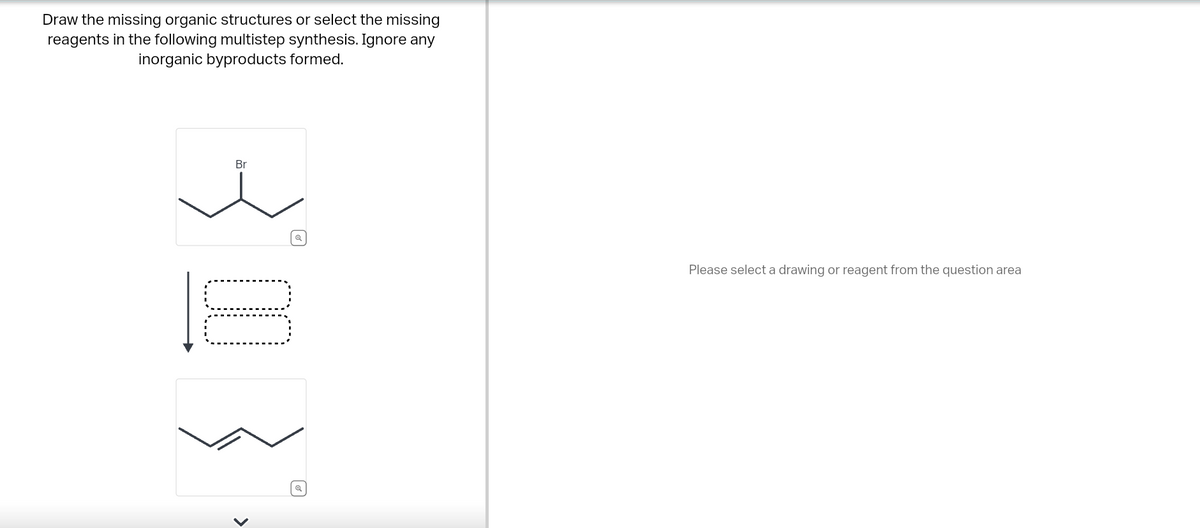
Draw the missing organic structures or select the missing reagents in the following multistep synthesis. Ignore any inorganic byproducts formed. Br ધ > Q Please select a drawing or reagent from the question area
Q: What are the products of a mononitration reaction of the following reactants? -OCH3 aia NO2 F F F F
A:
Q: 3. Indicate: a. The position of the IR peak of the C=O group of benzaldehyde and dibenzalacetone.…
A:
Q: In an aqueous solution of a certain acid the acid is 11.% dissociated and the pH is 1.96. Calculate…
A:
Q: 4. For the following electrocyclic ring closure reaction a. Give the curved arrow-pushing and the…
A: Explanation: It seems like you've presented a set of questions related to an electrocyclic ring…
Q: C) H3C CH2 CH3 LiBH4 Et₂O, 0 °C H3C then H3O+ work up provid provide the drawing of stereochemical…
A: All the two reactions are electrophilic addition reactions of carbobyl compounds.To find out the…
Q: Three atoms (I, II, and III) are indicated in the molecule shown below. O || ||| What is the…
A:
Q: show how for molecule C11H14O2 the following fragments may be obtained given the mass spec highest…
A:
Q: Question 18 Identify the product (or multiple products) for each of the following reactions: 1. 03…
A: Lindlar's catalyst are used for the hydrogenation of alkynes to form cis alkenes. The reduction does…
Q: TRANSMITTANCEI Compound A has 4 carbons and one heteroatom (not C or H). a. Please give appropriate…
A: The objective of the question is to identify the structure of compound A, its reaction scheme with…
Q: What is the partial pressure (in atm) of gasoline if 1 gallon of gasoline completely evaporates into…
A: The objective of this question is to calculate the partial pressure of gasoline in a room after 1…
Q: A combinatorial library was built by reacting the carbonyl compounds in group A with the Grignard…
A: 1. When a halide reacts with Mg metal, it forms Grignard reagent. The given halides will form the…
Q: 6. A solution of 0.1000 M HCl was used to titrate 25.00 mL of Na2CO3 (0.0800 M). Calculate the pH of…
A: The objective of this question is to calculate the pH of the solution at different stages of the…
Q: 2. [Protecting groups] Provide the plausible step-by-step synthesis to produce the product (shown on…
A: (a)Starting Material:The starting material has an OH group attached.Desired Product:The product has…
Q: Two sets of ionizations are shown in the tables below. Complete the tables by ordering each set of…
A: Ionisation energy is the amount of energy required to remove the electron from an isolated gaseous…
Q: Calculate to 2 decimal places the concentration of the conjugate acid and conjugate base for a total…
A:
Q: 6. A solution of 0.1000 M HCl was used to titrate 25.00 mL of Na2CO3 (0.0800 M). Calculate the pH of…
A: The objective of this question is to calculate the pH of the solution at different stages of the…
Q: 10. Show how you would accomplish the following transformation. More than one step is required. Show…
A: First aldehyde oxidized into acid then react with alcohol to give ester
Q: c) Draw the structure of each of the following complexes. Clearly show the stereocentre of central…
A: Structure of complex compound:A complex compound is a molecular structure with a central atom…
Q: Will a precipitate form when 50.0 mL of 2.0 x 10-2 M NaF is mixed with 10.0 mL of 1.0 × 10-2 M…
A: Precipitate formation between two solutions when we mixed to them depends up on the solubility…
Q: Question 8 Please predict the products for each of the following reactions: 1.03 2. H₂O NaNH, 6 Pd…
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: 4. What is the product of the following reaction? Hint: This is a SN1 reaction. OH HCI ?
A: A substitution reaction, a nucleophile replaces a leaving group in substrate to give a new product.…
Q: What mass of NiCO, will dissolve in 1.4 L of 0.502 M NaCO₂ solution? K of NICO, is 6.6×10 9 sp Mass…
A:
Q: Draw the products formed when each ester is treated with lithium hydroxide and water.
A: Ester is a condensation product of alcohol and carboxylic acid. The general reaction in ester…
Q: Which of the following has the (R) configuration? a. t of CO₂H H₂N!! C CH₂OH H b. CO₂H H₂N H CH₂OH…
A: The objective of the question is to determine which of the given compounds has the (R)…
Q: need help show charges and hydrogens on the oxygen or hydrogen atoms where appropriate .
A: The objective of the question is to identify the species found in the mixture.
Q: draw the major product of this reaction. ignore inorganic products ( Step by step answer please)
A: Thank You.
Q: Draw the major organic product(s) of the following reaction. 1 eq. NaNH2, NH3(1) H-CEC-H…
A: When ethyne reacts with NaNH2 (sodium amide) in liquid ammonia (NH3), it forms sodium acetylide…
Q: Draw the major product of the following sequence of reactions. If the reaction is likely to give you…
A: In the given reaction sequence the substrate is toluene and it is reacted with two sets of reagents.…
Q: If 30 g of acetone was combusted with a stoichiometric equivalence of oxygen, how many grams of…
A: The objective of the question is to find out the amount of oxygen required to combust 30 g of…
Q: HD OH NalO4 Et OH Me DCM OH OH (PCy3)2Cl2RUCHPh (Grubbs Cat.) Pd(PPh3)4, PhBr K2CO3 Toluene, 80 C…
A:
Q: These are synthesis questions. You need to show how the starting material can be converted into the…
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: A 1000 mL solution contains 0.500 M K,CrO, and 0.500 M Na₂SO. A concentrated Ba(NO3)2 solution is…
A: The question is based on the concept of solubility product principle.It states that when a weak…
Q: Question 15 Identify the product (or multiple products) for each of the following reactions: A B C D…
A: Step 1: Step 2: Step 3: Step 4:
Q: A Please predict the products for each of the following reactions: Hint: Treat D (deuterium atom)…
A:
Q: If NaOH is combined with CH3OCH 3, which of the following reaction diagrams best represents the…
A: In the given question we need to find out the reaction diagram of NaOH with CH3OCH3.
Q: Draw structural formulas for the major organic product of the reagents shown. CH3 Give detailed…
A:
Q: Three atoms (I, II, and III) are indicated in the molecule shown below. || III What is the oxidation…
A:
Q: Show work, thank you!
A: the equation for the dissociation of SrF2 is: SrF2(s) <=> Sr2+(aq) + 2F-(aq) the ionic…
Q: Draw structural formulas for the major organic product(s) of the reaction shown below. OCH3 • •…
A: Step 1: Step 2: When anisole is nitrated with a mixture of HNO3 and H2SO4 it gives a mixture of…
Q: Identify the two unique common elementary steps in the above mechanism, by sorting them in the order…
A: Detailed explanation: Electrophilic addition: This mechanism involves the addition of an…
Q: Classify the following substituents according to whether they are electron donors or electron…
A: Inductive effect is defined as the permanent displacement of electron density towards more…
Q: Draw one of the two enantiomers of the major product from this reaction. Use a dash or wedge bond to…
A: Step 1:Step 2: Step 3: Step 4:
Q: 2) Consider the following reaction and associated rate law: X (g) + Y(g) → Z Rate = k[X] [Y] b Use…
A:
Q: please help me draw and understand which is the enantiomer of this product
A: For the given product, the enantiomer is drawn below, which should be a mirror image of the parent…
Q: In each row check off the boxes that apply to the highlighted reactant. reaction Ag* (aq) + 2…
A: Ag+ act as Lewis acid (as accepts electron from NH3). CH3NH2 acts as Bronsted-Lowry base.(as…
Q: H3C I HH H3C NH2 I H3C CH3I H HH Na N :: O Select to Add Arrows NaOH, H2O
A: Amines are converted into quaternary ammonium salts by reacting with alkyl halides..The treatment of…
Q: This question concerns part 1 of this lab. You need to make a buffer solution by mixing both a weak…
A: Given,molarity of sodium acetate = 0.100 Mvolume of sodium acetate = 5.00 mLpH of the solution =…
Q: 20. A vial of anesthetic contains 10 mL. The pharmacist asks that you add 30 mL of distilled water…
A: The problem involves finding the total volume of a prepared medication after adding distilled water…
Q: PLs help me solve the given question, explain it, make sure its correct pls thanks
A: Given:…
Q: Draw a skeletal structure for the product formed when the following alcohol is dehydrated with H2SO4…
A: We have been given an alcohol and it is asked to find out dehydrated product.
Step by step
Solved in 2 steps with 1 images

- Draw the missing organic structures or select the missing reagents in the following multistep synthesis. Ignore any inorganic byproducts formed.When some sugars dissolve in water they spontaneously undergo changes in optical rotation called mutarrotation. The Mutarrotation of D-glucopyranose is catalyzed by acid and bases. 2-Hydroxypyridine is a more effective catalyst than phenol and pyridine for this reaction because: a.Both oxygen and N in 2-hydroxypyridine act as bases increasing the rapid interconversion of sugar b. The OH of 2-hydroxypyridine serves as the base while the current N as the acid. c. 2-hydroxypyridine acts both as a base to remove the proton from the hydroxyl group in the hemiacetal and as an acid to provide a proton to the oxygen in the hemiacetal. d.Phenol and pyridine are very expensive.One of the later steps in glucose biosynthesis is the isomerization of fructose 6-phosphate to glucose 6-phosphate. Propose a mechanism, using acid or base catalysis as needed.
- please explain why this happends, im having trouble in this section. Part A Chymotrypsin, an enzyme that hydrolyzes peptide bonds in proteins, functions in the small intestine at an optimum pH of 7.7 to 8.0. How is the rate of the chymotrypsin-catalyzed reaction affected by each of the following conditions?Draw the product of the reactions of succinic anhydride with each of the listed reagents. Assume in all cases that the reagent is present in excess and an aqueous workup takes place after each reaction. A. Reaction with (CH3)2CHOH. \table[[Select,Draw,Templates More,,],[/,//,I//,H,Erase]] B. Reaction with NH3. \table[[Select,Draw,tes More,Erase],[/,//,III,C,H,N,0]] Question Source: Vollhardt 7e0 Organic Chemistry: Structure And Function Publishęr: W. Freemar C. Reaction with phenylmagnesium bromide in THF, followed by H+,H2o. \table[[Select,Drav,v T,lates,,,Erase],[1,II,III,C,H,0,]] D. Reaction with LiAlH4 in ether, followed by H+,H2O \table[[Select,Draw,Templates More],[/,//,III]] \table[[3,0]] \table[[,2,QProvide a multistep synthesis.
- does structure E represent fructofuranose? explainWhich of the following substituent is an ortho/para-directing activator and an ortho/para-directing deactivator? a amino group b halogen group c nitro group d carboxyl groupDraw the missing organic structures in the following multistep synthesis. Ignore any inorganic byproducts formed.
- Polyunsaturated fatty acids (PUFA's) in oils 0.g. sunflower oil are prone to rancidity because they contain conjugated double bond systems which are easily oxidized in the presence of light or peroxides. BHT Is used as an antioxidant in oils to prevent oxidative rancidity. How do BHT prevent oils from reacting with peroxides so that they do not become rancid? Show the mechanism.Co2+ catalyzes the hydrolysis of the lactam shown here. Propose a mechanism for the metal-ion catalyzed reaction.Place a box in fhe functional groups responsible for its solubility (Hydrophilic or Lipophilic)

