Q: A chemist adds 105.0 mL of a 0.721M barium chloride (BaCl₂) solution to a reaction flask. Calculate…
A: The volume of the barium chloride solution is = 105.0 mlThe molarity of the barium chloride solution…
Q: la How would you accomplish the following conversion? A) H+/H20; HCN B) -OH/NaCN C) Br2/H+, H20;…
A: The question is based on the concept of organic synthesis.We need to synthesize the product using…
Q: What volume of oxygen gas is produced when 0.682 mol of hydrogen peroxide (H2O2) reacts completely…
A: For the given reaction,Temperature, T = = (0 + 273.15) K…
Q: Write down all the steps of the reaction of 2-Methyl-2-hexene with sulfuric acid and name the…
A: We have to give the product of the reaction between 2-methyl-2-hexene and sulfuric acid.So, the…
Q: You are asked to prepare a 1.000 L solution of 4.5 M C6H12O6 (glucose; molar mass = 180.16 g/mol) in…
A: The number of moles of glucose is calculated as shown below. The number of moles of glucose is 4.50…
Q: If the following molecule cyclized, which of the following statements would be true? H- HO- HO H-…
A: it is the structure of galactose. It is a monosacharide. cyclization results when C5-OH doing…
Q: The following electron configurations represent excited states. Identify the element and enter its…
A: Filling of electrons in atomic orbital follow Aufbau principle.It states that electrons are filled…
Q: Which of the following is the best leaving group? A) H2O B) Cl C) OH D) All are equal What type of…
A: In the context of leaving groups, the best leaving group is typically one that can stabilize the…
Q: Use Le Chatelier’s principle to determine whether the equilibrium for the exothermic reaction below…
A: The given chemical reaction is N2(g) + 3H2(g) → 2NH3(l).We have to determine the equilibrium…
Q: 3) Predict the % of the following products, based on Bromination selectivity. Note: Show your…
A: Alkanes can be halogenated in the presence of light or heat. The alkane does not contain any…
Q: 5. Propose a mechanism to explain the given byproduct. H,(OSI/AI), андан OH CH₂ CH3
A: The given reaction scheme is shown below.We have to give the mechanism of the above reaction.
Q: Lewis Dot Structure NH₂ 3-D VSEPR Structure (with Labeled Bond Angles) 4 Lewis Dot Structure SO,¹…
A: Information of question
Q: cl HO
A: The question is based on organic reactions. We need to identify the product and explain its…
Q: 5. Give the IUPAC names of the fallowing alkanes. a) CH3C(CH3)2CH(CH₂CH3)CH₂CH₂CH(CH3)2 b) c) tâ K…
A:
Q: What is the molar solubility of AgBr in 0.180 M NaBr? 0.09 M 8.0 x 10-11 M 4.2 × 10-¹2 M
A: Information of question
Q: Identify whether each species functions as a Brønsted-Lowry acid or a Brønsted-Lowry base in this…
A: According to Bronsted and Lowry concept,Acid is a substance that has the tendency to donate a…
Q: CH3 Br ОН
A:
Q: Which shows the resonance hybrid of nitrate ion, NO? Select the correct answer below: 0 [0.0.0] Ö N…
A: Resonance structures are the collection of Lewis structures which represents the delocalisation of…
Q: A student proposes the following two-step synthesis of an ether from an alcohol A and another…
A: The question is based on the concept of organic synthesis.We need to synthesize the product using…
Q: Br2 (1 equiv.) CH3CO₂H Na₂CO3, LiBr
A: 1st step is the alpha bromination step in presence of acid.2nd step is the elimination step in…
Q: Draw one product of an elimination reaction between the molecules below. Note: There may be several…
A: Elimination reaction of alkyl chloride.We have to draw one alkene product.
Q: ©Macmillan Learning The reaction for producing glucose in plants, called photosynthesis, is light…
A:
Q: a) Which molecule(s) could successfully form Grignard reagents without any issues? A) ia D) B) CI…
A: The question is based on the concept of organic reactions. We need to identify the compounds which…
Q: lick on the central atom that has a trigonal planar molecular geometry. H :Ο: Η Η H---0---H Η Η Η
A: Click on the central atom that has a trigonal planar geometry.
Q: Consider the following reaction between an alkyl halide and a weak base. -I + CH₂OH
A: Reaction of alkyl halide and methanol is given.We have to identify the product and draw the…
Q: 5.0 g of cream at 23.3 °C are added to an insulated cup containing 150.0 g of coffee at 84.7 °C.…
A: Given: Mass of cream= 5.0 g The temperature of cream = 23.3°CMass of coffee=150.0 g The temperature…
Q: Question 13 Triacylglycerols can undergo hydrolysis. True O False Question 14 Fatty acids can…
A: The given two statements are shown below.Triacylglycerols can undergo hydrolysis.Fatty acids can…
Q: I H. H H
A: The given compound is .We have to give the IUPAC name of the given compound.
Q: Draw all likely products of the following reaction. CH₂ -OH B H₂SO, heat Draw the molecules on the…
A: Alcohol gives dehydration reactions when it reacts with a strong acid like H2SO4 or H3PO4 to form an…
Q: Draw the major E2 reaction product formed when cis-1-chloro-2-ethylcyclohexane (shown) reacts with…
A:
Q: I I I I I I 1. NaOCH₂C H3 2. C6H5CH₂I (1 equiv) Ph Select to Draw H3O+ heat Select to Draw × × Ρήτ
A: Given,The reaction:
Q: Predict the product(s) for the following reaction. Br₂ / H₂O
A:
Q: Draw structures for the products of this reaction (no mechanism necessary. CH₂ 1) LIAIH/E₂O 2) H₂O*…
A: LiAlH4 is strong reducing reagent, useful for the reduction of ketone, aldehyde, amide, acid…
Q: Homework Problems 4.1 Calculate the number of moles in a 5.00-g sample of each of the following: a.…
A:
Q: Predict the product of this organic reaction: _i_ CH3-C−CH2NH, + ₂-i-OH CH3-CH₂-C-OH P + H₂O…
A: Carboxylic acid reacts with amine to form amide.
Q: Draw the structures of the amino acids formed at physiological pH when the tripeptide below is…
A: If the pH is greater than pKa then deprotonation occurs and it exists in deprotonation form. While…
Q: Give detailed Solution with explanation needed..don't give Handwritten answer...
A: Molecular geometry is three dimensional arrangement of atoms around the central atom in a…
Q: Hi, I am in chemistry 2 and am working on acids and bases. However im struggling with identifying a…
A: The question is based on the concept of salt hydrolysis. We need to identify the nature of given…
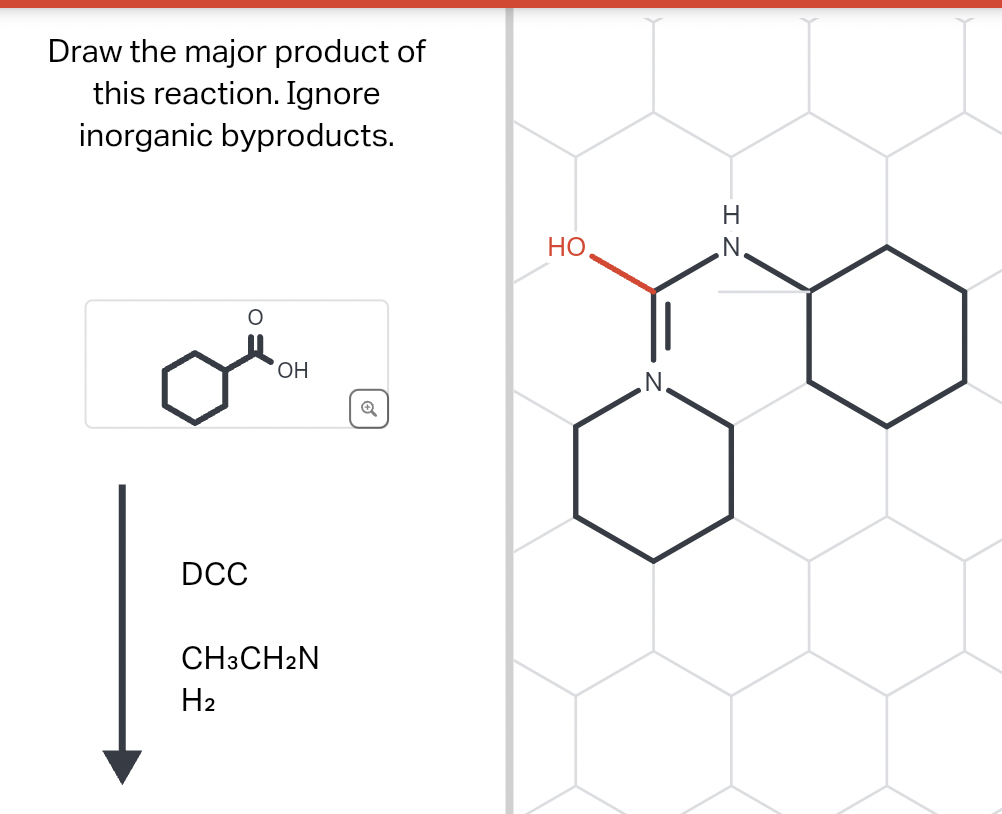
Q: Draw the major product of this reaction. Ignore inorganic byproducts and the amine side product. 'N…
A: Acidic hydrolysis of amide give Carboxylic acid and amine.
Q: -Fe is cubic. Is it FCC or BCC? 20 30 110 sommy kambarys 40 50 60 200 Diffraction angle 20 70 211…
A: The type crystal system can be decided by seeing the peaks in the powder xrd spectrum. For BCC type…
Q: For the following electrocyclic ring closure reaction a. Give the curved arrow pushing and the major…
A: The given reaction is .We have to give the product of the given reaction.
Q: What is the desired theoretical yield of carbon dioxide?
A: % yield of CO2 = 85%Actual yield of CO2 = 540 g
Q: Answer is incorrect I already lose the point for question. Kindly please give m ethe answer for…
A: Given that, for an electrochemical cell, the cell potential is E = 1.1 V.The number of electrons…
Q: CN NH3 OH OMe H SOCI₂ i. MeMgBr ii. H3O+ OH NaOH
A: The given reaction scheme is shown below.We have to provide the missing reagent and the products.
Q: Pls help me Relative Reduction Potential Assuming standard conditions, and considering the table of…
A: Reducing agent is the one which reduces others and itself gets oxidised by losing electrons. For an…
Q: What is the relationship between the parent structure on the left and the structure on the right? Me…
A:
Q: :O: H3O+ OH H I 'Н Draw Tautomer X
A: Tautomers are the isomers which involve the interchange of H-atom.
Q: What are the roles of the kinetic parameters on the reduction of the concentration of NO? This…
A: Kinetic parameters concerned with the reduction of the concentration of NO are:1) Reaction rate…
Q: Given the following thermochemical equation: 2H₂(g) + O₂(8) - 2H₂O(1) ΔΗ° = -571.6 kJ Determine the…
A:
Q: Draw a structural formula for the more stable carbocation intermediate formed in the reaction shown.…
A: Alkene gives an electrophilic addition reaction with hydrogen halide to form an alkyl halide.The…
#10 help ,I got it wrong
Step by step
Solved in 3 steps with 2 images


