Q: Make an electron-flow-mechanism for this synthetic scheme. This involves predicting major and…
A: Synthesis of Ascorbic acid from D - glucose with mechanism is given below.
Q: Draw a stepwise mechanism for the conversion of heptane-2,6-dione to 3-methylcyclohex-2-enone with…
A: First, the general equation is as follows,
Q: Which is NOT true of keto-enol tautomerism? Select the correct response: Rapid interconversion. The…
A:
Q: Draw a stepwise mechanism for the biological conversion of linalyl diphosphate to limonene.
A:
Q: Draw the mechanism that shows how α-D-allopyranose converts to α-D-allofuranose under acidic…
A:
Q: J. Answer questions A-D with respect to the following reaction (requires more than one step). Br Br…
A: Here we have to synthesize the following given compound from propyne in multistep reactions by retro…
Q: Draw the mechanism for how a-pinene is biosynthesized from geranyl pyrophosphate. a-Pinene
A: Alpha –Pinene (α-pinene) is an organic compound of the terpene class, one of two isomers of pinene.…
Q: Draw a stepwise mechanism for the biological conversion of linalyl diphosphate to limonene.
A: The first step of the mechanism involves the removal of OPP molecule from the reactant molecule…
Q: Draw a stepwise mechanism for the biological conversion of linalyldiphosphate to limonene.
A: The mechanism is as follows: 1) The C-OPP undergoes heterolytic cleavage and that produce a stable…
Q: Explain the Mechanism of Tautomerization ?
A: In this question, we will explain the mechanism of Tautomerization. You can see details explanation…
Q: Propose a retrosynthesis of the following compound using the michel reaction.
A:
Q: Use the curved arrow formalism to draw the complete mechanism for the Claisen condensation reaction…
A: Given Reaction = Claisen Condensation Reactant methyl acetate methyl propionate Solvent Ether…
Q: how mechanism for synthesis of acetic acid using CH3COONa and HSO4
A: To illustrate: Mechanism for synthesis of acetic acid using CH3COONa and HSO4
Q: If squalene is synthesized in a medium containing acetate with a 14C-labeled carbonyl carbon, which…
A:
Q: Draw the Robinson annulation product formed from the following starting materials.
A: Given reaction,
Q: Explain the mechanism of Epoxidation of an Alkene with a Peroxyacid ?
A: Concept Introduction: The epoxidation of an alkene is the conversion of the C=C double bond to an…
Q: Draw the cyclohexanone oxime synthesis reaction mechanism.
A: The overall reaction is as follows:
Q: nthesis, show two different synthesis strategies. In the first synthesis strategy, include e…
A: The questions based on organic reactions. We have to convert the reactant into product by a series…
Q: Draw the product formed from the ring-closing metathesis of eachcompound. Then, devise a synthesis…
A: 1- First we will see tha Products for each by using ring closing metathesis.
Q: What starting materials are needed to synthesize each compound using a Robinson annulation?
A:
Q: Determine the mechanism of nucleophilic substitution of attached reactionand draw the products,…
A:
Q: Match the reactions with the correct description. Match each item to a choice: Acetoacetic ester…
A:
Q: Show the mechanism of keto-enol tautomerism that includes the formation of enolate conjugate ions…
A: keto-enol tautomerism is a reaction in which the ketone and its enol forms are present in…
Q: Show the detailed reaction mechanism symmetrical aldol condensation of propanal to…
A: Solution: Symmetrical Aldol condensation is nothing but the Aldol condensation of same substrate ( a…
Q: Draw the mechanism for the formation of the epoxide of cyclopentene with first Br2/H2O followed by…
A: We have to predict the mechanism for the formation of epoxide of cyclopentene.
Q: Draw the mechanism of the favorable reaction of CH3CO2H and sodium ethoxide (CH3CH2O-Na+) through…
A: Bronsted-Lowry acid-base theory: The Bronsted-Lowry acid-base theory states that the acid is a…
Q: Draw a reaction mechanism to account for the formation of the dipeptide (cy = cyclohexyl)
A:
Q: Show the process of synthesizing an enolate compound from cyclohexanone using an LDA catalyst.
A:
Q: Nitration of aniline is carried out after acylation because :Select one a. O-and p-products are…
A: Given, Nitration of aniline is carried out after acylation because ? select one a). o- and…
Q: С. Claisen condensation
A:
Q: the product is given predict from what alkene reactant be made from, write the complete reaction on…
A:
Q: Provide the complete mechanism using curved arrow formalism for the reaction of pyruvic acid treated…
A:
Q: How do you know whether or not two ketones or two aldehydes or a combination was used to synythesize…
A: Ozonolysis process can be used to determine whether two ketone/ aldehyde/ combination is been used…
Q: Draw the mechanism for the dehydration step in the Wohl degredation.
A: Wohl degradation as it name suggests is a carbon reduction step used in carbohydrates, specially…
Q: Draw one resonance form of the Wheland intermediate formed during the para–nitration of anisole…
A: See the solution given below
Q: Please select the appropriate reagent to complete the following transformations. A a B b C D | d…
A: The transformation taking place is given as,
Q: draw reaction mechanism of cyclohexanone being reduced by sodium borohydride to form cyclohexanol.
A: Ketones on reduction with sodium borohydride give alcohols. Hence Cyclohexanone on reduction with…
Q: Draw the mechanism for the formation of diphenylacetylene
A: It is a 2- step process in which we generate triple bond of an alkyne by brominating the double bond…
Q: Aldol Mechanisms: Synthesis of 2-ethyl-hex-2-enal
A:
Q: Make an electron-flow-mechanism for this synthetic scheme. This involves predicting major and…
A: Mechanism of synthesis of ascorbic acid from glucose is given below.
Q: What product is formed by ring-closing metathesis of each compound?
A: Alkenes metathesis is a reaction between two alkenes which results in the interchange of double…
Q: Define each term and give an example: i) aldolic condensation j) cross aldolic condensation k)…
A: As per the policy, we are allowed to answer only 1st three subparts of the question. To get the…
Q: Draw the product formed from the ring-closing metathesis of each compound. Then, devise a synthesis…
A: (a) Synthesis:
Q: Choose one answer from each parenthesis. Th
A: The two reactions that are taking place are: 1. Epoxidation of alkene 2. Acidic hydration of alkene
Q: How To Synthesize a Compound Using the Robinson Annulation ?
A: Given: Synthesize a Compound Using the Robinson Annulation
Q: Draw the product when the following compound undergoes ring-closing metathesis.
A: The product of the reactions using ring-closing metathesis has to be given,
Q: Draw a reasonable mechanism to form the terpene shown from the biologičal precursors shown: Several…
A: First two substrate will undergo additive reaction followed by cyclization and hydration dehydration…
Q: CH3 CH2 CH3 CH3 CH3 H3C H3C CHs OPP
A: The reaction is intramolecular cyclization.
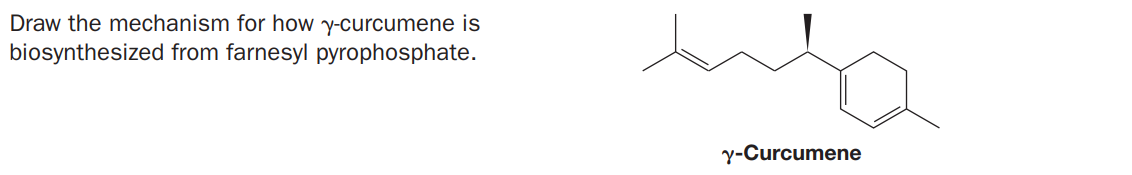
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 4 images

- draw an electron-pushing mechanism that shows the formation of the anti looking products using AE then SN2 mechanistic pathwayDraw the mechanism that shows how α-D-allopyranose converts to α-D-allofuranose under acidic conditions.Draw a mechanism for the conversion of glucose from its aldehyde form to its hemiacetal form and then to its methyl glycoside (acetal) form under acidic conditions. Show all bond breaking and bond forming steps; show all intermediates.
- Draw the product formed when attached compound is treated with two equivalents of CH3CH2CH2CH2MgBr followed by H2O.Draw the mechanism that shows how α-D-allopyranose converts to β-D-allopyranose under acidic conditions.What ring-closing metathesis product is formed when each substrate is treated with Grubbs catalyst under high-dilution conditions?
- Choose one answer from each parenthesis. The identity of Structure A is represented by the structure labelled (SAT / MAT / CAT / BAT / WAT / RAT / NAT) while that of Structure B is labelled (SAT / MAT / CAT / BAT / WAT / RAT / NAT). The missing reagent in the first reaction is (dilute H2SO4 / H2O / MCPBA / Br2 and water).identify the reagents that will accomplish the following eneatioselective transformationThe following isomerization reaction, drawn using D-glucose as starting material, occurs with all aldohexoses in the presence of base. Draw a stepwise mechanism that illustrates how each compound is formed.
- Draw a mechanism for the Fischer esterification to synthesize Wintergreen.Draw a detailed mechanism for the bromination of ethyl phenylacetate with NBS.Draw mechanism for fischer esterification using the reagents benzoic acid, methanol and sulfuric acid using the general mechanism attached as reference. HINT: You just need to replace the Rs in the general mechanism for the actual groups in your reagents.

