Taking into account anti periplanar geometry, predict the major E2 product formed from each starting material. b. с. d. a. "CH CH3 ČH(CH3)2 ČH(CH3)2
Q: PBR3, py ? Nal 1. ÕH DMF, 0 °C BNOH 2. ? ? 120 °C 'BuONa CH3 NaCN 3. ? ? H3CH2C CI DMF, 0 °C 80 °C
A: Organic reactions are carried out in presence of appropriate reagents. Reagents have their distinct…
Q: Draw the products of the following reactions, including all stereoisomers:
A: Hello. Since the question consists of multiple sub-parts, the first three sub-parts shall be only…
Q: Consider the E2 elimination of 3-bromopentane with hydroxide. :Br CH3 product + Br+ H2O H CH3CH2…
A: E2 mechanism is a bimolecular elimination method. It is also known as concerted mechanism because…
Q: Explain why the reaction of 2-bromopropane with NaOCOCH3 gives (CH3)2CHOCOCH3 exclusively as…
A: The reason why reaction of 2-bromopropane with NaOCOCH3 forms CH32CHOCOCH3, whereas the reaction of…
Q: Arrange the following compounds in their ease of undergoing an SN2 reaction: a. CH3CH2CH2-Cl b.…
A:
Q: a) Explain in detail why reaction of methylcyclopentane with Cl2/light gives a mixture of products…
A: Reactivity of hydrogens : tertiary > secondary > primary. Reactivity of halogens: Chlorine…
Q: CH3 -CH3 CH3
A:
Q: +Which statement(s) is/are true regarding leaving groups? 1. Weak bases make good leaving groups.…
A:
Q: CH3 HBr Peroxides CH3 H3C Br -Br CH3 CH3 C. А. ÇH3 ÇH3 Br CH3 `CH2B D. В.
A:
Q: D ОН ČI
A:
Q: Draw an energy diagram for an E2 reaction: (CH3)3CBr + −OH →(CH3)2C=CH2 + H2O + Br−
A:
Q: Design the synthesis of: (a) CH3C≡CCH2CH3 from HC≡CH and appropriate alkyl halides.…
A: The details solution for this is provided below in attach image.
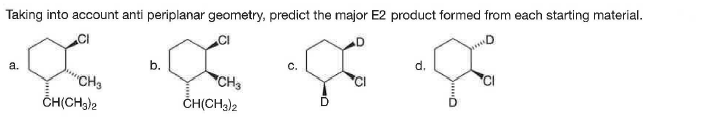
Q: Taking into account anti periplanar geometry, predict the major E2 product formed from each starting…
A: The compounds in which the leaving group and the adjacent hydrogen atom are anti-periplanar to each…
Q: (üi) from NH2 CH3
A: 3) The mechanism of formation of isoquinoline has been outlined below
Q: CH2CI CH2CI CH2CI CH2CI CIH2C CH2CI CH2CI CH2CI A в D I. Which of the heterocycles would be the most…
A: Q2 i) Diels alder reaction refers to a chemical reaction takes place between a conjugated diene and…
Q: 5. Write out the stepwise mechanism, including intermediates or transition state for: CH3O Br OCH3
A: Given: To find: stepwise mechanism with intermediates and transition state.
Q: In the E2 reaction of 2-bromopentane with sodium tert-butoxide, 1-pentene is favored over 2-…
A:
Q: What is the major product of the following reactions? Disregard stereoisomers:
A: Here in these reactions, NBS is used which is known as N-Bromosuccinimide. N-Bromosuccinimide or…
Q: CH2l2 ? Zn/Cu The starting material necessary for this transformation is: A в D E
A: This reaction is known as Simmons Smith reaction. In this reaction alkene is get converted into…
Q: Arrange the following compounds in their ease of undergoing an SN2 reaction: a) CH3CH2CH2-CI b)…
A:
Q: Peroxides) CH3 CH3 (b) CHCH3 HI ? (a) HBr CH3CH2C=CHCHCH3
A:
Q: In the alkene below, why is HB more easily abstracted by a halogen atom than HẠ ? НА HB A.…
A:
Q: 10. Arrange each set of compounds in order of reactivity towards E2 process: Br Br Br CI (a) (b)
A: In this question, the given set of compounds is to be arranged in the order of reactivity towards E2…
Q: a) b)
A: In photochemistry, we learn about cycloaddition reactions. The one important reaction of this type…
Q: Name each alkene and specify its configuration by the E,Z system
A: While writing the IUPAC name first identify the parent chain and the functional group.
Q: What reagent/reagents is/are necessary to transform the starting molecule into the desired product?…
A:
Q: Br H;C. .CH3 `CH3 H
A: A<C<D<B This the order of decreasing reactivity for E2 elimination
Q: Arrange the following compounds in their ease of undergoing an SN2 reaction: a) CH3CH2CH2-CI b)…
A: Please note- As per our company guidelines we are supposed to answer only one question. Kindly…
Q: но NBS HBr CCI, ether а. b.
A: ->HBr give substitution reaction . ->NBS/CCl4 used for alpha bromination.
Q: Rank the alkyl halides in order of increasing E2 reactivity. Then do the same for E1 reactivity.
A: The factors that influence the order of ( E1 and E2 ) elimination reactions are: 1) The reactivity…
Q: Rank the following carbocations in order of decreasing stability, putting the most s II Multiple…
A: Stability of carbocation is as 3°>2°>1° We can see that I is a 3° Carbocation as the carbon…
Q: Draw all constitutional isomers formed in each E2 reaction, and predict the major product using the…
A: Since we only answer up to 3 sub-parts, we’ll answer the first 3 sub-parts. Please resubmit the…
Q: Internal alkynes are able to selectively form one product when subjected to most reaction…
A: Alkynes are unsaturated hydrocarbons that can carry out electrophilic addition reaction by…
Q: Rank the following in order from slowest to fastest rate of reaction in a Diels–Alder reaction with…
A: Introduction: Diels-Alder reaction is a cycloaddition reaction of diene and dienophile to form a…
Q: Complete these SN2 reactions, showing the configuration of each product.
A: SN2 reaction or bimolecular nucleophilic substitution has following features: Formation of a…
Q: butan-2-ol → butan-2-one, CH3COCH2CH3. NaOCI НОАС EtMgBr H,CrO4 ether or PCC excess NaOCI ТЕМРО…
A: The oxidation of alcohol can be done via oxidizing agents like chromic acid (H2CrO4). Sodium…
Q: Does cis- or trans-1-bromo-4-tert-butylcylohexane react faster in an E2reaction?
A: Cis- or trans-1-bromo-4-tert-butylcylohexane that reacts faster in an E2 reaction has to be…
Q: Explain why α-pyrone reacts with Br2 to yield a substitution product (likebenzene does), rather than…
A: Two compounds can show similar types of reactions only if they have similar properties. Both Benzene…
Q: Taking into account anti periplanar geometry, predict the major E2 product formed from each starting…
A: E2 reactions are the elimination reactions in which the reactant and the reagent or the nucleophile…
Q: Give the Reagents. When finished, choose "true". SH -s-
A: The given reaction is the synthesis of disulfide by S-S coupling from thiol. The reagents used in…
Q: Explain why α-pyrone reacts with Br2 to yield a substitution product (like benzene does), rather…
A: The given compound is aromatic as it involves delocalization of pi-electrons.
Q: The reaction that converts cyclopentanol into cyclopentene is a(n)
A:
Q: 5) Carbocation forms in the reaction of: A) SN1 B) SN2 C) Both D) Cannot determined
A: Given that : We have to identify in which reaction(s) carbocation is formed : A) SN1 B) SN2 C) Both…
Q: Reaction of the cyclic product with a nitron (see below) yields a mixture of heterocyclic…
A: Given reaction:
Q: Diels–Alder reaction of a monosubstituted diene (such as CH2=CH–CH=CHOCH3) with a monosubstituted…
A:
Q: H3C CH3 IBr KO-t-Bu ID HO-t-Bu а. CH3 NaOEt b. Ph-c-Br CH3 c. Show the curved arrow mechanism for…
A:
Q: 6. 7. product in #6 CH3 HNO3, H₂SO4 SnCl₂ H* OH ?
A: 6. Electrophilic substitution: The resonance hybrids for acetophenone is given below. As acetyl…
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

- a. How many s bond orbitals are available for overlap with the vacant p orbital in 1. the isobutyl cation? 2. the n-butyl cation? 3. the sec-butyl cation? b. Which of the carbocations in part a is most stable?Give a clear explanation of how the followingcarbocation is stabilized:Does cis- or trans-1-bromo-4-tert-butylcylohexane react faster in an E2reaction?
- Rank these alkyl halides in order of increasing reactivity in an SN2 reactionSpiderweb chemistry . Reagents needed to get the major product and show stereochemistry.Addition of HBr to allene (CH2=C=CH2) forms 2-bromoprop-1-ene ratherthan 3-bromoprop-1-ene, even though 3-bromoprop-1-ene is formed froman allylic carbocation. Considering the arrangement of orbitals in theallene reactant, explain this result.

