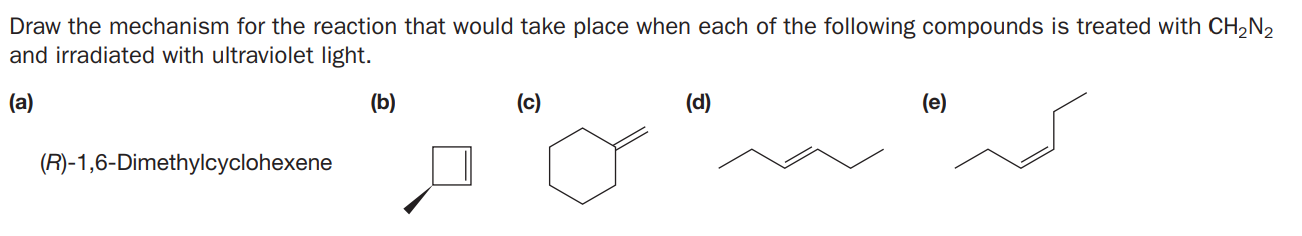
Draw the mechanism for the reaction that would take place when each of the following compounds is treated with CH2N2 and irradiated with ultraviolet light. (a) (b) (c) (d) (e) (R)-1,6-Dimethylcyclohexene
Q: Provide the complete mechanism using curved arrow formalism for the formation of the product shown…
A: -> Carbonyl compound has electrophilic center at which it can give nucleophilic addition reaction…
Q: Provide a plausible, stepwise mechanism to account for the following transformation. Use curved…
A: The mechanism can be shown as:
Q: When alkyl fluorides are reacted with strong bases, it was found out that the mechanism does follows…
A: E1cb reactions are elimination reactions in which the first step is the loss of proton and making of…
Q: 211. Draw all the possible chair-like conformations of compounds 1-2. In the presence of a strong…
A:
Q: Consider the following transformation and answer the questions that follow. OTBS OTBS OTBS OTBS Bno…
A:
Q: Draw the SNl mechanism of the reaction between 2,5-dimethyl-2,5-hexanediol and hydrochloric acid.…
A:
Q: Which of the two isomers would you expect to undergo E2 elimination faster? trans-1-bromo-…
A: Out of these two, trans-1-bromo-4-tertbutylcyclohexane will undergo faster E2 elimination.
Q: OH в
A:
Q: When allyl bromide is refluxed with magnesium metal in ether solvent, the product formed is…
A: At first grignard reagent is formed, and it become nucleophilic, so further attack in allyl bromide…
Q: 2. (a). Compound B is an effective medication used to treat major depression by inhibiting the…
A:
Q: Decane undergoes thermal cracking to produce compound D and compound E with equal number of carbon…
A: Thermal cracking is the process where hydrocarbons are subjected to high temperature and pressure to…
Q: a) Draw both chair conformations of the starting alkyl bromide. Indicate which conformation, if…
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: Provide the mechanism for the following transformation. D = 2H
A: At first D+ present in D-Br adds to the alkene to give tertiary carbocation. Then 1,2-Me- shift…
Q: Draw all the products that are expected when 2-ethyl-3-methyl-1,3-cyclohexadiene is treated with HBr…
A: Draw all the products that are expected when 2-ethyl-3-methyl-1,3-cyclohexadiene is treated with HBr…
Q: The epoxide starting material shown here is a synthetic derivative of the antimalarial drug…
A: Synthetic derivative of antimalarial drug artemisinin.
Q: What starting material is required in order to synthesize each of the following compounds by…
A: Alkene Metathesis or (olefin metathesis): In this reaction breaking of the double bond of an alkene…
Q: Which of the following structures would be considered an enol tautomer of cyclopentanone он OH A…
A: • Tautomers ⇒→ Tautomens are the structunal isomer which are in enconvent readily.→ In…
Q: B) Write the most stable enol forms of the following compound, and choose which of them is more…
A: Enolisation occurs via tautomerism (it is a phenomenon in which different structures ahave relative…
Q: 4. Each of the following alkyl bromides gives only one product in via an E2 mechanism. Provide the…
A: The different 3-dimensional arrangement of atoms that results due to free rotation about C-C single…
Q: Rank the compounds in each group in order of increasing reactivity in electrophilic aromatic…
A: a). The reactivity of compound toward electrophilic aromatic substitution increases with the…
Q: Write a detailed mechanism for the following reaction. Include the slow and fast steps. Draw the…
A: In the given reaction, the HBr is added to alkene to produce alkyl bromide. It is called…
Q: Which of the following are achiral? And identify the relationship between each of them. Ex. A-B,…
A: The first step in solving this problem is assigning the R,S configuration to each chiral center. For…
Q: COOME CHO CO2ME
A: Electrocyclic reaction is unimolecular... And diels alder reaction is biomolecular... Diels alder…
Q: Consider the following chemical transformation: The transformation takes place via two sequential…
A: PERICYCLIC REACTIONS
Q: Compound A yields the following two products upon ozonalysis. Which of the following is the correct…
A: Solution: We know alkene on ozonolysis gives carbonyl compounds by cleaving alkene C=C bond &…
Q: Glucuronic acid is an oxidized derivative of glucose found in animal secretions such as saliva.…
A: First step of this reaction is protonation where glucuronic acid is attacked by H+ ion in the…
Q: Draw both chair conformations of the starting alkyl bromide. Indicate which conformation, if either…
A: Hello. Since your question has multiple sub-parts, we will solve the first three sub-parts for you.…
Q: Elimination, stereochemical aspects. Draw the major product that is obtained when…
A: We have given that Draw the major product that is obtained when (2R 3R)-2-Bromo-3-phenylbutane is…
Q: Considering the stereochemistry, what is the name of the product when cyclopentene undergoes…
A: Bromination of alkene is defined as when the alkene is treated with bromine then carbon to carbon…
Q: Q17. Compound 1 can undergo an intramolecular reaction to give cyclic product 2. Using curly arrows,…
A: Applying concept of cyclation reaction of formation of cycloamide.
Q: The reaction of (S)-2-bromopentane with potassium cyanide to yield 2-methylpentanenitrile…
A: Stereospecific reaction is defined as a reaction in which the reactant's stereochemistry controls…
Q: Friedel–Crafts alkylation of benzene with (R)-2-chlorobutane and AlCl3 affords sec-butylbenzene.a.…
A: Friedel – Crafts alkylation of benzene with (R) -2- Chlorobutane and AlCl3 gives Sec-butyl benzene.
Q: a) Draw both chair conformations of the starting alkyl bromide. Indicate which conformation, if…
A: Since you have posted a question with multiple sub-parts, we will solve first three subparts for…
Q: Answer the following questions about the mechanism for the acid-catalyzed hydration of an alkene: a.…
A: The hydration of alkene results in the formation of alcohol. Consider an example of ethylene that…
Q: When treated with NaOH, the bromide below gives an alkene by the E2 mechanism, by elimination of the…
A: A-E all points are explained. It is cis as two higher priority groups are present on same side of…
Q: Consider the substitution reaction that takes place when (R)-3-bromo-3-methylhexane is treated with…
A: The reactant given is (R)-3-bromo-3-methylhexane.
Q: Propose the mechanism for the mechanism for the reaction of the hydroxyl groups of compound 2 in the…
A: In this question, we will draw the mechanism of the Compound-2 to 3 and 4 to 5
Q: Rank the compounds in each group in order of increasing reactivity in electrophilic aromatic…
A:
Q: 4. Each of the following alkyl bromides gives only one product in via an E2 mechanism. Provide the…
A:
Q: What starting material is required in order to synthesize each of the following compounds by…
A: Alkene Metathesis or (olefin metathesis): In this reaction breaking of the double bond of an alkene…
Q: (1R,2R)-1-Bromo-2-methylcyclopentane is reacted with sodium methoxide. Given the product(s) and show…
A: An elimination reaction in organic chemistry is a reaction where a base abstract An acidic proton…
Q: Provide a mechanism for the following reaction. Please explain it in detail 1. CI NH3 NH CI N
A: When N,N-dichloroethyl-N-methylamine treated with Ammonia then a cyclic product is formed with two…
Q: The specific angle of rotation of (R)-2-bromobutane is -23.1°. Treatment of (R)-2-bromobutane with…
A: The specific angle of rotation of (R)-2-bromobutane is -23.1 degrees. Treatment of (R)-2-bromobutane…
Q: Draw the expected major product of catalytic hydrogenation of the following alkene. Use wedged and…
A:
Q: ONa
A: Here I have provided n arrow-pushing mechanism for the following reaction. This is favourable…
Q: Provide a reasonable stepwise mechanism for the following transformation. Show the structure of any…
A: Solution: We know all kinds are are electron reached due to buy bonds so they can act as nucleophile…
Q: The following alkene is treated with one equivalent of N-Bromosuccinimide in dichloromethane in the…
A:
Q: Draw the expected major product of catalytic hydrogenation of the following alkene. Use wedged and…
A:
Q: For the following reaction, provide a detailed step-wise mechanism showing t electron flow using the…
A:
Trending now
This is a popular solution!
Step by step
Solved in 5 steps with 5 images

- Draw a detailed mechanism for the FeBr3@catalyzed reaction of ethylbenzene with bromine, and show why the sigma complex (and the transition state leading to it) is lower in energy for substitution at the ortho and para positions than it is for substitution at the meta position.If compound 1 is instead reacted with strong aqueous acid a different product is formed, with the formula C12H1O. Identify the product and draw the full curly arrow mechanism for its formationIdentify R and S in the following reaction sequence, and draw a mechanism for the conversion of R to S (molecular formula C6H10O3). S was used in the synthesis of darunavir (trade name Prezista), used to treat HIV.
- Explain in detail which of the following compounds (A or B) when reacted with acetophenone under basic conditions will undergo a 1,4 addition reaction. Predict the product of the reaction and propose a mechanism.Provide the complete mechanism using Curved Arrow Formalism for the reaction of (R)-3,3-dimethyl-2-hexanol treated with aqueous phosphoric acid. If more than one product is formed state which is major, minor, very minor, etc. Name the product(s).The bicyclo [3.1.0] hexane ring system, highlighted in compound 3, is found in several natural products, including sabinene, a compound partially responsible for the flavor of ground black pepper. One method for preparing this ring system involves the conversion of compound 1 to compound 2, as shown below. Draw the structure of compound 2 and provide a reasonable mechanism for its formation. Add any remaining curved arrows to complete step one of the mechanism, and modify the given drawing as needed to show the intermediate that is formed in this step.
- When alkyl fluorides are reacted with strong bases, it was found out that the mechanism does follows a rather unconventional E1 mechanism. It was found out that the compound first forms a carbanion before the elimination. Which of the following will be the most likely product when 2-fluoropentane undergoes a reaction with an strong base? (E)-pent-2-ene Pent-1-ene Both B and C (Z)-pent-2-eneConsider the following chemical transformation:The transformation takes place via two sequential pericyclic reactions. Identify the two reactions and give a critical explanation whether the reactions are allowable or not. Explain the stereochemistryDrawing an SN2 product with More Complex Reactants Identify C, the product of an SN2 reaction in the synthesis of raloxifene, a drug used to reduce the risk of invasive breast cancer in postmenopausal women.
- Although there are nine stereoisomers of 1,2,3,4,5,6hexachlorocyclohexane, one stereoisomer reacts 7000 times more slowly than any of the others in an E2 elimination. Draw the structure of this isomer and explain why this is so.When allyl bromide is refluxed with magnesium metal in ether solvent, the product formed is 1,5-hexadiene. (C6H10). What is the curved arrow mechanism for this reaction?Friedel–Crafts alkylation of benzene with (R)-2-chlorobutane and AlCl3 affords sec-butylbenzene.a. How many stereogenic centers are present in the product?b. Would you expect the product to exhibit optical activity? Explain, with reference to the mechanism.

