Q: When the following equation is balanced properly under basic conditions, what are the coefficients…
A: First we need to balance the given redox reaction by half reaction method. We can balance redox…
Q: Topics] [References] Use the References to access important values if needed for this question. ula…
A: Bronsted Lowry acid base theory, Bronsted Lowry acid is the substance which donate proton while…
Q: H b. . . C. d. i Hlor e. f. 2 H + H3C-C=N 1) NaOCzHg/C,H5OH 2) H₂O/H* NaOC₂H5 base & OH C2H5OH OH…
A: We have to draw the structure of the given products.
Q: 19) (1) Provide the majo Reduction, Clemme (A) Me2N (C) (E) (II) Provide the mechanism
A:
Q: One mixes aqueous NaCl with aqueous AgNO3. NaCl. AgNO3and NaNO3all have appreciable solubility in…
A: First we are write reaction and then use Ksp of AgCl to determine which Compound would precipitate.
Q: 5. A certain mass of oxygen was collected over water when potassium chlorate was decomposed by…
A: When a gas is collected over water some water vapors are also present along with the gas. In such…
Q: Propose an efficient synthesis for the following transformation: do The transformation above can be…
A: Step 1 : Grignard addition to aldehyde FG. Step 2 : OH is converted into OTs (good leaving group)…
Q: The most suitable method of separation of 1:1 mixture of ortho and para-nitrophenols is (a)…
A: 1.) We would check nature of ortho and para nitrophenol to predict method of separation . 2.) To…
Q: Which among the given molecules can exhibit tautomerism? Ph Ph I II III (a) III only (c) Both I and…
A:
Q: molecular geometry
A:
Q: In the reaction shown, the radiation produced is a(an) Ti 51V+ 23 a. gamma ray. b. neutron. O c.…
A:
Q: Which of the molecules shown below would be expected to have the highest water solubility A CH3CH₂OH…
A: Water is a polar solvent and it can dissolve an ionic or polar covalent compound.
Q: 40. Balance the following nuclear reactions, a. 233NpB + ? b. Be + He→→→→→ ? c. P+ ? - → 33P 15- d.…
A:
Q: The half-life of protactinium-234 is 6.69 hours. What fraction of a given amount remains after 26.76…
A:
Q: Covert 0.00823 moles of HCL to g Convert 0.00911 moles Mg to g
A: Using formula: No Of moles (n)= given mass/molar mass No of moles of HCl is 0.00823mole No. Of…
Q: 6. What is the relationship between A and B? HO OH II A) They are constitutional isomers They are…
A:
Q: What is the first step in solid-phase peptide synthesis? 1-washing C-terminus to purify 2-attach…
A: Solid Phase Peptide Synthesis Solid phase peptide synthesis (SPPS), developed by R. B. Merrifield,…
Q: НО ft:1:05:02 НО A О 600 0 В к х 300 2 НО :0 Lama Abdulhai Kajah: Attempt 1 J 600 L (higher) B > C>A…
A: The melting point of longer chains is higher than the shorter chain due to the greater…
Q: If one has measured the concentration based equilibrium constant (Kc) to calculate the…
A: Kp = Kc (RT)∆n R is ideal gas constant, T is temperature, ∆n = difference of reactant and product…
Q: Write a balance net ionic equation for the following redox reaction that takes place in a basic…
A: The reaction in which both oxidation and reduction reaction takes place is known as redox reaction.…
Q: Use the temperature and atmospheric pressure data to identify which of the following bodies of water…
A:
Q: An external water pipe is built of aluminum (Al). In order to prevent corrosion, the nails that hold…
A: In order to prevent corrosion of aluminium metal pipe, the nails must be made of metal with more…
Q: How many coulombs of charge are necessary to produce 341 g of Al from Al³+ (aq)? (F= 96,500 C/mol) 1…
A:
Q: Tc-99m is commonly used in nuclear medicine. It has a half-life of 6 hours. The radioactivity dose…
A:
Q: Write the chemical formula of the following complex ions. formula 0 name hexaamminechromium(III)…
A:
Q: 8. Which one of the following will be most acidic and why? 2-Chlorobutanoic acid, 4- Chlorobutanoic…
A:
Q: The arrangement of ions, atoms or molecules in crystals are best determined by which instrumental…
A: The arrangement of ions, atoms or molecules in crystals are best determined by x-ray diffraction…
Q: Which alkyl halide in each set is more reactive in an SN2 reaction with hydroxide ion? or Br 오아 or…
A:
Q: KBr AgNO3 Ej: KBr + AgNO3 →→KNO3 AgBr Na2CO3 Cas NHẠCH
A: First we would write products by double displacement reaction . Then using solubility rules, we…
Q: B. Carboxylic acid
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: For the reaction A+B=C+D, various initial rate measurements where taken using A and B only, and C…
A:
Q: [Review Topics] Draw the product(s) of the following reactions. H₂C -CEC-CH3 Nal NH3(l) . Consider…
A:
Q: What is used as a form of basis when reading the output IR Spectroscopy? Choose one answer only. A.…
A: What is used as a form of basis when reading the output IR Spectroscopy ?
Q: view - Black X P12.5: Melting, Fre X PExercises X P Answers to Odd- X Email-Jessica Sa X C Solve 30.…
A: Given, 61. The heat required to convert 2.55 g. of water at 28.0 °C to steam at 100.0 °C is:
Q: i need help to choose the required regions necessary to prepare the following compound in the…
A: Here we are required to find the Starting material for the synthesis of imine
Q: Which of the following molecules is E configuration? NC CH₂ HOH₂C CH₂ HOH₂C OMe Z. H.CH,C CH₂OH H₂C…
A: 1. Assign priority to the groups attached to each doubly bonded carbon atom according to the CIP…
Q: A gas sample has a temperature of 29∘C with an unknown volume. The same gas has a volume of 444 mL…
A:
Q: Provide the reaction (reactants -> products) with detailed mechanism for the following, with AICI3…
A: Chemical reactions 1 and 2 involve the substitution mechanism :
Q: Draw three molecules of each compound below. a. propane, CH 3 CH 2 CH 3 b. heptane, CH 3 CH 2 CH 2…
A: All these compound the molecular strucutre are as follows
Q: -OCH 3 0-00- CI- OCH, fuming H₂SO4: Br₂/FeBra: excess H30'/heat: NaNO₂/HCI/< 5°C; H₂SO4: Cul…
A:
Q: Draw a picture of and describe with at least one sentence London Dispersion Forces (LDF) between two…
A:
Q: What is the electron pair geometry, lewis structure, molecular geometry, and hybridization of NO? Is…
A:
Q: 4) What is the K, for disodium asparaginate? asparagine: FM 132.119 TZ 2.00
A: Given: To determine: kb for disodium asparaginate.
Q: Attempt 1 Calculate the cell potential for the galvanic cell in which the reaction Fe(s) + Au³+ (aq)…
A: we need to calculate cell potential for the given cell
Q: Chemistry 4. Predict the structure of all the intermediates in the synthesis of the following triol.…
A:
Q: a. Predict the structure of the unknown compound. b. Explain the fragmentation pattern and identify…
A: M = 107, Odd mass, odd no of nitrogens are present. Loss of hydrogen gives m/z = 106. Therefore the…
Q: HF is a weak acid. KOH is a strong base. One would suspect that the pH of an 0.1M solution of KF…
A: If pH is less than 7 then it is acidic in nature and when pH is greater than 7 the nature of…
Q: [Review Topics] [References] Use the References to access important values if needed for this…
A:
Q: 7. Arrange the following acids in the increasing order of their solubility in water: p- CIC6H4COOH,…
A: In the given question we have to arrange the following acids in the increasing order of their…
Q: Write the chemical formula of the following complex ions. formula name hexafluoromanganese (VII)…
A: Multiple question given here.
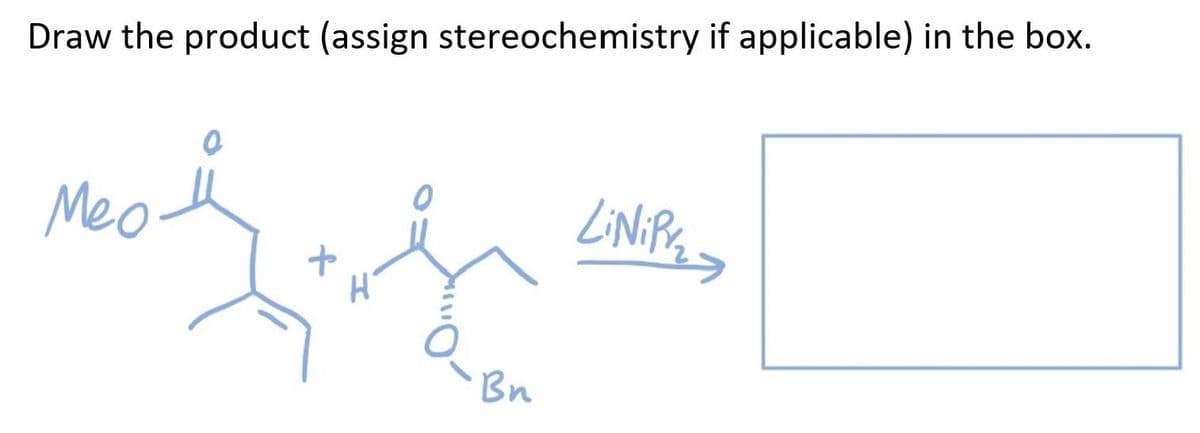
Step by step
Solved in 2 steps with 1 images

- Draw the reaction products for each SN1 reaction and indicate the appropriate stereochemistry.Name attached compound and decide which stereoisomer will react faster inan E2 elimination reaction. Explain your choice.Draw the products of attached SN1 reaction and indicate the stereochemistrywhen necessary.

