Q: A chemist makes 460. mL of barium chloride (BaCl2) working solution by adding distilled water to…
A:
Q: i Draw the missing curved arrow notation. 120 ད Cz + :CI: H-CI: H C N O S F P P Cl Br I
A: Step 1:This reaction is the friedel craft alkylation reactin of aromatic compound.This reaction…
Q: Please correct answer and don't use hend raiting
A:
Q: Using ethyl bromide and formaldehyde as your only source of carbons, show the complete synthesis of…
A: Step 1: Write the objectiveThe objective of the question is to predict the synthesize the following…
Q: Sulfuric acid is essential to dozens of important industries from steelmaking to plastics and…
A: World production of sulfuric acid = Mass of solid sulfur = 3.1 kgPressure of oxygen gas = 10.0…
Q: How would you synthesize the following substances from benzaldehyde and reagents from the table? In…
A: The given target molecules are shown below.The given reagents are shown below.We have to synthesize…
Q: Calculate the pH of a solution made by mixing 100.0 mL of 0.049 M NH3 with 100.0 mL of 0.121 M HCI.…
A: Answer:Number of moles of any specie in the solution is equal to the product of its molar…
Q: Draw structures for the carbonyl electrophile and enolate nucleophile that react to give the aldol…
A: Aldol condensation reaction is a reaction between two carbonyl compounds to form an alpha-beta…
Q: 0.14 M CH3NH2 (where K for CH3NH2 is 4.4 x 10-4) Express your answer using two decimal places. pH =…
A: The objective of the question is to calculate the pH of a 0.14 M CH3NH2 solution. CH3NH2 is a weak…
Q: Phosphate buffers are commonly used to mimic biological systems. Given that phosphoric acid is a…
A: Step 1:Phosphate buffers are indeed commonly used in biological research and experimentation due to…
Q: Но -OH
A: Ketone react with HCN to give cyanohydrin then further hydrolysis to give alpha hydroxy acid
Q: Draw structural formulas for the major organic product(s) of the reaction shown below. CI • • •…
A: Step 1:Step 2: Chlorobenzene can undergo electrophilic substitution reaction with Br2 in presence…
Q: What would be the major product of the following reaction? i KMO, OH, heat ii. HO i. Er P ? ii H₁₂D
A: The question is based on organic reactions.We need to identify the product and explain its…
Q: 44 ut of Classify each pair of molecules as diastereomers, enantiomers, or same. C H 'H H CI H, H H…
A: The objective of the question is to classify the given pair of molecules as diastereomers,…
Q: None
A:
Q: A sheet of gold weighing 9.5 g and at a temperature of 16.5 °C is placed flat on a sheet of iron…
A: Approach to solving the question:Please see attached photos for detailed solutions. Thank you.…
Q: Draw the product of the reaction below. Ignore inorganic byproducts. OH H. H H HO- -OH OH H OH H…
A:
Q: Identify the three steps from the following options
A: Step 1: Step 2: Step 3: Step 4:
Q: 16. Draw the major product of the reaction below? SO3, H2SO4 major product PTO
A: Step 1:As we know that tert butyl group ortho para directing group so income Electrophile attached…
Q: Predict the major organic product for the following reaction sequence. Be sure your answer accounts…
A:
Q: For a certain chemical reaction, the standard Gibbs free energy of reaction at 20.0 °C is -50.9 kJ.…
A:
Q: A chemist must prepare 500.0 mL of potassium hydroxide solution with a pH of 12.80 at 25 °C. She…
A: The pH of KOH solution = 12.80V = 500.0 mL
Q: For each chemical reaction in the table below, decide whether the highlighted reactant is a…
A: Bromsted lowry acid Which donates H+Bromsted lowry base which accept H+
Q: 4 HBr(g) + O2(g) 2 Br(g) + 2H₂O(g) Also, a chemist finds that at a certain temperature the…
A: Hydrogen bromide and oxygen react to form bromine and water,4 HBr(g) + O2(g) → 2 Br2(g) + 2 H2O(g)
Q: Predict the major organic product for the following reaction sequence. Be sure your answer accounts…
A: Step 1: Step 2: Step 3: Step 4:
Q: 12. In each box write: a) the formula and, b) the names of the compounds formed in the following…
A: Answer:Ionic compound is made of ions and electrically neutral in nature, so, cation and anion…
Q: Calculate the solubility at 25 °C of CuBr in pure water and in 0.33 M KCN. You'll probably find some…
A: Given:Concentration of KCN =0.33 MWe have to calculate the solubility of CuBr in pure water and in…
Q: just need standard error for K
A:
Q: 13. Write the correct formula for each of the following compounds: A. diphosphorus pentachloride: C…
A:
Q: Make the following changes on the molecule in the drawing area below: 1. First make sure this is…
A:
Q: Question 1. Calculate the catalyst loading (in mol %) employed in the following experiment and the…
A: The objective of the question is to calculate the catalyst loading in mol % and the turnover number…
Q: The base protonation constant K₁ of lidocaine (C14H21 NONH) is 1.15 × 10 Calculate the pH of a 0.88…
A: Step 1: The base lidocaine (C14H21NONH) is a weak base, which is obvious from its small value of the…
Q: How would you prepare the following compound using either an acetoacetic ester synthesis or a…
A: The objective of this question is to provide the preparation method for the given reaction through…
Q: 1. For each of the following, show the predominant polymer structure that results. Include all end…
A: Let's tackle this chemistry problem involving polymer structures. The image you provided contains a…
Q: 6. (13 pts) A solution of 0.1000 M HCl was used to titrate 25.00 mL of Na2CO3 (0.0800 M). Calculate…
A: Given data:Reaction: (A - {Products})Rate law: ({Rate} = -{d[A]}/{dt} = k[A]^x)Ka1 = (4.46 *…
Q: Rank the following in order of increasing reaction rate with Br2 (slowest to fastest)
A: The objective of the question is to rank the given alkenes in order of increasing reaction rate with…
Q: What is the IUPAC name of the following compound? NH, 0 CH3CHCHCOH CH₂
A: Write the name of the compound according to the given compound Total number of carbon = 4 Functional…
Q: Draw the organic product(s) of the following reaction. H-CEC-H 1 eq. NaOH/H2O
A:
Q: Consider the reaction below that has a Keq of 7.55.10^-2. 3.1 mol of A and 8.2 mol of B are added to…
A: The reaction is :A + 2B <=> CKeq = 7.55*10-23.1 mol of A8.2 mol of BV = 7 L
Q: Please correct answer and don't use hend raiting
A: Step 1: Step 2: Step 3: Step 4:
Q: IN O OCH2CH3 NH2
A: Hydrolysis are a class of reactions where water is used to break other Molecular bond.Hydrolysis can…
Q: 20. A vial of anesthetic contains 10 mL. The pharmacist asks that you add 30 mL of distilled water…
A: The problem involves finding the total volume of a prepared medication after adding distilled water…
Q: During the reduction experiment you add 1.5 mL of cyclohexanone (MW-98.15 g/mol, density 0.948 g/…
A: The objkective of the question is to calculate the percent yield.
Q: None
A: Step 1: understanding concept Step 2: find the formula Step 3: put the values Step 4: get the…
Q: Draw the major product of this reaction. Ignore inorganic byproducts. NH2 1. CHзI (excess) 2. Ag2O,…
A: Step 1: Step 2: Step 3: Step 4:
Q: What is ∆G° for the reaction CaCO₃(s) → CaO(s) + CO₂(g)? CaO(s): --603.3 CO2(g): -394.4 CaCO3…
A:
Q: 1. How many microliters of a 25 \mu M MgCl2.6H2O solution would be needed to provide 152.48 ng of…
A: The objective of the question is to find out the volume of a 25 µM MgCl2.6H2O solution required to…
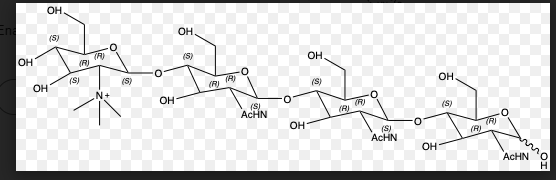
Q: 1. Draw L-Glucose in the Fisher projection and Haworth diagram 2. Connect D-glucose and D-fructose…
A:
Q: b) OTf N-Pt-OTf + PMe3 OTf + 3. Predict the products of the following reactions: a) CI 72- CI-Pt-CI…
A: The objective of this question is to predict the products of reactions involving coordination…
Q: dont provide handwriting solution ...
A: The objective of the question is to identify the reagent(s) that would be most likely to complete…
please help me draw and understand which is the enantiomer of this product
Step by step
Solved in 2 steps with 1 images

- compared to HCO3-, H2co3 has a A Stronger conjugated base B Weaker conjugated base C Higher ka D Lower ka E Both b and c4-Methylphenol, CH3C6H4OH (pKa 10.26), is only slightly soluble in water, but its sodium salt, CH3C6H4O- Na+, is quite soluble in water. In which solution(s) will 4-methylphenol dissolve? Q.) Aqueous NaOHThe key step in a reported laboratory synthesis of sativene, a hydrocarbon isolated from the mold Helminthosporium sativum, involves the following base treatment of a keto tosylate. What kind of reaction is occurring? How would you complete the synthesis?
- Complete each acid-base reaction and predict whether the position of equilibrium lies toward the left or toward the right. (a) CH3CCH+CH3CH2ONa+CH3CH3OH (b) CH3CCCH2CH2OH+Na+NH2NH3(l)1. which compound is more soluble in water: CH3 (CH2)3 COO- Na+ Or CH3(CH2)3 COOH 2. which compound is more soluble in an organic solvent: CH3 (CH2)3 COO- Na+ Or CH3(CH2)3 COOHEthyl butyrate, CH3CH2CH2CO2CH2CH3CH3CH2CH2CO2CH2CH3, is an artificial fruit flavor commonly used in the food industry for such flavors as orange and pineapple. Its fragrance and taste are often associated with fresh orange juice, and thus it is most commonly used as orange flavoring. It can be produced by the reaction of butanoic acid with ethanol in the presence of an acid catalyst (H+H+): CH3CH2CH2CO2H(l)+CH2CH3OH(l)H+⟶CH3CH2CH2CO2CH2CH3(l)+H2O(l)CH3CH2CH2CO2H(l)+CH2CH3OH(l)⟶H+CH3CH2CH2CO2CH2CH3(l)+H2O(l) A chemist ran the reaction and obtained 5.40 g of ethyl butyrate. What was the percent yield, The chemist discovers a more efficient catalyst that can produce ethyl butyrate with a 78.0% yield. How many grams would be produced from 7.45g of butanoic acid and excess ethanol?
- Ethyl butyrate, CH3CH2CH2CO2CH2CH3CH3CH2CH2CO2CH2CH3, is an artificial fruit flavor commonly used in the food industry for such flavors as orange and pineapple. Its fragrance and taste are often associated with fresh orange juice, and thus it is most commonly used as orange flavoring. It can be produced by the reaction of butanoic acid with ethanol in the presence of an acid catalyst (H+H+): CH3CH2CH2CO2H(l)+CH2CH3OH(l)H+⟶CH3CH2CH2CO2CH2CH3(l)+H2O(l) Part A Given 7.30 gg of butanoic acid and excess ethanol, how many grams of ethyl butyrate would be synthesized, assuming a complete 100%% yield? Express your answer in grams to three significant figures. Part B A chemist ran the reaction and obtained 5.95 gg of ethyl butyrate. What was the percent yield? Express your answer as a percent to three significant figures. Part C The chemist discovers a more efficient catalyst that can produce ethyl butyrate with a 78.0%% yield. How many grams would be produced from 7.30 gg of…Ethyl butyrate, CH3CH2CH2CO2CH2CH3CH3CH2CH2CO2CH2CH3, is an artificial fruit flavor commonly used in the food industry for such flavors as orange and pineapple. Its fragrance and taste are often associated with fresh orange juice, and thus it is most commonly used as orange flavoring. It can be produced by the reaction of butanoic acid with ethanol in the presence of an acid catalyst (H+H+): CH3CH2CH2CO2H(l)+CH2CH3OH(l)H+⟶CH3CH2CH2CO2CH2CH3(l)+H2O(l) Given 8.45 gg of butanoic acid and excess ethanol, how many grams of ethyl butyrate would be synthesized, assuming a complete 100%% yield? Express your answer in grams to three significant figures. A chemist ran the reaction and obtained 5.50 gg of ethyl butyrate. What was the percent yield? Express your answer as a percent to three significant figures. The chemist discovers a more efficient catalyst that can produce ethyl butyrate with a 78.0%% yield. How many grams would be produced from 8.45 gg of butanoic acid and excess…Label each statement as True or False. a. CH3CH2+ is the conjugate acid of CH2=CH2. b.CH3CH2− is the conjugate base of CH3CH2+. c. CH2=CH2 is the conjugate base of CH3CH2−. d. CH2=CH− is the conjugate base of CH2=CH2. e. CH3CH3 is the conjugate acid of CH3CH2−.
- Rank the following alcohols from strongest to weakest acid. b. Explain the relative acidities. CH2 = CHCH2OH CH3CH2CH2OH HC=CCH2OH1.(a) Which of the following groups has the LOWEST IUPAC priority?(A) CH3 (B) NH2 (C) OH (D) COOH (E) Br (b)Which of the following corresponds to the strongest acid?(A) (CF3)3C-COOH (B) (CF3)3 C-OH(C) CH3COOH (D) CH3OH(E) HOCH2CH3Name the following molecules, then rank their basicities in decreasing order (1 = most basic, 3 = least basic).





