Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Andrei Straumanis
Chapter14: Elimination
Section: Chapter Questions
Problem 47CTQ
Related questions
Question
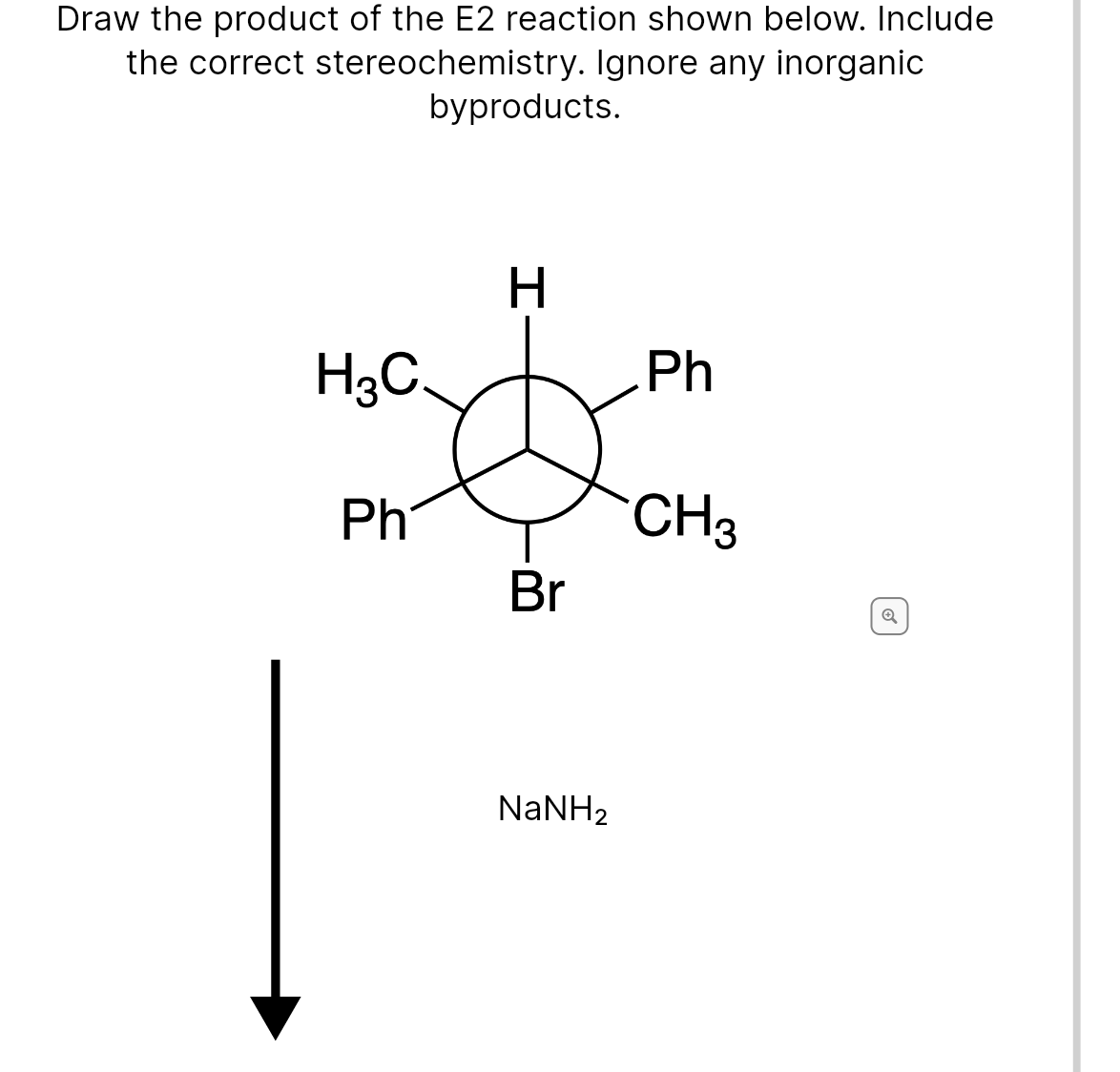
Transcribed Image Text:Draw the product of the E2 reaction shown below. Include
the correct stereochemistry. Ignore any inorganic
byproducts.
H.
H3C.
Ph
Ph
CH3
Br
NaNH2
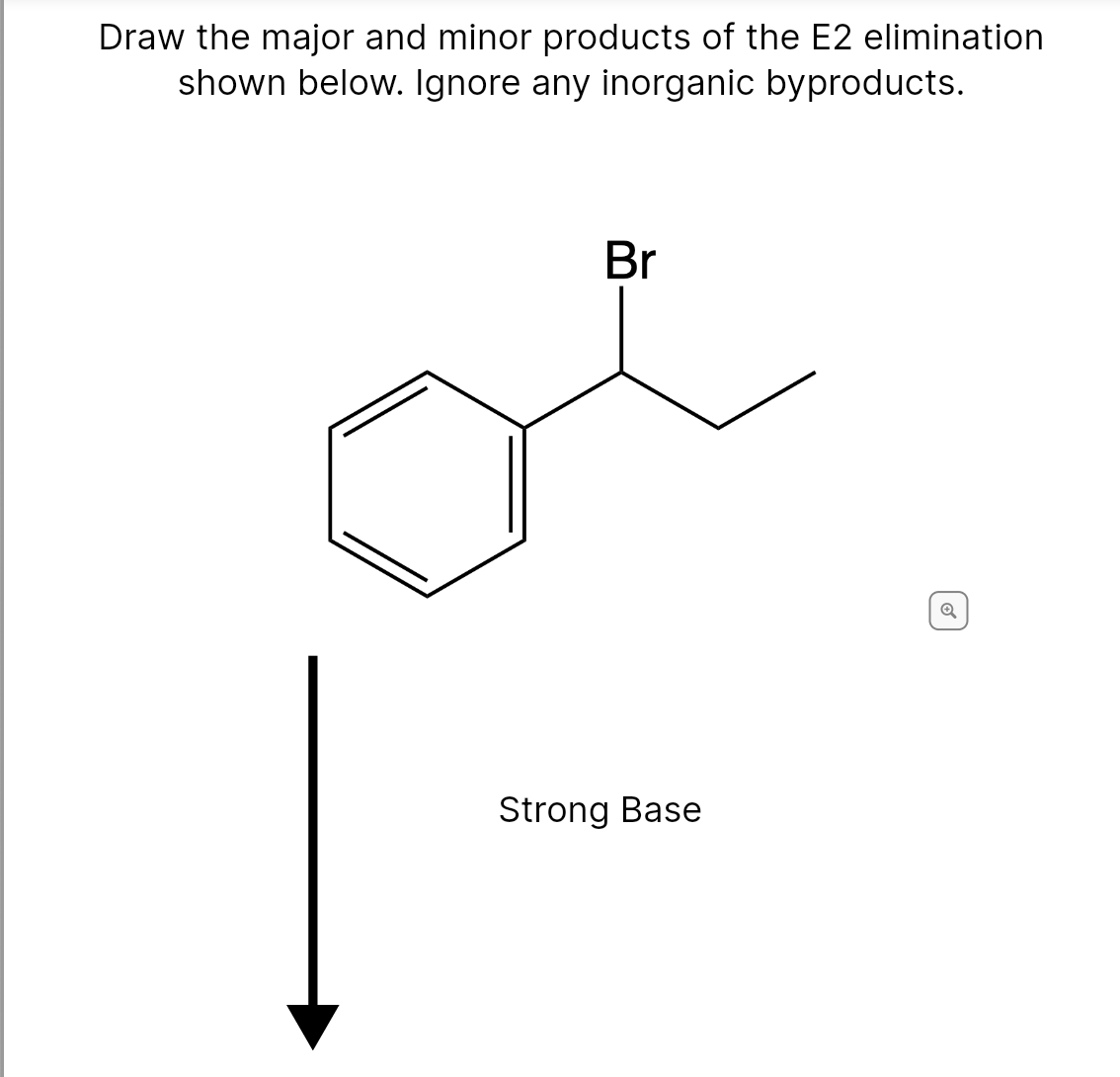
Transcribed Image Text:Draw the major and minor products of the E2 elimination
shown below. Ignore any inorganic byproducts.
Br
Strong Base
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning