Draw the product formed in each reaction. [1] NANO2, HCI [2] CuBr а. -NH2 [1] NANO2, HCI [2] H2O b. -NH2 с. CH3O [1] NaNO2, HCI [2] HBF, -NH2 [1] CUCN [2] LIAIH4 [3] H2O d. N2* C- CI
Reactive Intermediates
In chemistry, reactive intermediates are termed as short-lived, highly reactive atoms with high energy. They rapidly transform into stable particles during a chemical reaction. In specific cases, by means of matrix isolation and at low-temperature reactive intermediates can be isolated.
Hydride Shift
A hydride shift is a rearrangement of a hydrogen atom in a carbocation that occurs to make the molecule more stable. In organic chemistry, rearrangement of the carbocation is very easily seen. This rearrangement can be because of the movement of a carbocation to attain stability in the compound. Such structural reorganization movement is called a shift within molecules. After the shifting of carbocation over the different carbon then they form structural isomers of the previous existing molecule.
Vinylic Carbocation
A carbocation where the positive charge is on the alkene carbon is known as the vinyl carbocation or vinyl cation. The empirical formula for vinyl cation is C2H3+. In the vinyl carbocation, the positive charge is on the carbon atom with the double bond therefore it is sp hybridized. It is known to be a part of various reactions, for example, electrophilic addition of alkynes and solvolysis as well. It plays the role of a reactive intermediate in these reactions.
Cycloheptatrienyl Cation
It is an aromatic carbocation having a general formula, [C7 H7]+. It is also known as the aromatic tropylium ion. Its name is derived from the molecule tropine, which is a seven membered carbon atom ring. Cycloheptatriene or tropylidene was first synthesized from tropine.
Stability of Vinyl Carbocation
Carbocations are positively charged carbon atoms. It is also known as a carbonium ion.
Draw the product formed in each attached reaction.
![Draw the product formed in each reaction.
[1] NANO2, HCI
[2] CuBr
а.
-NH2
[1] NANO2, HCI
[2] H2O
b.
-NH2
с.
CH3O
[1] NaNO2, HCI
[2] HBF,
-NH2
[1] CUCN
[2] LIAIH4
[3] H2O
d.
N2* C-
CI](/v2/_next/image?url=https%3A%2F%2Fcontent.bartleby.com%2Fqna-images%2Fquestion%2Fc6b0bf3c-0355-470f-8c1d-f1d82cad1d6b%2Fbea6f1b4-3458-4a94-9fec-67ed23b5d411%2F9nculv_processed.png&w=3840&q=75)
Since we only answer up to 3 sub-parts, we’ll answer the first 3 sub-parts. Please resubmit the question and specify the other subparts (up to 3) you’d like answered.
Given reactions:
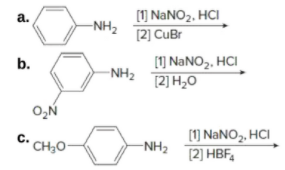
The reaction of amines and substituted amines with NaNO2, HCl leads to the formation of diazonium salt. This reaction is called Sandmeyer reaction.
Step by step
Solved in 5 steps with 3 images


