Q: 4. Thermodynamic data for C(graphite) and C(diamond) at 298 K is given in the table below. 6) Will…
A: The objective of the question is to calculate the change in enthalpy (ΔH°), entropy (ΔS°) and Gibbs…
Q: Question 2 A radioactive sample at a lab contains a mixture of 223 Ra and 47 Ca. There are initially…
A: Step 1:Radium-223 (223Ra) has a half-life of 11.4 days. Calcium-47 (47Ca) does not decay…
Q: The rate constant for this first-order reaction is 0.720 s -1 at 400 °C. A → products How long, in…
A: Detailed explanation:Given the first-order rate law, the time t can be calculated using the…
Q: b). What is the conjugate base of NH4? c) The Ka for NH4* is 5.5x10-10. What is the pKa for its…
A: b) Conjugate base is formed when a species releases a proton (H+).NH4+ →−H+ NH3so, conjugate base of…
Q: • Provide conditions for one of these transformations OCH3 a) LOCH3 H3CO H3CO SO3H OCH3 b) OCH3 H3CO…
A: 1) Electrophilic aromatic substitution reaction in which methoxy groups are ortho, para directing…
Q: Determine the ground state configuration and number of the unpaired electrons in the following…
A: The arrangement of the electrons in the different orbits around the nucleus of an atom is known as…
Q: g) 1) NaOH 2) H₂C H3C Η
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: The free radical chlorination of isopentane yields isomeric monochlorinated isopentanes, namely:…
A: Step 1: Step 2: Step 3: Step 4:
Q: Draw and label the chemical structure of the following reaction (include the curly arrow reaction…
A: The chemical reaction, including the curly arrow mechanism: H H \ / C=C / \…
Q: Predict the major product.
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: 7. Predict the products for the reactions below. 1) excess EtMgBr 2) H20 excess NH3 ОН pyridine 1)…
A: Step 1: a) Propanoic anhydride with excess of ethyl magnesium bromide gives 3-ethylpentan-3-ol.First…
Q: Predict the products of the following reactions. CH3 CH3 CH CH2 -NH2 -CH2 + H₂O CH3 + H₂O CH2 N CH3…
A: Items 1 and 2: Amines have a lone electron pair on their nitrogen atoms and can accept a proton from…
Q: 26. MATHEMATICAL Determine the values of Ky and Vmax for the de- carboxylation of a ẞ-keto acid…
A: The objective of the question is to determine the values of Ky and Vmax for the decarboxylation of a…
Q: 2. Consider the following reaction: 4 HCN(/) + 5 O2(g) → 2 H₂O(g) + 4 CO₂(g) + 2 N2(g) (a) Using the…
A:
Q: Predicting qualitatively how entropy changes with temperature and... 1/3 Izabella For each system…
A: Here are the things you need to know about the entropy of a system:When temperature is increased,…
Q: Ascorbic acid, H₂C6H6O6, is a diprotic acid usually known as vitamin C. For this acid, pk1 is 4.10…
A:
Q: Data Buffer pH without adding HCL = 3.56 pH of buffer with 1 ml HCL = 2.14 pH of buffer with 2 ml…
A: The ability of a buffer solution to withstand pH changes upon the addition of either an acid or a…
Q: −4 A chemistry graduate student is given 125. mL of a 0.60M nitrous acid (HNO 2) solution. Nitrous…
A: The objective of this question is to find out the mass of sodium nitrite (NaNO2) that needs to be…
Q: how can i plot this data for concentration with depth?
A: Step 1:It is needed to plot this data for concentration with depth. Step 2:Enter the given data in…
Q: Cp = 2.07+ T-673 1480 kJ kgK T-673 kJ Cy=1.6085+ 1480 kgK The specific heat of superheated water…
A: Given Data:The specific heat of water vapor at constant pressure is, .The specific heat of water at…
Q: For each of the following compounds, decide whether the compound's solubility in aqueous solution…
A: To determine whether the solubility of a compound changes with pH, we need to consider the effect of…
Q: from Br b)
A: Step (i): In the first step, a longer carbon chain alkane (pentane) is formed by the given reactant…
Q: The best known acrylic is polymethylmethacrylate, which is sold as a product known as lucite. (a)…
A: (a) The structure of polymethylmethacrylate (PMMA), which is also known as acrylic or Lucite, is…
Q: I need help with part b
A: 2. After adding 15 mL of KOH: At this point, 15 mL of 0.100 M KOH reacts with the barbituric acid.…
Q: Draw the first round of beta -oxidation for lauroyl-CoA
A: Approach to solving the question: Detailed explanation:The first round of beta-oxidation for…
Q: 2+ Fe (aq) + H2O = FeOH(aq) + H(aq) (C) FeOH + e = FeOH (surface) (E) (aq) FeOH(surface) + H(aq) + e…
A: The objective of the question is to determine the step that depicts the overall rate.
Q: Calculate the volume in liters of a 0.15M barium chloride solution that contains 225 mmol of barium…
A: The objective of this question is to calculate the volume of a 0.15M barium chloride solution that…
Q: If the pH of a 0.15M acid is 3.6, what is the Ka
A: The objective of this question is to calculate the acid dissociation constant (Ka) for an acid given…
Q: 3 NEXT > < PREV 1 2 Based on your ICE table (Part 1) and the definition of Ka, set up the expression…
A: For Parts 1 to 3, a buffer system was prepared with HC7H5O2 and NaC7H5O2. The reaction for the…
Q: A chemist adds 0.10 L of a 4.51 mol/L silver perchlorate (AgC104) solution to a reaction flask.…
A:
Q: Part A For a single substance at atmospheric pressure, classify the following as describing a…
A: Approach to solving the question: Detailed explanation: Examples:
Q: how to thioether oxidated by meta - chloroperoxybenzonic acid to sulfuroxides, please draw its…
A: 1. **Approach of mCPBA to the Thioether:** - The thioether sulfur atom, which is nucleophilic,…
Q: What is shown below is an interesting way of making lactone B from lactone A. The reaction takes…
A: Step 1: Step 2: Step 3: Step 4:
Q: 3. Calculate the Packing Factor of the Zinc Blenda structure to know the degree of occupancy of the…
A: ### Steps to Calculate the Packing Factor1. **Determine the Number of Atoms per Unit Cell:** - In…
Q: None
A: Step 1:this reaction is called an E2 (elimination) reaction. In this reaction, 1-bromohexane (an…
Q: What is the wavelength (in nm) of a photon if the energy is 7.45 × 10⁻¹⁹ J? (h = 6.626 × 10⁻³⁴ J • s…
A: The objective of this question is to calculate the wavelength of a photon given its energy. The…
Q: None
A: Reaction: Answer:
Q: y" 3y 18y = sin(8) = y(0) 3, y'(0) = -1 Using Y for the Laplace transform of y(t), i.e., Y =…
A: Step 1: Step 2:
Q: Does resolution of the paper chromatography method can be great enough for complete separation of…
A: The objective of the question is to understand if the resolution of the paper chromatography method…
Q: Calculate the volume in millimeters of a 1.01 mol / L aluminum chloride solution that contains 100 g…
A: The objective of this question is to calculate the volume of a 1.01 mol/L aluminum chloride solution…
Q: Consider the reaction: 2SO2(g) + O2(g) ⇌ 2SO3(g) K = 1.8 x 102The reaction chamber was initially…
A: The objective of the question is to calculate the partial pressure of O2(g) at equilibrium given the…
Q: −4 A chemistry graduate student is given 125. mL of a 0.60M nitrous acid (HNO 2) solution. Nitrous…
A: A chemistry graduate student can use the Henderson-Hasselbalch equation to determine the amount of…
Q: None
A: Step 1:(a) To calculate the solubility-product constant (Ksp) for Pb(IO3)2, we use the…
Q: The equation for the overall reaction that occurs in Complex III is shown below: QH2+2 cytochrome c…
A: The redox couples [Ubiquinol (QH2)/Ubiquinone (Q)] and [Cytochrome c (Fe3+)/Cytochrome c (Fe2+)]…
Q: The structure of the amino acid tyrosine is shown below in its completely protonated form. PK 16 OH…
A: The question is asking for the structure of the amino acid tyrosine in its completely protonated…
Q: Draw structures for the carbonyl electrophile and enolate nucleophile that react to give the aldol…
A:
Q: :):):));;$:&:&$:
A:
Q: what is given off during radioactive decay?
A: Step 1:
Q: True or false, explain your answer.
A: The compound contains a stereocenter, specifically the carbon atom bonded to the oxygen atom (C∗).…
Q: None
A:
Draw the mechanism of the transformation from 3 to 4 .
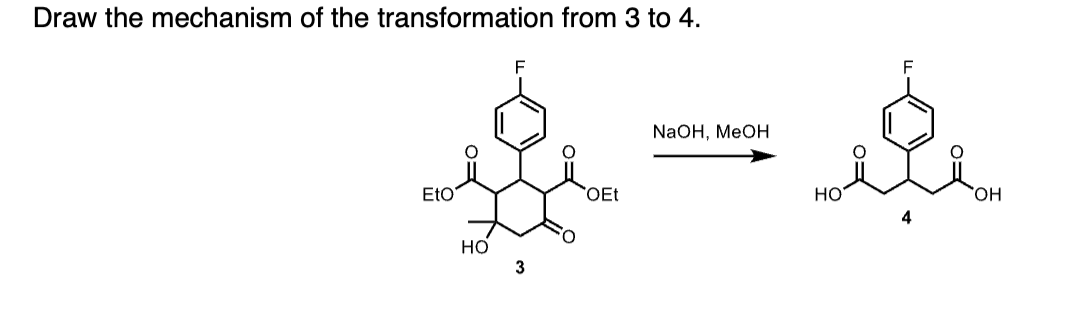
Step by step
Solved in 2 steps with 1 images




