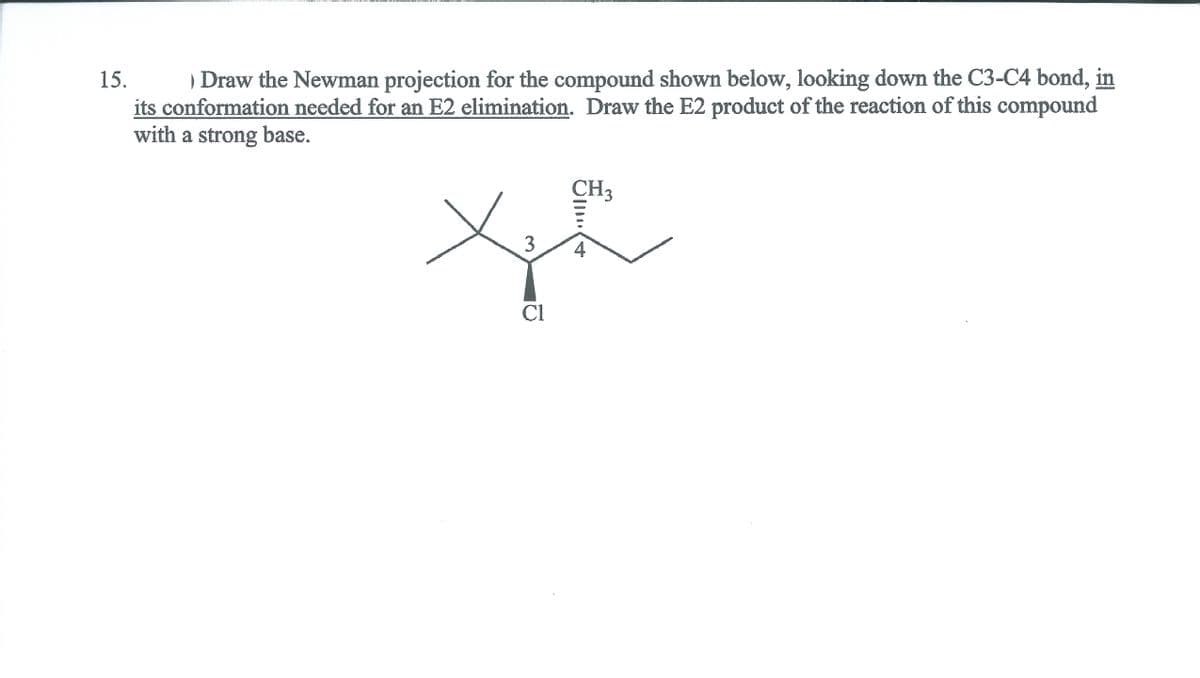
) Draw the Newman projection for the compound shown below, looking down the C3-C4 bond, in its conformation needed for an E2 elimination. Draw the E2 product of the reaction of this compound with a strong base. 15. CH3 Cl
Q: When alkyl fluorides are reacted with strong bases, it was found out that the mechanism does follows…
A: E1cb reactions are elimination reactions in which the first step is the loss of proton and making of…
Q: e. Of the three compounds shown below, Compound A does not undergo E2 elimination, Compound B…
A: Compound that do not have any kind of symmetry are chairal compounds . For E2 reaction there must…
Q: e. Of the three compounds shown below, Compound A does not undergo E2 elimination, Compound B…
A: Thankyou for submitting an excellent question but as per our guidelines only one question was…
Q: Consider the E2 elimination of 3-bromopentane with hydroxide. :Br CH3 product + Br+ H2O H CH3CH2…
A: E2 mechanism is a bimolecular elimination method. It is also known as concerted mechanism because…
Q: Redraw the structure of the starting material in the required conformation (using a Newman…
A: We are given an alkyl halide which undergoes elimination reaction with NaOH giving an alkene as a…
Q: Which of the two isomers would you expect to undergo E2 elimination faster? trans-1-bromo-…
A: Out of these two, trans-1-bromo-4-tertbutylcyclohexane will undergo faster E2 elimination.
Q: Consider the elimination reaction shown here, which CH3 H3C Base produces a mixture of…
A:
Q: Draw an Energy diagram for an E1 reaction: (CH3)3CI + H2O ⇢ (CH3)2C=CH2+ H3O+ + I−
A: E1 is the unimolecular elimination reaction, the removal of HX from the substrate leads to the…
Q: -CF3 (7) Please select the major product in the nitration to CF3 CF3 NO2 ÇF3 ÇF3 NO2 A. В. C. `NO2…
A: Given organic compound is (Trifluoromethyl)benzene. It is a deactivating substituent. Deactivating…
Q: Although there are nine stereoisomers of 1,2,3,4,5,6-hexachlorocyclohexane, one stereoisomer reacts…
A: To find: The structure of that stereo isomer with explanation
Q: Both compounds A and B below give the same carbocation intermediate C in the SN1 reaction. However,…
A: In this question, we will Identify which Compound ( A or B ) give carbocation intermediate ( C )…
Q: With 2° or 3° alkyl groups bonded to the ether oxygen, the C—O bondis cleaved by an SN1 mechanism…
A: The replacement of any atom or group of atoms by a nucleophile is known as nucleophilic substitution…
Q: Draw one of the two enantiomers of the major product from this reaction. Use wedge and dash bonds to…
A:
Q: What is the product of the following [1,3] sigmatropic rearrangement, A or B?
A: The above thermal isomerization a [1,3] shift through apperantly forbidden proceeds with inversion…
Q: Draw an energy diagram for an E2 reaction: (CH3)3CBr + −OH →(CH3)2C=CH2 + H2O + Br−
A:
Q: Draw an alkyl bromide with proper stereochemistry that can be used to synthesize the given alkene as…
A: Answer: To form the desired alkene we need a strong non-bulky base that attacks on alkyl bromide…
Q: 2. Arrange these carbocations in order of increasing stability. CH2 CH3 -CH3 C. b. a.
A: Stability of carbocation can be compared using hyperconjugation effect. Carbocations with having…
Q: Be sure to answer all parts. Given that an E2 reaction proceeds with anti periplanar…
A:
Q: Taking into account anti periplanar geometry, predict the major E2 product formed from each starting…
A: Introduction : We have to tell the major eliminated product formed .
Q: What is the major product(s) for the following three-step reaction sequence. I and its enantiomer…
A: Given : We have to make products for the following given reaction.
Q: 6. Draw the most stable enol tautomer of 8-methyleycloocta-3,5-dien-1-one.…
A: Ketone undergoes tautomerization to form enol.
Q: 5.) For the following molecules V-Z, please circle which organic molecules would function well as…
A: Dienophiles- The alkene which has an electron-withdrawing group is known as Dienophiles. The…
Q: Given that an E2 reaction proceeds with anti periplanar stereochemistry, draw the products of each…
A: Elimination reactions: Organic reactions contain the removal of a group of atoms from a molecule by…
Q: Draw the major product formed when the structure shown below undergoes E1 elimination in benzene…
A:
Q: CI КОН ELOH H Нeat
A:
Q: Which of the ff. halides will most probably react via an SN1 mechanism? a) CH3CH=CHCH2Cl b)…
A: Given Which of the ff. halides will most probably react via an SN1 mechanism? a)…
Q: Draw the starting structure that would lead to this major product (and its enantiomer) under these…
A: We have to draw the starting structure for the following given product (and its enantiomer) as…
Q: Which isomer reacts more rapidly in an E2 reaction: cis-1-bromo-4-tert-butylcyclohexane or trans-1-…
A: E2 elimination reaction : It is a type of elimination reaction that is mostly observed in the case…
Q: Given that an E2 reaction proceeds with anti periplanar stereochemistry, draw the products of each…
A: Ethoxide ion is a strong base and E2 mechanism is favored by strong, negatively charged bases. It…
Q: 4. Rank the following leaving groups from best to worst. OH2 -OH -CH3 -NH2 LG LG LG LG LG A B C D E
A: Ability to leave of a leaving group depends upon: Electronegativity of the leaving group: A high…
Q: Draw all constitutional isomers formed in each E2 reaction, and predict the major product using the…
A: Since we only answer up to 3 sub-parts, we’ll answer the first 3 sub-parts. Please resubmit the…
Q: Although there are nine stereoisomers of 1,2,3,4,5,6hexachlorocyclohexane, one stereoisomer reacts…
A: The one stereoisomer of 1,2,3,4,5,6hexachlorocyclohexane, reacts 7000 times more slowly than any of…
Q: Qustion below attached .
A: Hello. Since your question has multiple sub-parts, we will solve first three sub-parts for you. If…
Q: If the -CH3 group is an activator and an ortho-, para- director (directs the position of the…
A: -CH3 group is an electron-donating group because of the + I effect .
Q: 1. As we've seen, SN1/E1 reactions can be quite messy and can generate multiple products. Consider…
A: Alkyl halide gives elimination and substitution reactions. Both E1 and SN1 reactions proceed by the…
Q: Draw the major organic product of the indicated reaction conditions. Be sure to clearly depict the…
A:
Q: Draw the main organic products of the reaction. Indicate the stereochemistry, including all hydrogen…
A:
Q: 3. The haloalkane below is heated in a solution of ethanol and undergoes SN1 and EiS a) Draw the…
A: Note: The above alkyl halide is heated with ethanol (weak nucleophile) results E1 products as major…
Q: One of these molecules reacts faster via an E2 reaction. Draw the products from each E2 reaction.…
A:
Q: Give the systematic name for reactant A. and the final product B. in this synthesis. Specify any…
A: In the first step of the reaction, the alkyne forms a nucleophile and gives an SN2 reaction, in the…
Q: Draw the major organic product of the indicated reaction conditions. Be sure to clearly depict the…
A: In given reactant We have, amide function and alkyne function. And given reagent is lindlar…
Q: 4. Each of the following alkyl bromides gives only one product in via an E2 mechanism. Provide the…
A:
Q: major and minor alkenes formed from the E2-elimination of cis-1
A: More substituted alkene results as a major product whereas less substituted alkene would be a minor…
Q: Draw all the possible chair-like conformations of compounds 1-2. In the presence of a strong base,…
A: In this question we have clarify the proper reason for the formation of more stable product by the…
Q: H3C CH3 IBr KO-t-Bu ID HO-t-Bu а. CH3 NaOEt b. Ph-c-Br CH3 c. Show the curved arrow mechanism for…
A:
Q: Why is a substituion (carbocation) more stable for a (OH) more than a teritary substituion?
A: Tertiary carbocation is a more stable carbocation thats why OH substitution is faster for tertiary…
Q: C. Predict the major product of the reaction with sodium methoxide. NaOMe E2 conditions d. Predict…
A:
Q: Draw the Newman projection for the compound shown below, looking down the C3-C4 bond, in its…
A:
Q: 7. For each reaction, circle the correct product. Br N2OCH3 or NaOCH3 or E2 E2
A: A species with a larger size can easily accommodate negative charge. In other words, in a large size…
Q: Write out the two-step sequence that converts benzene to each compound: (a) C6H5CH2CH2CH2CH2CH3 (b)…
A: Converting benzene into C6H5CH2CH2CH2CH2CH3 and C6H5CH2C(CH3)3 follows two steps:- Friedal craft…
Step by step
Solved in 2 steps with 2 images

- Consider the E2 elimination of Compound A, and answer the following questions: (a) Label each stereocenter in Compound A with the correct R or S configuration. (b) Draw the structures of the major product with correct stereochemistry. Assume D and H have the same reactivity.Given that an E2 reaction proceeds with anti periplanar stereochemistry, draw the products of each elimination. The alkyl halides in (a) and (b) are diastereomers of each other. How are the products of these two reactions related?1a. Provide the products/reactants of the SN1 reactions below. Be very careful of the stereochemistry if applicable. (Recall that in SN1 reaction, the reactant’s chirality (aka configuration of the halo-carbon) is converted to both enantiomer – so you might need show both structures with dashed and wedge bonds 1b. Draw the mechanism (use curved arrow) of of EITHER c or d above
- If dehydration of 2-methylcyclohexanol were to occur under an E2 mechanism, draw the chair conformations of 2-methylcyclohexanol that would be necessary for the reaction to occur. 2-methylcyclohexanol is sold as a mixture of cis and trans isomersAlthough there are nine stereoisomers of 1,2,3,4,5,6hexachlorocyclohexane, one stereoisomer reacts 7000 times more slowly than any of the others in an E2 elimination. Draw the structure of this isomer and explain why this is so.which newman projection displays the proper antiperiplanar orientation for an E2 elimination of the reaction sequence below
- The E2 reaction is defined as stereoselective if the reacting alkyl halide contains? The E2 reaction is defined as regioselective if the reacting alkyl halide contains? only one β-hydrogen. two β-hydrogens attached to the same β-carbon atom. ...two β-hydrogens attached to different β-carbon atoms. ...no β-hydrogens.Draw an Energy diagram for an E1 reaction: (CH3)3CI + H2O ⇢ (CH3)2C=CH2+ H3O+ + I−a.Draw three-dimensional representations for all stereoisomers of 2chloro-3-methylpentane, and label pairs of enantiomers. b. Considering dehydrohalogenation across only C2 and C3, draw the E2 product that results from each of these alkyl halides. How many different products have you drawn? c. How are these products related to each other?
- Taking into account anti periplanar geometry, predict the major E2 product formed from each starting material.Draw a stepwise mechanism for the following reaction that illustrates how two substitution products are formed. Explain why 1-bromohex-2-ene reacts rapidly with a weak nucleophile (CH3OH) under SN1 reaction conditions, even though it is a 1° alkyl halide.The Hofmann product is favored by the E2 mechanism when 2-bromo-3-methylbutane is added: A. MeOH B. MeO:- in MeOH C. Nal in acetona D. (CH 3)3 CO:- in (CH 3),COH

