Q: Using appropriate reagents and sequencing, show reasonable synthetic routes for each of the…
A: Given: Benzoquinone from benzene p methylaniline from aniline To find: conversions. Solution:…
Q: Draw structures for the carbonyl electrophile and enolate nucleophile that react to give the enone…
A: enolate attacks the carbonyl electrophile and forms enone Generally this is resembles to aldol…
Q: Draw the major organic product formed when the compound shown below undergoes aromatic bromination.…
A:
Q: Draw the mechanism from benzaldehyde to this compound. Reactants: i) NaBH4 ii)PBr3
A:
Q: Outline methods for the preparation of acetophenone (phenyl methyl ketone) starting from the…
A:
Q: draw the product formed if this compound will undergo acid-catalyzed fischer esterification
A: When 3-isopropyl pentan-5-ol-1-oic acid undergoes acid catalyzed Fischer esterification in presence…
Q: Draw the starting structure that would make this product under these conditions, using all reagents.…
A:
Q: HO Br
A: Conversion of alkyl halide to grignard reagent Attack of grignard reagent as nucleophile to the…
Q: Illustrate the synthesis of triphenylmethanol via Grignard reactions. Select the appropriate…
A: In the synthesis of triphenylmethanol via Grignard reactions, following reagents will be used. 1)…
Q: H3O*, Br₂ NaOEt. wwwmmmm EtOH H3O+ NaOH, H2O
A: Step wise reaction explained:
Q: Draw the structure of the major product formed in the following reaction when the aromatic aldehyde…
A: The methoxide ion acts as a base and deprotonates the methyl acetate to form the enolate ion. The…
Q: Draw the major organic product formed when the compound shown below undergoes a reaction with…
A:
Q: Explain and show step by step how did the product formed when pentanoic anhydride is treated with…
A:
Q: Draw the reactant you would use to make the epoxide shown using a Sharpless epoxidation with t-BUOOH…
A:
Q: What is the first step in the mechanism of the reaction of DIBAL-H with an ester?
A: DIBAL-H = Di isobutyl aluminum hydride Reducing agent It reduce ester to aldehyde (also reduce…
Q: Acetylene reacts with sodium amide in the presence of propyl halide
A:
Q: If NaOH were reacted with the given molecule, could it react with the epoxide to form a vicinal…
A: Epoxide reacts with sodium hydroxide to give vicinal diol and carbonyl groups (only aldehyde and…
Q: -NH- HN H2N NH2 NaOH/ethanol benzil 5,5-diphenylhydantoin (Dilantin)
A:
Q: What is the name of the carbonyl compound that has served as common starting material for synthesis…
A: We have to find out the carbonyl which can form all the compound by reacting with different…
Q: Explain the effect of the structure of the alkyl halide on elimination through E1 and E2.
A:
Q: What starting materials are needed to prepare ar-turmerone using a directedaldol reaction?…
A: A species with a larger size can easily accommodate negative charge. In other words, in a large size…
Q: What is the reaction scheme of the synthesis of the following compound, starting from benzaldehyde?
A:
Q: Show how the m-chlorobenzenesulfonic acid can be synthesized from benzene:
A: The mechanism in which m-chlorobenzenesulfonic acid can be synthesized from benzene has to be…
Q: Is the following statement true or false? If the newly added group in the tetrahedral intermediate…
A: In a nucleophilic acyl substitution reaction, the upcoming nucleophile has to be stronger base than…
Q: What would be the bromination product of phenyl benzoate with Br2/AlBr3? Select one:
A: Answer is explained below.
Q: Draw structural formulas for the product formed by treating each compound with propylmagnesium…
A: Grignard reagent is used for the synthesis of many compounds with different functional groups. The…
Q: Draw the condensation product obtained when the following compound is heated in the presence of…
A:
Q: The Reformatsky reaction is an addition reaction in which an organozinc reagent is used instead of a…
A: a.
Q: Show how the 2-phenylpropene acid can be synthesized from benzene:
A: I consider the synthesis of 2-phenylpropene from benzene. Because a compound called 2-phenylpropene…
Q: C. Draw the structure for the product formed in each of step of the following synthetic sequences.…
A:
Q: 1) Draw AND name the product that would result upon treatment of R-3-methylheptanal with Excess…
A: We’ll answer the first question since the exact one wasn’t specified. Please submit a new question…
Q: Use a sheet of paper to answer the following question. Take a picture of your answers and attach to…
A:
Q: Draw structures for the carbonyl electrophile and enolate nucleophile that react to give the enone…
A:
Q: Complete the synthesis of the following product from benzene and the given reagents. COOH CH3C1 KMnO…
A: The correct answer is given below
Q: Show how the benzyl methyl ether can be synthesized from benzene:
A: To show the preparation of benzyl methyl ether from benzene:
Q: The reaction of a Grignard reagent with a carboxylic acid does not give a secondary alcohol because…
A: The reaction of grignard reagent with a carboxylic acid does not give secondary alcohol because of…
Q: Propose an arrow-pushing mechanism for the following transformation. НО H30+
A: The above transformation is associated with the carbocation. In this transformation hydroxyl group…
Q: What product is formed as a result of nitration reaction of 2,5-dimethylbenzoic acid?
A: The structure of 2,5-dimethylbenzoic acid is as follows:
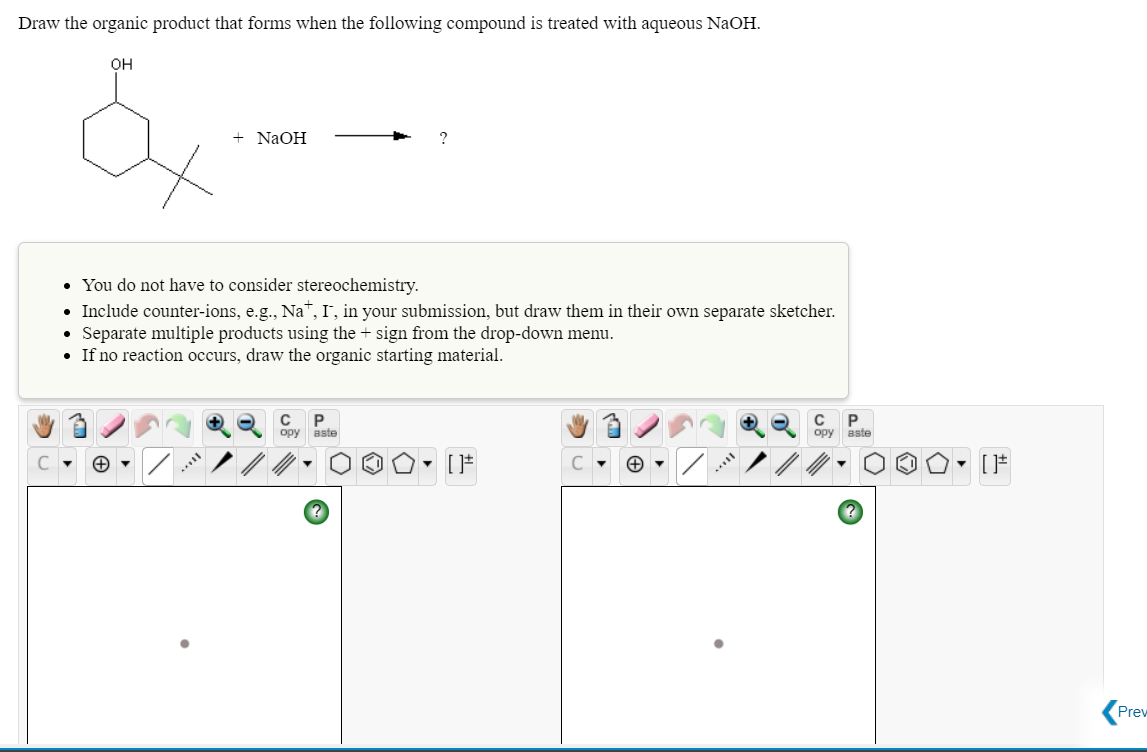
Q: Draw the organic product that forms when the following compound is treated with aqueous NaOH. он +…
A: Note: Alcohols react with sodium hydroxide give alkoxide ion (Here OH is having acidic proton,…
Q: Draw structural formulas for the a,ß-unsaturated aldehyde or ketone and the lithium diorganocuprate…
A: The product formed is given as,
Q: what carbonyl compounds might you start with in a Grignard synthesis of the following compound?…
A: carbonyl compounds might you start with in a Grignard synthesis of the following compound?…
Q: Draw the structure of following compounds:- the (E)-hydrazone of benzaldehyde
A: Structure of benzaldehyde is as follows.
Q: What are the two possible constitutional isomers formed by dehalohydrogenation of the following…
A:
Q: Draw the structure of the product that is formed when the compound shown below is treated with the…
A: ->reagent -1-PBr3,Br2 2-H2O
Q: Which molecule is hydrolyzed most slowly with aqueous NaOH?
A: Hydrolysis reaction is one the important class of organic reactions. In hydrolysis reaction, water…
Q: What product prepared for preparation of p-Nitroaniline.
A: In preparation of p-nitroaniline first p-nitroacetanilide is prepared from acetanilide by nitration…
Q: Show how the benzyl alcohol can be synthesized from benzene:
A: Benzyl alcohol is represented as a -CH2OH group attached to the benzene ring.
Q: What is the net charge of the product made after an organometallic reagent reacts with an epoxide,…
A: After addition of water/acid, the net charge of the product will be zero. Correct answer is 0. For…
Q: Identify the two compounds below that can be prepared from the reaction between a Grignard reagent…
A:
please answer the question correctly
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- Draw the organic product(s) of the following reaction. You do not have to consider stereochemistry. Include cationic counter-ions, e.g., Na+ in your answer, but draw them in their own sketcher. If no reaction occurs, draw the organic starting material. Separate multiple products using the + sign from the drop-down menu.Consider the following aromatic compounds a para-substituted compound?, the most reactive towards EAS? an ortho-substituted compound? does not react with RX, FeX3?Compound X is insoluble in cold KMnO4, Br2 in CCl4, and conc. H2SO4. Compound X is most likely: a. an alkane b. none of these c. an alkene d. an alcohol e. an alkyl halide Indicate which of the ff. statements regarding nucleophilicity is incorrect. F- is more nucleophilic, hence, more reactive towards methyl iodide than Cl-. Second row elements are more nucleophilic than first row elements of comparable basicity. The rate of SN2 reaction may be markedly affected by the nucleophilicity of the attacking atom. Nucleophilicity is the affinity of a nucleophile to an electrophilic carbon Which of the following alkynes can be deprotonated by NaNH2 in liquid NH3? 3-Methylhex-2-yne Pent-2-yne 3-Methylbutyne none of these Hex-3-yne
- PLEASE ASAP Major products for electrophilic addition transformations. Clearly show stereochemistry where appropriate (using wedged or dashed bond lines, or by writing axial or equatorial bonds). If no reaction is expected, write “NR”.Rank the following (in image one, the one with the chlorines) from most to least reactive in SN 1 reactions. How about ranking reactivity in nucleophilic addition (for example, Grignard) reactions in the second image?A hydrocarbon of unknown structure has the formula C8H10. On catalytichydrogenation over the Lindlar catalyst, 1 equivalent of H2 is absorbed. Onhydrogenation over a palladium catalyst, 3 equivalents of H2 are absorbed.(a) How rnany degrees of unsaturation are present in the unknown?(b) How many triple bonds are present?(c) How many double bonds are present?(d) How many rings ar e present?(e) Draw a structure that fits the data.
- You do not have to consider stereochemistry. You do not have to explicitly draw H atoms. Do not include lone pairs in your answer. Draw phosphate groups, when present, as in the question: in basic form and with a phosphorous oxygen double bond.The above synthesis was designed using the Organic Chemistry Roadmaps in the appendix of your textbook. In this synthesis starting with acetylene, reagents from the table are used to carry out the indicated steps (shown in blue). In the box below, draw the structure of compound j. Use the wedge/hash bond tools to indicate stereochemistry where it exists. In cases where there is more than one answer, just draw one. Show stereochemistry in a meso compound. Do not include counter-ions, e.g., Na+, I-, in your answer.(10pts) Compound A, C10H16, was found to be optically active. On catalytic reduction over a palladium catalyst, 2 equivalents of hydrogen were absorbed, yielding compound B, CioH2o. On ozonolysis of A, two fragments were obtained. One fragment was identified as acetic acid (CHCOOH). The other fragment, compound C, was an optically active carboxylic acid, C8H14O2. Write reactions, and draw the correct structures for A-C, explain your answer in detail.
- Please indicate if it will also be an endo or exo product or both (50:50) and why. Thank you.A difficult problem in the synthesis of PGF2α is the introduction of the OH group at C15 in the desired conguration.a. Label this stereogenic center as R or S.b. A well known synthesis of PGF2α involves reaction of A with Zn(BH4)2, a metal hydride reagent similar in reactivity to NaBH4, to form two isomeric products, B and C. Draw their structures and indicate their stereochemical relationship.c. Suggest a reagent to convert A to the single stereoisomer X.Ignoring stereoisomers, draw the two possible enols for butan-2-one (CH3COCH2CH3), and predict which one is more stable.

