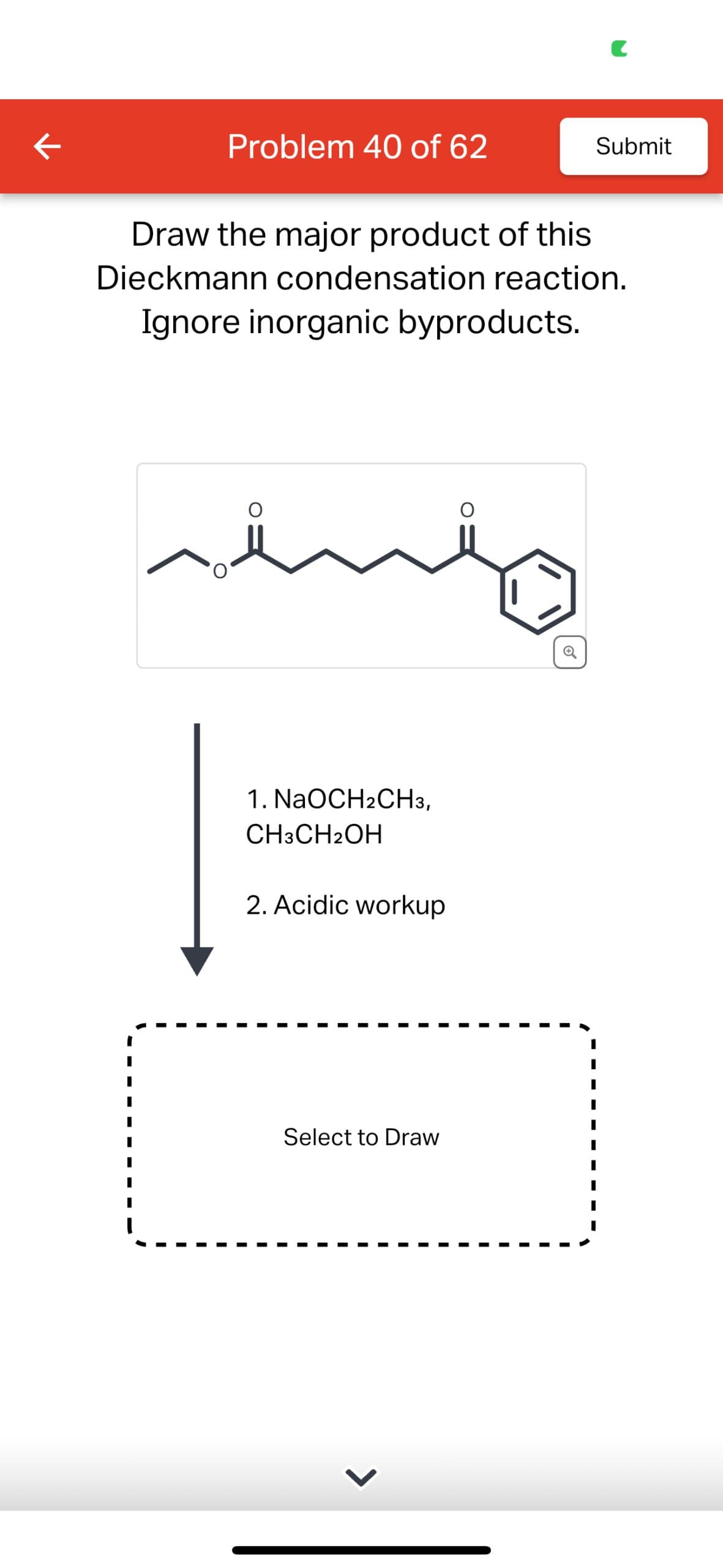
Q: The rate of a reaction is how quickly the reaction goes to completion. If two reactions have the…
A: The objective of this question is to understand how the concentration of reactants affects the rate…
Q: Calculate the value of the potential for the following electrochemical cell under the conditions…
A: Answer:Galvanic cell is a type of electrochemical cell that converts chemical energy into electrical…
Q: Predict an organic product obtained from the following reaction. 1. DIBAH, toluene CH3(CH2)10COCH3…
A: The organic product obtained from the reaction is given below.Explanation:Step 1: DIBAL-H is a…
Q: 28. a) 60u Calculate the molecular mass of HNO3 b) 63u c) 65u
A:
Q: Name the compound using the IUPAC system. O₂N. Br NO 2 NH₂
A: 4-bromo-3,5-dinitroaniline Explanation:For other monosubstituted benzenes, the presence of the…
Q: Which, if any, of the following would not typically have a -3 charge in an ionic comp All have a -3…
A: These are composed of both positive and negatively charged ions together by electrostatic forces.…
Q: CI OH yes Would elimination take place at a significant rate between these reactants? If you said…
A: An elimination reaction can occur via two different pathways ---- E1 and E2.E2 : This bimolecular…
Q: What is the major organic product obtained from the following reaction? OH 1. LiAlH4 2. H₂O*
A:
Q: Predict the major products of the following organic reaction: + .x. A ?
A: The objective of this question is to draw the product obtained from the given reaction.
Q: The rate of a reaction is how quickly the reaction goes to completion. If two reactions have the…
A: The rate of a chemical reaction refers to how quickly the reactants are transformed into products.…
Q: Addition of 2° amine Can you please help me solve this? If possible, can you please draw the…
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: Choose the correct reagent(s) to perform the following transformation. 1. Choose the correct…
A: Reagent for I : MCPBAReagent for II : 1. MeMgBr 2. H3O+Explanation:Thank You.
Q: OH H OH H2, Lindlar's catalyst Functional groups such as alkynes react the same in complex molecules…
A: The above complex structure of compound is treated with H2/ Lindlar's catalyst (Pd, BaSO4) . The H2,…
Q: could you answer both if possible i am stumped on them.
A: Please see the attached files.Explanation:Thank You.
Q: Using any data you can find in the ALEKS Data resource, calculate the equilibrium constant K at 25.0…
A: Given,
Q: A student dissolved 4 drops of an unknown compound in 1 mL of methylene chloride (dichloromethane)…
A: As per Q&A guidelines, we can solve one question and its three sub parts at a time. Please…
Q: The reaction that was on the screen when you started and its derivative demonstrate that the change…
A: The objective of the question is to calculate the change in enthalpy (ΔH) for Reaction 2 using the…
Q: Identify the best reagent(s) to achieve the following transformation. O NaNH2, NH3 O H2, Lindlar's…
A:
Q: Include all lone pairs and charges as appropriate. Ignore stereochemistry. Ignore inorganic…
A: A reaction mechanism describes the step-by-step sequence of elementary reactions by which an overall…
Q: Please predict the products for each of the following reactions. Clearly show the regiochemistry…
A: Reaction of alkene with a cold, dilute solution of potassium permanganate (KMnO4) under basic…
Q: 6) For each of the following carbocations determine if it is likely to rearrange. If so, draw the…
A:
Q: urgent, will rate multiple choice
A: Answer given belowExplanation:Step 1: Step 2: Step 3: Step 4:
Q: Draw the major product of this reaction. Include stereochemistry if applicable. Ignore byproducts.…
A: A nucleophilic substitution reaction is a type of organic reaction where an electron-rich…
Q: Draw the organic products formed in each reaction. NBS =CH2 hv HBr HBr ROOR
A: The question is based on organic reactions.We need to identify the product and explain its…
Q: Which reagent would you use to accomplish each of the following transformations? Options (please…
A: The given reaction scheme is shown below.We have to identify the reagents required for the above…
Q: Predict the major products of the following organic reaction: ཏི ' ། ཡཾ + NC ?
A: The Diels–Alder reaction is a pericyclic reaction (cycloaddition) between a conjugated diene and a…
Q: Splitting of a signal in a proton NMR spectrum tells us the number of chemically non-equivalent…
A: In proton NMR, the splitting pattern gives an insight into the structure of a molecule. The number…
Q: Draw the structure(s) for the major final product(s) formed in the following reaction sequence.
A: Electrophilic aromatic substitution reactions are the characteristic feature of benzene and other…
Q: Give the structural formula for the product formed (if any) from the reaction of acetone with…
A: Carbonyl compounds (aldehyde and ketone) react with an alcohol to form hemiacetal in the presence of…
Q: Question 11 Please predict the products for each of the following reactions. Clearly show the…
A: A reaction during which an addition of H2 occurs across a C=C bond or C≡C bond of an organic…
Step by step
Solved in 3 steps with 2 images

- Percent yield: 68 melting point:115-122 what is the lit melting point for this product? Did you successfully make pure 2,3-Diphenylquinoxaline? ExplainAdol Condensation Lab, Organic Chemistry 2: The same physical property that helps drive the reaction to completion can also stall out the reaction before it starts. What do we do in the procedure that helps minimize this concern? (initially I said that the driving force is 1) reactivity of carbonyl compounds 2) stability of the product please be as detailed as possible, will mark as helpfulI need help with drawing the major product(s) of hexane, iodine and light please
- Rearrangements can occur during the dehydration of 1° alcohols even though no 1° carbocation is formed—that is, a 1,2-shift occurs as the C— OH2+ bond is broken, forming a more stable 2° or 3° carbocation, as shown in Equation [1]. Using this information, draw a stepwise mechanism for the reaction shown in Equation [2]. We will see another example of this type of rearrangement in Section 16.5C.Please help with the mechannismFill in the fields (reagent or products) left blank in the reaction below.
- COMPLETE THE TWO REACTIONS BELOW• What if there is no stability in the emulsion? • How does an emulsion work? • How do you prepare an emulsion? • Provide and explain examples of defoamers that you recognize.What is the objective of this experiment in a few sentences? PART C & D (couldn't post picture) c. Acid HydrolysisIn a small erlenmeyer flask place 25 mL of the stock starch solution and 5 mL of 10%HCI and mix. Place in boiling water bath. After 30 minutes of heating, use a plasticpipette to withdraw a few drops (aliquot) and place them on a spot plate. Allow to coolfor a few seconds and add 2 drops of iodine solution. Observe color and interpret theresults. Withdraw and test an aliquot every 5 minutes until the test with iodine does not produce ablue color. Keep track of the total elapsed time. Interpret the results. Do not discardsample and proceed to part d. d. Fehlings Test on hydrolyzed sampleUpon completing part c. above and the test no longer gives a blue color, withdraw a 5 mLsample and neutralize with 10% NaOH until no longer acidic to litmus. Add 1 mL ofFehlings solution (A+B), place in boiling water bath, and time any color change asbefore. Compare this result with your…

